Aristolactams and Flavanones from Dasymaschalon wallichii (Hook.f. & Thomson) Jing Wang & R.M.K.Saunders and Their α-Glucosidase Inhibitory Activity
DOI:
https://doi.org/10.48048/tis.2024.8429Keywords:
Dasymaschalon wallichii, Anonaceae, α-glucosidase inhibitory activity, Aristolactam alkaloids, Flavonoids, ChalconeAbstract
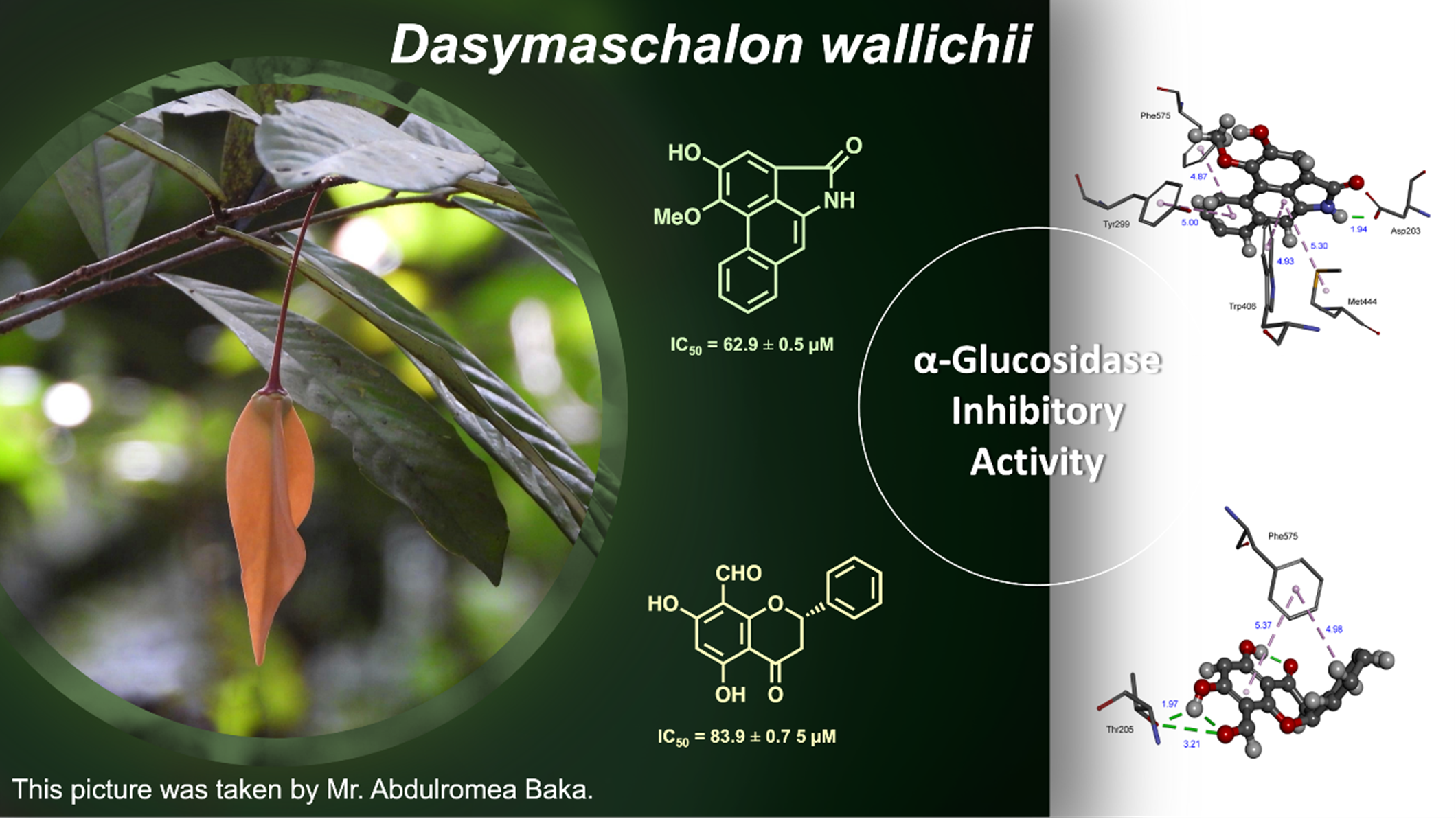
The first phytochemical investigation of the twig and leaf extracts of Dasymaschalon wallichii led to the isolation and identification of 10 compounds, including 6 airstolactam alkaloids (1-6), 3 flavonoids (7-9) and one chalcone (10). The structures were elucidated through the analysis of NMR spectroscopic data and comparisons with those reported in the literature. Compounds 1-4 and 7-10 were evaluated for their α-glucosidase inhibitory activity. Among them, aristolactam AII (1) and 8-formyl-5,7dihydroxyflavanone (8) showed α-glucosidase inhibitory activity with IC50 values of 62.9 and 83.9 μM, respectively, which are better than the positive control (acarbose, IC50 = 178.2 μM). The potential binding modes of active compounds with α-glucosidase were also analyzed by molecular docking.
