Leaf Defoliation Tolerance of Root-Cutting Chili Varieties (Capsicum annum L.) Planted in Lowland Areas Related to Improve Yield and Physiological Activity
DOI:
https://doi.org/10.48048/tis.2025.9389Keywords:
Chili yield, Invertase activity, Kinetin, Reducing sugar, SucroseAbstract
Lowland chili (Capsicum annum L.) cultivation in Indonesia has lower productivity than highlands. Root-shoot growth increment can enhance chili yield. Leaf defoliation with root cutting is modification technique to optimize root-shoot growth. This research aims to determine improving physiological activity and yield of root-cutting chili varieties tolerant to leaf defoliation in lowland areas. The factorial treatments were arranged in randomized complete block design with 3 replications. The first factor was lowland chili varieties (Kencana, Lembang-1, Tanjung-2, and Ungara). The second factor was leaf defoliation on root-cutting chili, consisting of 0, 20, 40, 60 %, and control (0 % leaf defoliation without root-cutting). Analysis of variance was used to determine effect of leaf defoliation on physiological activity and chili yield. The grouping chili varieties were analyzed through principal component and cluster analysis based on yield, stress selection indices, and physiological activity. The result identified Lembang-1 and Ungara as tolerant varieties to leaf defoliation, while Kencana and Tanjung-2 were intolerant. Ungara and Lembang-1 could increase fresh weight of fruit by 39.26 and 29.30 %, respectively under 40 and 60 % leaf defoliation compared to control. The increased chili yield and root-shoot growth in tolerant varieties were caused by improving physiological activity. Root growth was driven by increased invertase activity, reducing sugar, indole acetic acid, and kinetin hormones, while shoot growth was influenced by increased sucrose, invertase activity, reducing sugar, gibberellin, and kinetin hormones. Leaf