Growth Factors Optimization and Antioxidant Evaluation of β-Glucan Extracted from Lignosus rhinocerus Mycelium using Microwave-Assisted Extraction
DOI:
https://doi.org/10.48048/tis.2025.8912Keywords:
Lignosus rhinocerus, β-glucan, Mycelium cultivation, Antioxidant activity, Response surface methodology, Microwave-assisted extractionAbstract
This study aimed to optimize the cultivation of Lignosus rhinocerus mycelium at a laboratory level and evaluate the antioxidant activity of β-glucan extracted from the mycelium. Using the Response Surface Methodology (RSM) and Central Composite Design (CCD), optimal conditions for mycelium growth were identified at a temperature of 31.5 °C, 15 % potato powder, and 3.5 % glucose concentration, yielding a maximum mycelial biomass of 44.7 g/L and β-glucan production of 15.08 g/L. The β-glucan was extracted using a household microwave, significantly improving the extraction efficiency. The extracted β-glucan demonstrated notable antioxidant activity, with DPPH radical scavenging activity at approximately 53.31 % and ABTS radical scavenging activity around 47.31 %, albeit slightly lower than the 71.94 % activity of ascorbic acid. Comparative analyses highlighted the consistency in antioxidant capacities across different morphological stages, with aqueous methanol extracts of mycelium (LR-MH, LR-MT) and culture broth (LR-BH, LR-BT) showing either higher or comparable antioxidant activities to sclerotium extracts (LR-SC). Field Emission Scanning Electron Microscopy (FESEM) and Fourier Transform Infrared (FTIR) spectroscopy confirmed the structural integrity of the extracted β-glucan. The findings support the feasibility of using mycelium for β-glucan extraction, offering a sustainable alternative to traditional methods.
HIGHLIGHTS
- Optimized Mycelium Growth: Achieved optimal growth conditions for Lignosus rhinocerus mycelium, maximizing biomass and β-glucan production.
- Microwave Extraction: Used a household microwave for efficient β-glucan extraction, improving yield and reducing costs.
- Strong Antioxidant Activity: Extracted β-glucan showed significant antioxidant activity, comparable to ascorbic acid.
- Comparable Antioxidant Levels: Mycelium extracts displayed antioxidant activities similar to those of sclerotium extracts.