HIGHLIGHTS
- The first phytochemical investigation of Dasymaschalon wallichii resulted in the isolation and identification of 10 compounds.
- Compounds 1 and 8 showed α-glucosidase inhibitory activity with IC50 values of 62.9 and 83.9 μM, respectively.
- The molecular docking of compounds 1 and 8 were studied.
GRAPHICAL ABSTRACT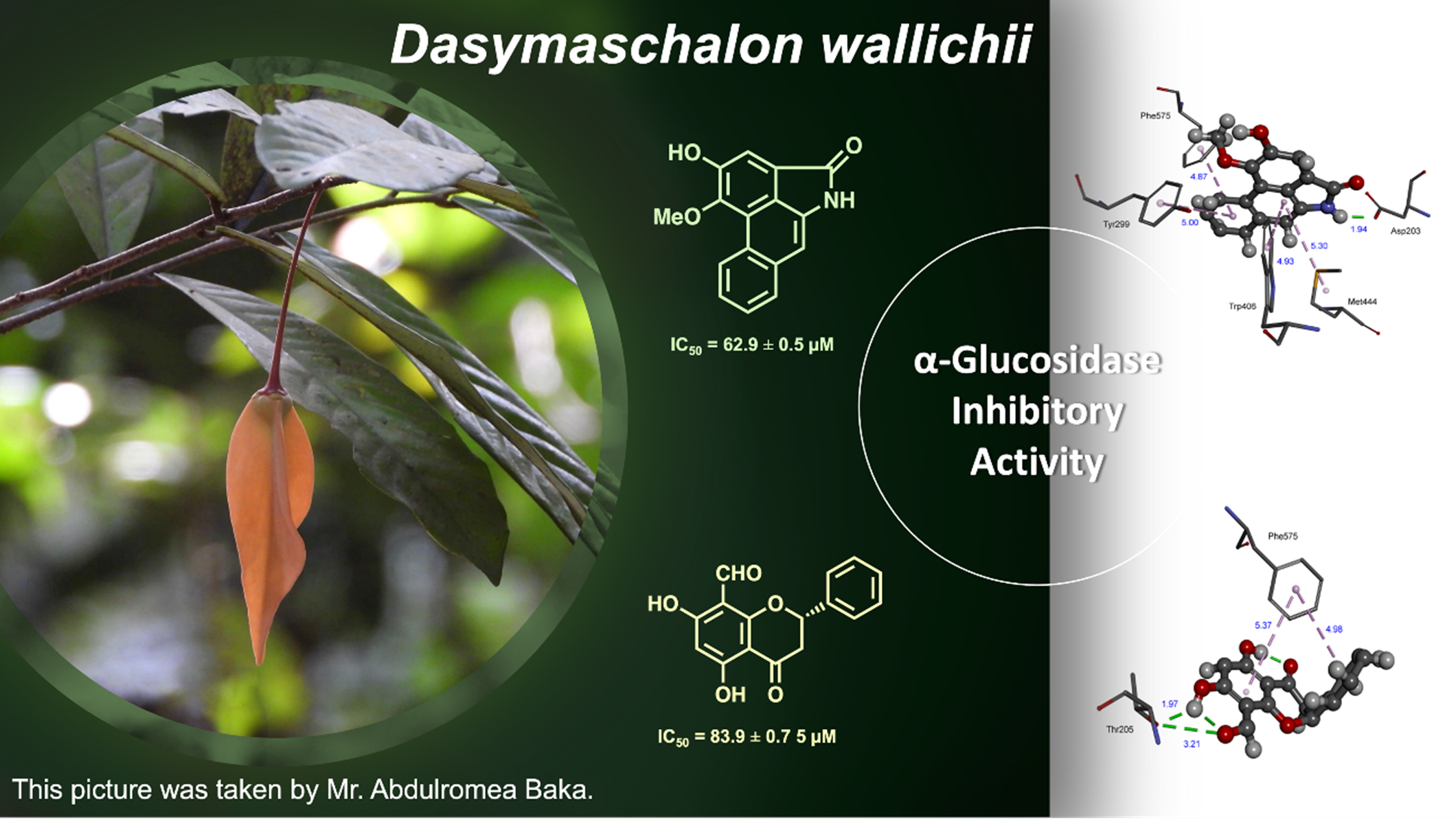
Downloads
References
S Wild, G Roglic, A Green, R Sicree and H King. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004; 27, 1047-53.