defoliation until 60 % on intolerant varieties (Tanjung-2 and Kencana) decreased chili yield by 9.53 and 19.64 %, respectively. It was concluded that 40 and 60 % leaf defoliation treatment were toleranted by root-cutting chili varieties of Ungara and Lembang-1, which increased chili yield in lowland areas.
HIGHLIGHTS
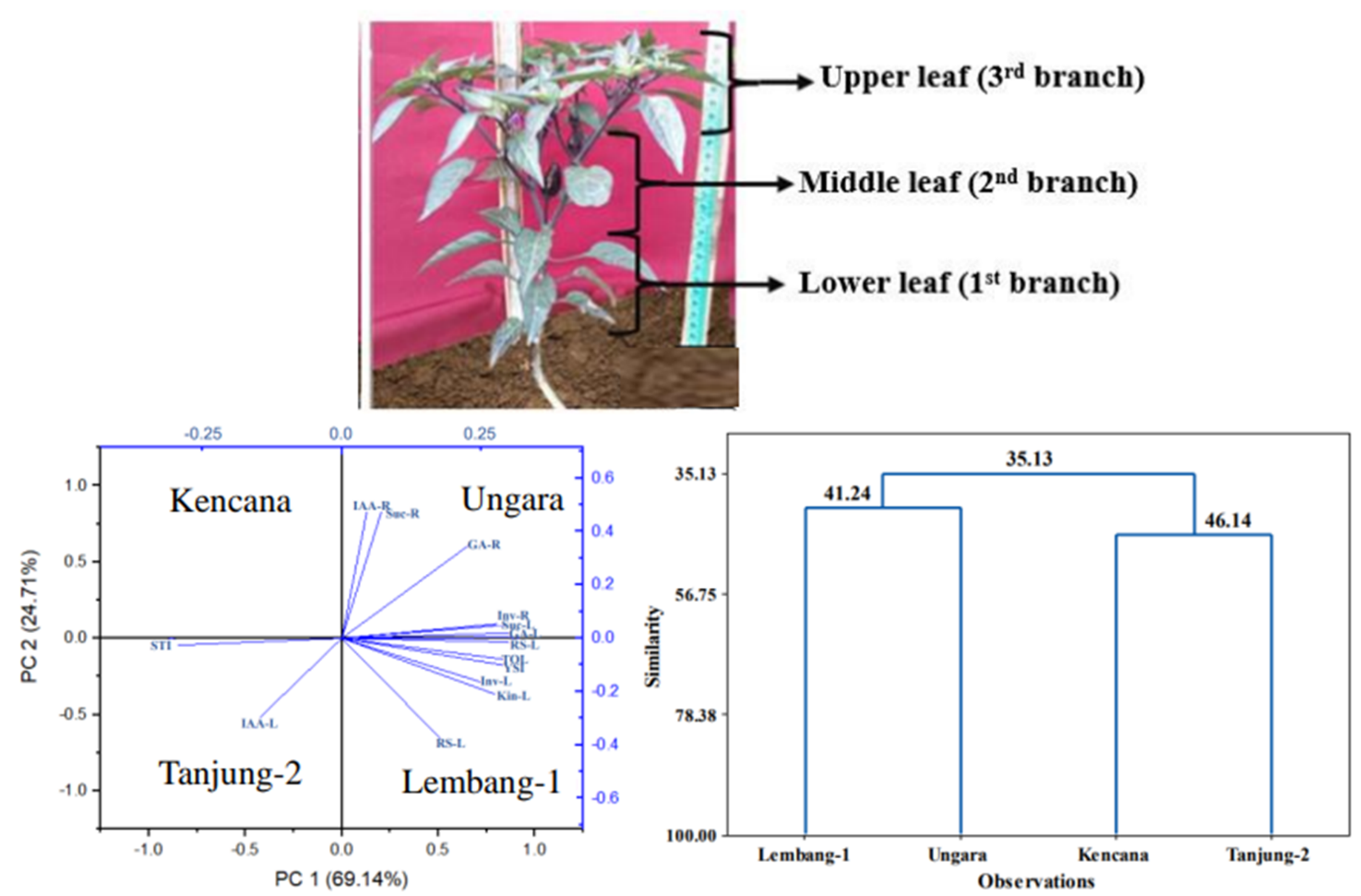
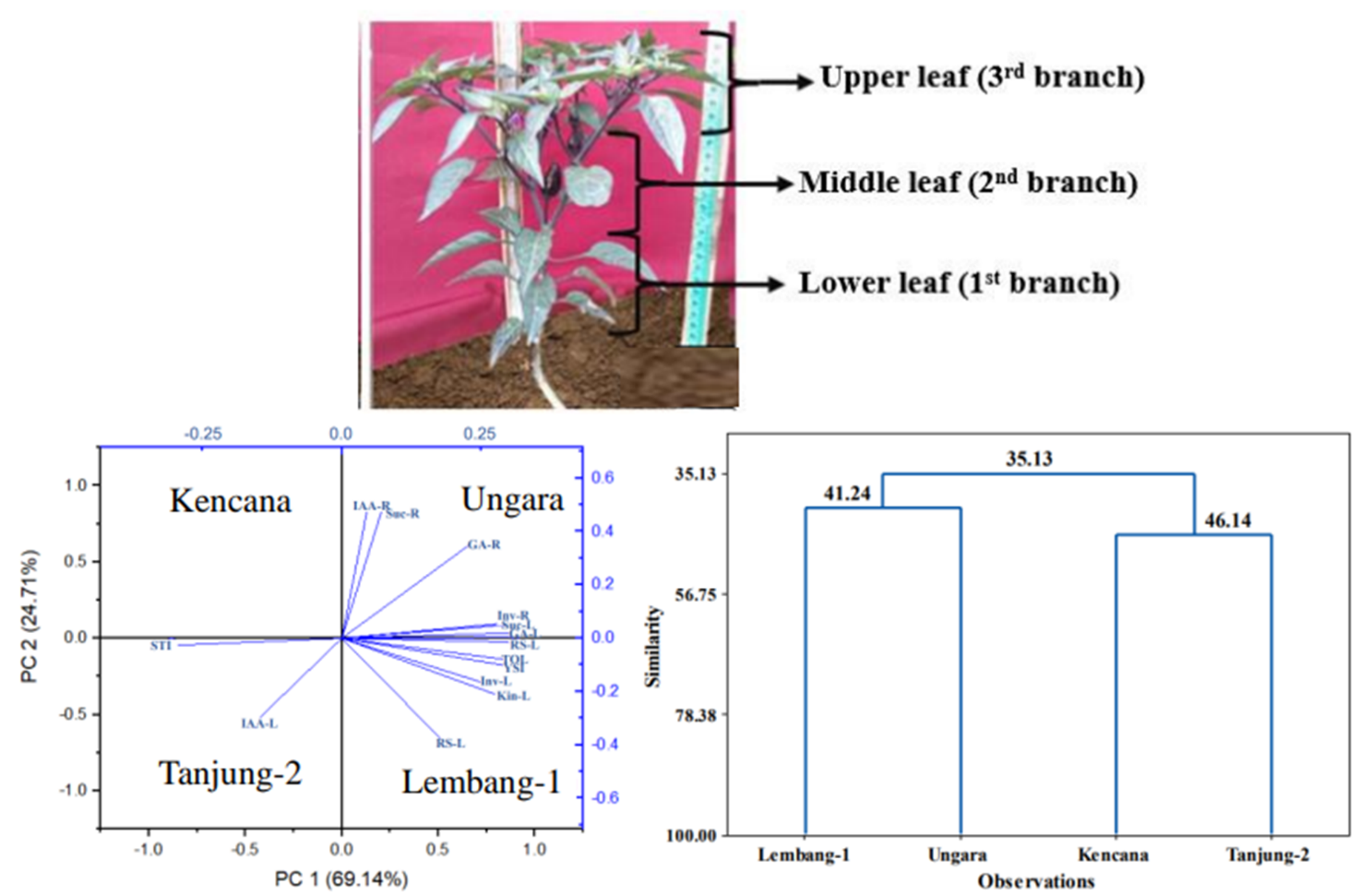
This research provides information on chili production improvement in lowland areas using root-shoot growth modification through root cutting and leaf defoliation. Response root-cutting chili yield based on stress selection indices, phytohormones, sucrose content, invertase activity, and reducing sugar under leaf defoliation was classified into tolerant (Lembang-1 and Ungara) and intolerant (Tanjung-2 and Kencana). The tolerant varieties (Lembang-1 and Ungara) could increase fresh weight of fruit by 29.30, 39.26 %, respectively under 60 and 40 % leaf defoliation compared to control (0 % leaf defoliation without root-cutting). The increased chili yield and root-shoot growth were due to physiological activity. Root growth was driven by increased invertase activity, reducing sugar, indole acetic acid, and kinetin hormones, while shoot growth was influenced by increased sucrose, invertase activity, reducing sugar, gibberellin, and kinetin hormones. Therefore, leaf defoliation technique can be used to optimize root-cutting chili production in lowland areas through improving physiological activity in root and shoot.
GRAPHICAL ABSTRACT
Downloads
References
AD Irjayanti, AS Wibowo, DA Khairunnisa, H Stiyaningsih, IM Putri, SK Areka and Z Nurfalah. Statistics of horticulture 2022, BPS Statistics Indonesia, Jakarta, Indonesia, 2023.
R Kirana, N Carsono, Y Kusandriani and D Liferdi. Improvement of yield potency of pure line pepper with heterosis fenomenon at highland on dry season. Jurnal Hortikultura 2024; 24(1), 10-15.
