Minocycline Inhibits Glial Scar Formation Through CNTF Expression and Ameliorates Cognitive Impairment in Traumatic Brain Injury Rats
DOI:
https://doi.org/10.48048/tis.2024.7945Keywords:
CNTF, Cognitive impairment, Glial scar, Minocycline, Rats, Traumatic brain injuryAbstract
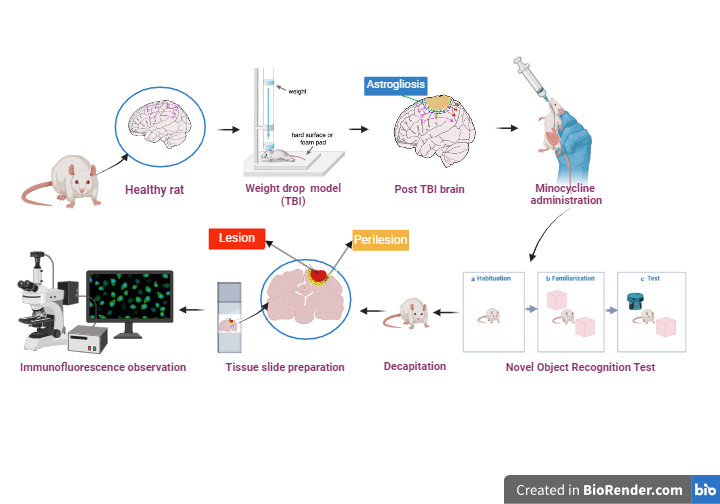
Traumatic brain injury (TBI) is a chronic condition that causes permanent disability, particularly cognitive impairment resulting from glial scar formation. Minocycline treatment inhibits glial scar formation through the Ciliary Neurotropic Factor (CNTF) pathway in multiple sclerosis. We hypothesized that minocycline could also inhibit CNTF, which would play a role in the inhibition of glial scar formation in TBI. The objective was to evaluate the role of minocycline in inhibiting glial scar formation through the CNTF signaling pathway and ameliorating cognitive impairment in TBI model rats. Male Sprague Dawley rats (n = 16) were divided into 4 groups (n = 4/group). TBI through the weight drop model is performed on day 0, followed by minocycline treatment of 25 mg/kg (MNO1 group), 50 mg/kg (MNO2 group) and 100 mg/kg (MNO3 group) given for 14 days. The NOR test is performed on day 15, followed by immunofluorescence double staining on day 16. Minocycline plays a role in inhibiting glial scar formation in TBI model rats. Minocycline inhibits the formation of CNTF with an effect proportion of 66.3 %, which plays a role in inhibiting glial scar in the perilesional area in TBI model rats. Inhibition of glial scar improves cognitive function impairment in TBI model rats. Administration of minocycline improves cognitive function in TBI model rats with an effect proportion of 46.7 %. It can be concluded that minocycline inhibits glial scar through the inhibition of CNTF expression and ameliorates cognitive impairment in a rat model of TBI.
HIGHLIGHTS
- Minocycline shown to inhibit CNTF formation in TBI model rats
- CNTF inhibition can inhibit the formation of glial scar in TBI model rats.
- Inhibition of glial scar formation can improve cognitive function impairment in TBI model rats.
- Minocycline administration can improve cognitive function through the NOR test.
GRAPHICAL ABSTRACT

Downloads
References
D Cortes and MF Pera. The genetic basis of inter-individual variation in recovery from traumatic brain injury. NPJ Regenerative Med. 2021; 6, 5.
Y Zhou, A Shao, Y Yao, S Tu, Y Deng and J Zhang. Dual roles of astrocytes in plasticity and reconstruction after traumatic brain injury. Cell Comm. Signaling 2020; 18, 62.
H Xu, G Tan, S Zhang, H Zhu, F Liu, C Huang, F Zhang and Z Wang. Minocycline reduces reactive gliosis in the rat model of hydrocephalus. BMC Neurosci. 2012; 13, 148.
J Meythaler, J Fath, D Fuerst, H Zokary, K Freese, HB Martin, J Reineke, J Peduzzi-Nelson and PT Roskos. Safety and feasibility of minocycline in treatment of acute traumatic brain injury. Brain Inj. 2019; 33, 679-89.
ZY Cai, Y Yan and R Chen. Minocycline reduces astrocytic reactivation and neuroinflammation in the hippocampus of a vascular cognitive impairment rat model. Neurosci. Bull. 2010; 26, 28-36.
K Erning and T Segura. Materials to promote recovery after stroke. Curr. Opin. Biomed. Eng. 2020; 14, 9-17.
S Pourkhodadad, S Oryan, G Kaka and SH Sadraie. Neuroprotective effects of combined treatment with minocycline and olfactory ensheathing cells transplantation against inflammation and oxidative stress after spinal cord injury. Cell J. 2018; 21, 220-8.
JW Squair, I Ruiz, AA Phillips, MMZ Zheng, ZK Sarafis, R Sachdeva, R Gopaul, J Liu, W Tetzlaff, CR West and AV Krassioukov. Minocycline reduces the severity of autonomic dysreflexia after experimental spinal cord injury. J. Neurotrauma 2018; 35, 2861-71.
SY Ng and AYW Lee. Traumatic brain injuries: Pathophysiology and potential therapeutic targets. Front. Cell. Neurosci. 2019; 13, 528.
M Leibinger, A Andreadaki, H Diekmann and D Fischer. Neuronal STAT3 activation is essential for CNTF- and inflammatory stimulation-induced CNS axon regeneration. Cell Death Dis. 2013; 4, e805.