P Saeedi, I Petersohn, P Salpea, B Malanda, S Kauranga, N Unwin, S Colagiuri, L Guariguata, AA Molata, K Ogurtsova, JE Shaw, D Bright, R Williams and IDFDA Committee. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the international diabetes federation diabetes atlas, 9th edition. Diabetes Res. Clin. Pract. 2019; 157, 107843.
K Papatheodorou, M Banach, E Bekiari, M Rizzo and M Edmonds. Complications of diabetes 2017. J. Diabetes Res. 2018; 2018, 3086167.
MS Ali, M Jahangir, SSU Hussan and MI Choudhary. Inhibition of α-glucosidase by oleanolic acid and its synthetic derivatives. Phytochemistry 2002; 60, 295-9.
J Wang, P Chalermglin, RMK Saunders. The genus dasymaschalon (annonaceae) in Thailand. Syst. Bot. 2009; 34, 252-65.
U Prawat, O Chairerk, R Lenthas, A Salae and P Tuntiwachwuttikul. Two new cycloartane-type triterpenoids and one new flavanone from the leaves of Dasymaschalon dasymaschalum and their biological activity. Phytochem. Lett. 2013; 6, 286-90.
A Sinz, R Matusch, L Witte, T Santisuk, S Chaichana, V Reutrakul and S Wangcharoentrakul. Alkaloids from Dasymaschalon sootepense. Biochem. Syst. Ecol. 1998; 26, 933-4.
Z Yu, Z Niu, X Li, C Zheng, X Song, G Chen, X Song, C Han and S Wu. New phenylpropanoid and 6H-dibenzo [b, d] pyran-6-one derivatives from the stems of Dasymaschalon rostratum. Fitoterapia 2017; 118, 27-31.
A Sinz, R Matusch, T Kampchen, W Fiedler, J Schmidt, T Santisuk, S Wangcharoentrakul, S Chaichana and V Reutrakul. Novel acetogenins from the leaves of Dasymaschalon sootepense. Helv. Chim. Acta. 1998; 81, 1608-15.
S Hongthong, C Kuhakarn, T Jaipetch, S Prabpai, P Kongsaeree, P Piyachaturawat, S Jariyawat, K Suksen, J Limthongkul, A Panthong, N Nuntasean and V Reutrakul. Polyoxygenated cyclohexene derivatives isolated from Dasymaschalon sootepense and their biological activities. Fitoterapia 2015; 106, 158-66.
A Jaidee, T Promchai, K Trisuwan, S Laphookhieo, R Rattanajak, S Kamchonwongpaisan, SG Pyne and T Ritthiwigrom. Cytotoxic and antimalarial alkaloids from the twigs of Dasymaschalon obtusipetalum. Nat. Prod. Commun. 2015; 10, 1175-7.
Z Yu, C Han, X Song, G Chen and J Chen. Bioactive aporphine alkaloids from the stems of Dasymaschalon rostratum. Bioorg. Chem. 2019; 90, 103069.
Y Wang, W Chen, X Song and C Han. Aristololactam alkaloids from the roots of Dasymaschalon rostratum. Nat. Prod. Rep. 2021; 35, 1084-9.
V Suthiphasilp, W Maneerat, RJ Andersen, P Phukhatmuen, SG Pyne and S Laphookhieo. Dasymaschalolactams A-E, aristolactams from a twig extract of Dasymaschalon dasymaschalum. J. Nat. Prod. 2019; 82, 3176-80.