S Kusumaningrum, E Sulistyaningsih, R Harimurti and K Dewi. Identification of chili varieties (Capsicum annum) tolerance to root cutting based on stress selection indices and morphological traits planted in lowland area. Biodiversitas 2024; 25(5), 2063-2073.
E Magana-Hernandez, S Zuloaga-Aguilar, R Cuevas-Guzman and JG Pausas. Variation in plant belowground resource allocation across heterogeneous landscapes: implications for post-fire resprouting. American Journal of Botany 2020; 107(8), 1114-1121.
NI Zakaria, MR Ismail, Y Awang, PEM Wahab and Z Berahim. Effect of root restriction on the growth, photosynthesis rate, and source and sink relationship of chili (Capsicum annuum L.) grown in soilless culture. BioMed Research International 2020; (1), 2706937.
RL Darnell, HE Alvarado-Raya and JG Williamson. Root pruning effects on growth and yield of red raspberry. HortScience 2008; 43(3), 681-684.
P O’brien, C Collins and RD Bei. Leaf removal applied to a sprawling canopy to regulate fruit ripening in cabernet sauvignon. Plants 2021; 10(5), 1017.
K Iseki, O Olaleye and R Matsumoto. Effect of leaf thinning on shoot growth and tuber yield of white Guinea yam. Plant Production Science 2022; 25(1), 2022.
N Wang, T Ji, X Liu, Q Li, K Sairebieli, P Wu, H Song, H Wang, N Du, P Zheng and R Wang. Defoliation significantly suppressed plant growth under low light conditions in two leguminosae species. Frontiers in Plant Science 2022; 12, 777328.
AFMS Islam, MM Haque, R Tabassum and MM Islam. Effect of defoliation on growth and yield response in two tomato (Solanum lycopersicum mill.) varieties. Journal of Agronomy 2016; 15(2), 68-75.
Y Luo, C Zohner, TW Crowther, J Feng, G Hoch, P Li, AD Richardson, Y Vitasse and A Gessler. Internal physiological drivers of leaf development in trees: understanding the relationship between non-structural carbohydrates and leaf phenology. Functional Ecology 2024; 00, 1-14.