- Confirmed Structural Integrity: Structural integrity of β-glucan was validated using FESEM and FTIR spectroscopy.
GRAPHICAL ABSTRACT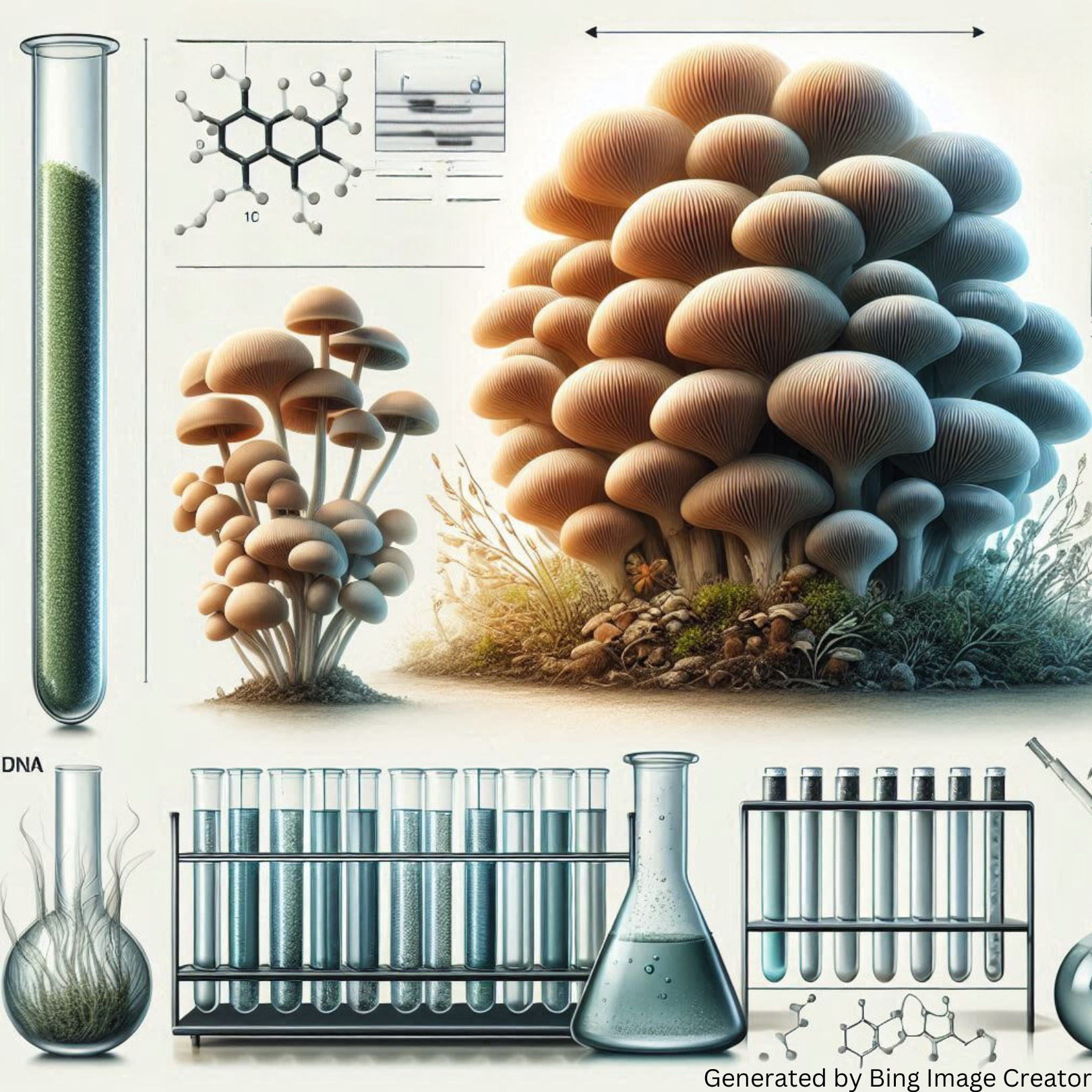
Downloads
References
D Davani-Davari, M Negahdaripour, I Karimzadeh, M Seifan, M Mohkam, SJ Masoumi, A Berenjian and Y Ghasemi. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods 2019; 8(3), 92.
F Zhu, B Du, Z Bian and B Xu. Beta-glucans from edible and medicinal mushrooms: Characteristics, physicochemical and biological activities. Journal of Food Composition and Analysis 2015; 41, 165-173.
F Zhu, B Du and B Xu. A critical review on production and industrial applications of beta-glucans. Food Hydrocolloids 2016; 52, 275-288.
J Boonyanuphap and C Hansawasdi. Spatial distribution of Beta glucan containing wild mushroom communities in subtropical dry forest, Thailand. Fungal Diversity 2010; 46, 29-42.
C Cerletti, S Esposito and L Iacoviello. Edible mushrooms and Beta-glucans: Impact on human health. Nutrients 2021; 13(7), 2195.
G Hetland, N Ohno, IS Aaberge and M Lovik. Protective effect of β-glucan against systemic Streptococcus pneumoniae infection in mice. FEMS Immunology and Medical Microbiology 2000; 27(2), 111-116.
SA Amer, GA Attia, AA Aljahmany, AK Mohamed, AA Ali, A Gouda, GN Alagmy, HM Megahed, T Saber and M Farahat. Effect of 1,3-Beta glucans dietary addition on the growth, intestinal histology, blood biochemical parameters, immune response, and immune expression of CD3 and CD20 in broiler chickens. Animals 2022; 12(22), 3197.
RP Singh and A Bhardwaj. β-glucans: A potential source for maintaining gut microbiota and the immune system. Frontiers in Nutrition 2023; 10, 1143682.
R Krishnan, YS Jang and MJ Oh. Beta glucan induced immune priming protects against nervous necrosis virus infection in sevenband grouper. Fish & Shellfish Immunology 2022; 121, 163-171.
J Richter, V Svozil, V Král, LR Dobiášová and V Vetvicka. β-glucan affects mucosal immunity in children with chronic respiratory problems under physical stress: Clinical trials. Annals of Translational Medicine 2015; 3(4), 52.
NA Mhd Omar, S Abdullah, N Abdullah, UR Kuppusamy, M Abdulla and V Sabaratnam. Lentinus squarrosulus (Mont.) mycelium enhanced antioxidant status in rat model. Drug Design, Development and Therapy 2015; 9, 5957-5964.