X He, J Chen, T Li, X Zhang, Y Guo, X Zhang, J Hu and C Geng. Nineteen new flavanol–fatty alcohol hybrids with α-glucosidase and PTP1B dual inhibition: One unusual type of antidiabetic constituent from Amomum tsao-ko. J. Agr. Food Chem. 2020; 68, 11434-48.
Z Hou, C Chen, J Ke, Y Zhang, Y Qi, S Liu, Z Yang, J Ning and G Bao. α-Glucosidase inhibitory activities and the interaction mechanism of novel spiro-flavoalkaloids from YingDe Green Tea. J. Agr. Food Chem. 2021; 70, 136-48.
P Kumboonma, T Senawong, S Saenglee and C Phaosiri. Discovery of new capsaicin and dihydrocapsaicin derivatives as histone deacetylase inhibitors and molecular docking studies. Org. Commun. 2021; 14, 133-43.
E Iqbal, LBL Lim, KA Salim, S Faizi, A Ahmed and AJ Mohamad. Isolation and characterization of aristolactam alkaloids from the stem bark of Goniothalamus velutinus (Airy Shaw) and their biological activities. J. King Saud Univ. Sci. 2018; 30, 41-8.
NM Cuong, PN Khanh, LTH Nhung, NX Ha, TT Huong, K Bauerova, YH Kim, DD Tung, TT Thuy and NTH Anh. Acetylcholinesterase inhibitory activities of some flavonoids from the root bark of Pinus krempfii Lecomte: In vitro and in silico study. J. Biomol. Struct. Dyn. 2023; 42, 4888-901.
U Prawat, D Phupornprasert, A Butsuri, Aw Salae, S Boonsri and P Tuntiwachwuttikul. Flavonoids from Friesodielsia discolor. Phytochem. Lett. 2012; 5, 809-13.
SK Talapatra, D Basu, P Chattopadhyay and B Talapatra. Aristololactams of Goniothalamus sesquipedalis wall. Revised structures of the 2-oxygenated aristololactams. Phytochemistry 1988; 27, 903-6.
P Thanuphol, Y Asami, K Shiomi, A Wongnoppavich, P Tuchinda and N Soonthornchareonnon. Marcanine G, a new cytotoxic 1-azaanthraquinone from the stem bark of Goniothalamus marcanii Craib. Nat. Prod. Res. 2018; 32, 1682-9.
S Omar, CL Chee, F Ahmed, JX Ni, H Jabar, J Huang and T Nakatsu. Phenanthrene lactams from Goniothalamus velutinus. Phytochemistry 1992; 31, 4395-7.
Y Lan, F Chang, Y Yang and Y Wu. New constituents from stems of Goniothalamus amuyon. Chem. Pharm. Bull. 2006; 54, 1040-3.
NH Sa, NT Tam, TD Quan, NTH Anh, NTT Linh, LTH Nhung, S Adrisio, DV Delfino, TV Sung and TT Thuy. Antiproliferative activity of isolated compounds from Pinus dalatensis and Pinus krempfii on acute myeloid Leukemia cells. Vietnam J. Chem. 2019; 57, 520-3.
S Gafner, JL Wolfender, S Mavi and K Hostettmann. Antifungal and antibacterial chalcones from Myrica serrata. Planta Med. 1996; 62, 67-9.
V Suthiphasilp, T Maneerat, RJ Andersen, BO Patrick, SG Pyne and S Laphookhieo. α-Glucosidase inhibitory activity of compounds isolated from the twig and leaf extracts of Desmos dumosus. Heliyon 2021; 7, e06180.
P Meesakul, C Richardson, SG Pyne and S Laphookhieo. α-Glucosidase inhibitory flavonoids and oxepinones from the leaf and twig extracts of Desmos cochinchinensis. J. Nat. Prod. 2019; 82, 741-7.
P Phukhatmuen, P Teerapongpisan, V Suthiphasilp, T Maneerat, T Duangyod, R Charoensup, S Cheepracha, J Zhu, YA Wang, S Deachathai, RJ Andersen and S Laphookhieo. Styryl lactone derivatives and aristolactam alkaloids from Goniothalamus tapis Miq. and their α-glucosidase inhibitory activity. Nat. Prod. Res. 2024. https://doi.org/10.1080/14786419.2024.2306172
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