MA Raza, H Gul, F Yang, M Ahmed and W Yang. Growth rate, dry matter accumulation, and partitioning in soybean (Glycine max L.) in response to defoliation under high-rainfall conditions. Plants 2021; 10(8), 1497.
T Liu, R Huang, T Cai, Q Han and S Dong. Optimum leaf removal increases nitrogen accumulation in kernels of maize grown at high density. Scientific Reports 2017; 7, 39601.
OO Aluko, C Li, Q Wang and H Liu. Sucrose utilization for improved crop yields: A review article. International Journal of Molecular Sciences 2021; 22(9), 4704.
N Wang, Q Li, X Liu, S Yi, M Zhao, X Sun, H Song, X Peng, P Fan, Q Gao, Y Wang, L Yu, H Wang, N Du and R Wang. Plant size plays an important role in plant responses to low water availability and defoliation in two woody leguminosae species. Frontiers of Plant Science 2021; 12, 643143.
ML Martin, L Lechner, EJ Zabaleta and GL Salerno. A mitochondrial alkaline/neutral invertase isoform (A/N-InvC) functions in developmental energy-demanding processes in Arabidopsis. Planta 2013; 237(3), 813-822.
X Xu, Y Ren, C Wang, H Zhang, F Wang, J Chen, X Liu, T Zheng, M Cai, Z Zeng, L Zhou, S Zhu, W Tang, J Wang, X Guo, L Jiang, S Chen and J Wan. OsVIN2 encodes a vacuolar acid invertase that affects grain size by altering sugar metabolism in rice. Plant Cell Reports 2019; 38(10), 1273-1290.
Y Klopotek, P Franken, H Klaering, K Fischer, B Hause, M Hajirezaei and U Druege. A higher sink competitiveness of the rooting zone and invertases are involved in dark stimulation of adventitious root formation in Petunia hybrida cuttings. Plant Science 2016; 243, 10-22.
L Shen, Y Qin, Z Qi, Y Niu, Z Liu, W Liu, H He, Z Cao and Y Yang. Genome-wide analysis, expression profile, and characterization of the acid invertase gene family in pepper. International Journal of Molecular Sciences 2019; 20(1), 15.
N Wang, M Zhao, Q Li, X Liu, H Song, X Peng, H Wang, N Yang, P Fan, R Wang and N Du. Effects of defoliation modalities on plant growth, leaf traits, and carbohydrate allocation in Amorpha fruticosa L. and Robinia pseudoacacia L. seedlings. Annals of Forest Science 2020; 77, 53.
N Hoidal, S Jacobsen, A Odone and G Alandia. Defoliation timing for optimal leaf nutrition in dual-use amaranth production systems. Journal of the Science of Food and Agriculture 2020; 100(13), 4745-4755.
I Cisneros-Hernandez, E Vargas-Ortiz, ES Sanchez-Martinez, N Martinez-Gallardo, D Soto Gonzalez and JP Delano-Frier. Highest defoliation tolerance in Amaranthus cruentus plants at panicle development is associated with sugar starvation responses. Frontiers Plant Science 2021; 12, 658977.
NA Khan, M Khan, HR Ansari and Samiullah. Auxin and defoliation effects on photosynthesis and ethylene evolution in mustard. Scientia Horticulturae 2002; 96(1), 43-51.
JP Ferreira, DM Marques, D Karam, E Borghi, PC Magalhaes, KRDD Souza, SD Arantes and TCD Souza. How does early defoliation influence the morphophysiology and biochemical characteristics of maize? Plant Growth Regulator 2024; 103, 747-761.
Y Sakashita, H Kurashima, M Fukuda, H Hirano, S Lamsal, N Katayama and T Fukao. Possible roles of carbohydrate management and cytokinin in the process of defoliation–regrowth cycles in rice. International Journal of Molecular Science 2024; 25(10), 5070.
SIH Putri and M Fitriana. Growth and yield of green eggplant applied to various compositions of growing media. Journal of Suboptimal Lands 2023; 12(2), 139-151.
RA Fischer and R Maurerac. Drought resistance in spring wheat cultivars grain yield responses. Australian Journal of Agricultural Research 1978; 29(5), 897-912.
ESH Dewi, P Yudono, ETS Putra and BH Purwanto. Physiological and biochemical activities of cherelle wilt on three cocoa clones (Theobroma cacao) under two levels of soil fertilities. Biodiversitas 2020; 21(10), 187-194.