ESS Tan, TK Leo and CK Tan. Effect of tiger milk mushroom (Lignosus rhinocerus) supplementation on respiratory health, immunity and antioxidant status: An open-label prospective study. Scientific Reports 2021; 11, 11781.
CBS Telles, D A Sabry, J Almeida-Lima, MSSP Costa, RF Melo-Silveira, ES Trindade, GL Sassaki, E Wisbeck, SA Furlan, EL Leite and HAO Rocha. Sulfation of the extracellular polysaccharide produced by the edible mushroom Pleurotus sajor-caju alters its antioxidant, anticoagulant and antiproliferative properties in vitro. Carbohydrate Polymers 2011; 85(3), 514-521.
SR Liu and WR Zhang. Optimization of submerged culture conditions involving a developed fine powder solid seed for exopolysaccharide production by the medicinal mushroom Ganoderma lucidum. Food Science and Biotechnology 2019; 28, 1135-1145.
W Manu-Tawiah and AM Martin. Nitrogen sources and the growth response of Pleurotus ostreatus mushroom mycelium. Canadian Institute of Food Science and Technology Journal 1988; 21(2), 194-199.
BF Lau, N Abdullah, N Aminudin, HB Lee, KC Yap and V Sabaratnam. The potential of mycelium and culture broth of Lignosus rhinocerotis as substitutes for the naturally occurring sclerotium with regard to antioxidant capacity, cytotoxic effect, and low-molecular-weight chemical constituents. PLoS One 2014; 9(7), e102509.
WH Lai, M J Siti Murni, D Fauzi, OA Mazni and NM Saleh. Optimal culture conditions for mycelial growth of Lignosus rhinoceros. Mycobiology 2011; 39(2), 92-95.
Ó Benito-Román, A Martín-Cortés, MJ Cocero and E Alonso. Dissolution of (1-3),(1-4)-β-glucans in pressurized hot water: Quantitative assessment of the degradation and the effective extraction. International Journal of Carbohydrate Chemistry 2016; 2016, 2189837.
M Magnani, CM Calliari, FCD Macedo, MP Mori, IMDS Cólus and RJH Castro-Gomez. Optimized methodology for extraction of (1→3) (1→6)-β-d-glucan from Saccharomyces cerevisiae and in vitro evaluation of the cytotoxicity and genotoxicity of the corresponding carboxymethyl derivative. Carbohydrate Polymers 2009; 78(4), 658-665.
TH Gamel, ESM Abdel-Aal, NP Ames, R Duss and SM Tosh. Enzymatic extraction of beta-glucan from oat bran cereals and oat crackers and optimization of viscosity measurement. Journal of Cereal Science 2014; 59(1), 33-40.
S Phothiphiphit, W Siriwatwechakul and S Boonyarattanakalin. Microwave extraction of oligosaccharides from grey oyster mushroom by microwave facilitated hydrolysis. Science & Technology Asia 2022; 27(3), 236-246.
MN Frioui, M Shamtsyan and NA Zhilnikova. Development of new methods for isolation of mushroom beta-glucans for the use in the food industry and their comparative evaluation. Journal of Hygienic Engineering and Design 2018; 25, 107-111.
Y Ookushi, M Sakamoto and J Azuma. Optimization of microwave-assisted extraction of polysaccharides from the fruiting body of mushrooms. Journal of Applied Glycoscience 2006; 53(4), 267-272.
A Gil-Ramírez, F R Smiderle, D Morales, M Iacomini and C Soler-Rivas. Strengths and weaknesses of the aniline-blue method used to test mushroom (1→3)-β-d-glucans obtained by microwave-assisted extractions. Carbohydrate Polymers 2019; 217, 135-143.
Megazyme Ltd. β-glucan (yeast and mushroom) assay protocol, Available at: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://www.megazyme.com/documents/Assay_Protocol/K-YBGL_DATA.pdf, accessed May 2024.
M Ziadi, T Bouzaiene, S M’Hir, K Zaafouri, F Mokhtar, M Hamdi and C Boisset-Helbert. Evaluation of the efficiency of ethanol precipitation and ultrafiltration on the purification and characteristics of exopolysaccharides produced by three lactic acid bacteria. BioMed Research International 2018; 2018, 1896240.
S Baliyan, R Mukherjee, A Priyadarshini, A Vibhuti, A Gupta, RP Pandey and CM Chang. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 2022; 27(4), 1326.
IR Ilyasov, VL Beloborodov, IA Selivanova and RP Terekhov. ABTS/PP decolorization assay of antioxidant capacity reaction pathways. International Journal of Molecular Sciences 2020; 21(3), 1131.
RH Nilsson, KH Larsson, AFS Taylor, J Bengtsson-Palme, TS Jeppesen, D Schigel, P Kennedy, K Picard, FO Glöckner, L Tedersoo, I Saar, U Kõljalg and K Abarenkov. The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Research 2018; 47(D1), D259-D264.
MAN Hayhoe, T Archbold, Q Wang, X Yang and MZ Fan. Prebiotics and β-Glucan as gut modifier feed additives in modulation of growth performance, protein utilization status and dry matter and lactose digestibility in weanling pigs. Frontiers in Animal Science 2022; 3, 855846.
S Natakankitkul, P Homdok, P Wandee, T Krisdaphong and T Toida. Development of skincare cosmetic from yeast beta-glucans. Thai Journal of Pharmaceutical Sciences 2016; 40, 9-12.
J Majtan and M Jesenak. β-Glucans: Multi-functional modulator of wound healing. Molecules 2018; 23(4), 806.
G Camilli, M Bohm, AC Piffer, R Lavenir, DL Williams, B Neven, G Grateau, S Georgin-Lavialle and J Quintin. β-Glucan-induced reprogramming of human macrophages inhibits NLRP3 inflammasome activation in cryopyrinopathies. Journal of Clinical Investigation 2020; 130(9), 4561-4573.
BGJ Moerings, PD Graaff, M Furber, RF Witkamp, R Debets, JJ Mes, JV Bergenhenegouwen and C Govers. Continuous exposure to non-soluble β-glucans induces trained immunity in M-CSF-differentiated macrophages. Frontiers in Immunology 2021; 12, 672796.
WC Bak, J H Park, YA Park and KH Ha. Determination of glucan contents in the fruiting bodies and mycelia of Lentinula edodes cultivars. Mycobiology 2014; 42(3), 301-304.
BF Lau, N Abdullah and N Aminudin. Chemical composition of the tiger’s milk mushroom, Lignosus rhinocerotis (Cooke) Ryvarden, from different developmental stages. Journal of Agricultural and Food Chemistry 2013; 61(20), 4890-4897.
HEF Abdel-Raheam, SA Alrumman, SI Gadow, Mohamed H. El-Sayed, DM Hikal, AEL Hesham and MMA Ali. Optimization of Monascus purpureus for natural food pigments production on potato wastes and their application in Ice Lolly. Frontiers in Microbiology 2022; 13, 862080.
SRA Usuldin, Z Ilham, AA Jamaludin, R Ahmad and WAAQI Wan-Mohtar. Enhancing biomass-exopolysaccharides production of lignosus rhinocerus in a high-scale stirred-tank bioreactor and its potential lipid as bioenergy. Energies 2023; 16(5), 2330.
IW Hwang, BM Kim, YC Kim, SH Lee and SK Chung. Improvement in β-glucan extraction from Ganoderma lucidum with high-pressure steaming and enzymatic pre-treatment. Applied Biological Chemistry 2018; 61, 235-242.
A Hamza, A Khalad and DS Kumar. Enhanced production of mycelium biomass and exopolysaccharides of Pleurotus ostreatus by integrating response surface methodology and artificial neural network. Bioresource Technology 2024; 399, 130577.
J Harasym and R Olędzki. Comparison of conventional and microwave assisted heating on carbohydrate content, antioxidant capacity and postprandial glycemic response in oat meals. Nutrients 2018; 10(2), 207.
S Özen, A Ünlü, HN Özbek and F Göğüş. β-glucan extraction from hull-less barley by a novel approach: Microwave-assisted pressurized CO2/H2O. Food and Bioprocess Technology 2024. https://doi.org/10.1007/s11947-024-03420-1
ML Lee, NH Tan, SY Fung, CS Tan and ST Ng. The antiproliferative activity of sclerotia of Lignosus rhinocerus (Tiger Milk Mushroom). Evidence-Based Complementary and Alternative Medicine 2012; 2012(1), 697603.
Q Wang, X Sheng, A Shi, H Hu, Y Yang, L Liu, L Fei and H Liu. β-glucans: Relationships between modification, conformation and functional activities. Molecules 2017; 22(2), 257.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