Miswar, B Sugiharto, T Handoyo and SA Made. The role of sucrose phosphate synthase (SPS) and acid invertase (AI) in sugarcane (Saccharum officinarum L.) in sucrose accumulation. Jurnal Ilmu-Ilmu Pertanian 2007; 26(4), 187-193.
GL Miller. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry 1959; 31(3), 426-528.
M Arai, H Mori and H Imaseki. Roles of sucrose-metabolizing enzymes in growth of seedlings. Purification of acid invertase from growing hypocotyls of mung bean seedlings. Plant and Cell Physiology 1991; 32(8), 1291-1298.
HF Linskens and JF Jackson. High performance liquid chromatography in plant sciences. Springer Nature, Berlin, Germany, 1987.
S Das, KW Sultana and I Chandra. In vitro micropropagation of Basilicum polystachyon (L.) Moench and identification of endogenous auxin through HPLC. Plant Cell, Tissue and Organ Culture 2020; 141, 633-641.
TL Olatunji and AJ Afolayan. The suitability of chili pepper (Capsicum annuum L.) for alleviating human micronutrient dietary deficiencies: A review. Food Science and Nutrients 2018; 6(8), 2239-2251.
N Iqbal, A Masood and NA Khan. Analyzing the significance of defoliation in growth, photosynthetic compensation and source-sink relations. Photosynthetica 2012; 50(2), 161-170.
LFM Marcelis and E Heuvelink. Concepts of modelling carbon allocation among plant organs. Springer Natrue, Dordrecht, Netherlands, 2007.
J Mariyono and S Sumarno. Chilli production and adoption of chili-based agribusiness in Indonesia. Journal of Agribusiness in Developing and Emerging Economies 2015; 5(1), 57-75.
K Lamba, M Kumar, V Singh, L Chaudhary, R Sharma, S Yashveer and MS Dalal. Heat stress tolerance indices for identification of the heat tolerant wheat genotypes. Scientific Reports 2023; 13, 10842.
SP Mishra, U Sarkar, S Taraphder, S Datta, DP Swain, R Saikhom, S Panda and M Laishram. Principal component analysis. International Journal of Livestock Research 2017; 7(5), 60-77.
A Rosati, A Paoletti, RA Hariri, A Morelli and F Famiani. Resource investments in reproductive growth proportionately limit investments in whole-tree vegetative growth in young olive trees with varying crop loads. Tree Physiology 2018; 38(9), 1267-1277.
CH Foyer and MJ Paul. Source-sink relationships. Rothamsted Research, Harpenden, 2001.
J Li, Y Guan, L Yuan, J Hou, C Wang, F Liu, Y Yang, Z Lu, G Chen and S Zhu. Effects of exogenous IAA in regulating photosynthetic capacity, carbohydrate metabolism and yield of Zizania latifolia. Scientia Horticulturae 2019; 253, 276-285.
X Wang, S Wang, Y Xue, X Ren, J Xue and X Zhang. Defoliation, not gibberellin, induces tree peony autumn reflowering regulated by carbon allocation and metabolism in buds and leaves. Plant Physiology and Biochemistry 2020; 151, 545-555.
Y Li, H Sun, TDP Protasio, PRG Hein and B Du. The mechanisms and prediction of non-structural carbohydrates accretion and depletion after mechanical wounding in slash pine (Pinus elliottii) using near-infrared reflectance spectroscopy. Plant Methods 2022; 18, 107.
W Liu, J Su, S Li, X Lang and X Huang. Non-structural carbohydrates regulated by season and species in the subtropical monsoon broad-leaved evergreen forest of Yunnan province, China. Scientific Reports 2018; 8, 1083.
RM Rosa, C Prado, G Podazza, R Interdonato, JA Gonzalez, M Hilal and FE Prado. Soluble sugars-metabolism, sensing and abiotic stress, review. Plants Signaling & Behavior 2009; 4(5), 388-393.
A Thomas, R Beena, G Laksmi, KB Soni, A Swapna and MW Viji. Changes in sucrose metabolic enzymes to water stress in contrasting rice genotypes. Plant Stress 2022; 5, 100088.
SS Sung, WJ Sheih, DR Geiger and CC Black. Growth, sucrose synthase, and invertase activities of developing Phaseolus vulgaris L. fruits. Plant, Cell and Environment 1994: 17(4), 419-426.
N Fabregas and AR Fernie. The interface of central metabolism with hormone signaling in plants. Current Biology 2021; 31(23), 1535-1548.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