DW Wardhana, HS Yudhanto, W Riawan, H Khotimah, HK Permatasari, TA Nazwar and N Nurdiana. Modification of the height of a weight drop traumatic brain injury model that causes the formation of glial scar and cognitive impairment in rats. BMC Neurol. 2023; 23, 439.
K Ceyzériat, L Abjean, MA Carrillo-de Sauvage, LB Haim and C Escartin. The complex STATes of astrocyte reactivity: How are they controlled by the JAK-STAT3 pathway? Neuroscience 2016; 330, 205-18.
M Moattari, F Moattari, G Kaka, HM Kouchesfahani, SH Sadraie and M Naghdi. Comparison of neuroregeneration in central nervous system and peripheral nervous system. Otorhinolaryngol. Head Neck Surg. 2018; 3, 1-3.
H Wang, G Song, H Chuang, C Chiu, A Abdelmaksoud, Y Ye and L Zhao. Portrait of glial scar in neurological diseases. Int. J. Immunopathol. Pharmacol. 2018; 31, 2058738418801406.
MV Sofroniew and HV Vinters. Astrocytes: Biology and pathology. Acta Neuropathol. 2010; 119, 7-35.
T Tanaka, K Murakami, Y Bando and S Yoshida. Minocycline reduces remyelination by suppressing ciliary neurotrophic factor expression after cuprizone-induced demyelination. J. Neurochem. 2013; 127, 259-70.
L Rachmany, D Tweedie, V Rubovitch, QS Yu, Y Li, JY Wang, CG Pick and NH Greig. Cognitive impairments accompanying rodent mild traumatic brain injury involve p53-dependent neuronal cell death and are ameliorated by the tetrahydrobenzothiazole PFT-α. PLos One 2013; 8, e79837.
K Eakin, R Baratz-Goldstein, CG Pick, O Zindel, CD Balaban, ME Hoffer, M Lockwood, J Miller and BJ Hoffer. Efficacy of N-acetyl cysteine in traumatic brain injury. PLos One 2014; 9, e90617.
S Edut, V Rubovitch, M Rehavi, S Schreiber and CG Pick. A study on the mechanism by which MDMA protects against dopaminergic dysfunction after minimal traumatic brain injury (mTBI) in mice. J. Mol. Neurosci. 2014; 54, 684-97.
S Schreiber, R Lin, L Haim, R Baratz-Goldstien, V Rubovitch, N Vaisman and CG Pick. Enriched environment improves the cognitive effects from traumatic brain injury in mice. Behav. Brain Res. 2014; 271, 59-64.
R Baratz, D Tweedie, JY Wang, V Rubovitch, W Luo, BJ Hoffer, NH Greig and CG Pick. Transiently lowering tumor necrosis factor-α synthesis ameliorates neuronal cell loss and cognitive impairments induced by minimal traumatic brain injury in mice. J. Neuroinflammation 2015; 12, 45.
T Cheng, B Yang, D Li, S Ma, Y Tian, R Qu, W Zhang, Y Zhang, K Hu, F Guan and J Wang. Wharton’s jelly transplantation improves neurologic function in a rat model of traumatic brain injury. Cell. Mol. Neurobiol. 2015; 35, 641-9.
R Baratz-Goldstein, H Deselms, LR Heim, L Khomski, BJ Hoffer, D Atlas and CG Pick. Thioredoxin-mimetic-peptides protect cognitive function after mild traumatic brain injury (mTBI). PLos One 2016; 11, e0157064.
X Ji, D Peng, Y Zhang, J Zhang, Y Wang, Y Gao, N Lu and P Tang. Astaxanthin improves cognitive performance in mice following mild traumatic brain injury. Brain Res. 2017; 1659, 88-95.
A Lesniak, CG Pick, A Misicka, AW Lipkowski and M Sacharczuk. Biphalin protects against cognitive deficits in a mouse model of mild traumatic brain injury (mTBI). Neuropharmacology 2016; 101, 506-18.
M Bader, Y Li, D Tweedie, NA Shlobin, A Bernstein, V Rubovitch, LB Tovar-y-Romo, RD DiMarchi, BJ Hoffer, NH Greig and CG Pick. Neuroprotective effects and treatment potential of incretin mimetics in a murine model of mild traumatic brain injury. Front. Cell Dev. Biol. 2020; 7, 356.
D Qubty, K Frid, M Har-Even, V Rubovitch, R Gabizon and CG Pick. Nano-PSO administration attenuates cognitive and neuronal deficits resulting from traumatic brain injury. Molecules 2022; 7, 2725.
DW Simon, MJ McGeachy, H Bayır, RSB Clark, DJ Loane and PM Kochanek. The far-reaching scope of neuroinflammation after traumatic brain injury. Nat. Rev. Neurol. 2017; 13, 171-91.
E Kovesdi, A Kamnaksh, D Wingo, NE Grunberg, JB Long, CE Kasper and DV Agoston. Acute minocycline treatment mitigates the symptoms of mild blast-induced traumatic brain injury. Front. Neurol. 2012; 3, 18424.
TI Lam, D Bingham, TJ Chang, CC Lee, J Shi, D Wang, S Massa, RA Swanson and J Liu. Beneficial effects of minocycline and botulinum toxin-induced constraint physical therapy following experimental traumatic brain injury. Neurorehabil. Neural Repair 2013; 27, 889-99.
A Kumar, P Rinwa and H Dhar. Microglial inhibitory effect of ginseng ameliorates cognitive deficits and neuroinflammation following traumatic head injury in rats. Inflammopharmacology 2014; 22, 155-67.
MA Sangobowale, NM Grin’kina, K Whitney, E Nikulina, KS Laurent-Ariot, JS Ho, N Bayzan and PJ Bergold. Minocycline plus N-acetylcysteine reduce behavioral deficits and improve histology with a clinically useful time window. J. Neurotrauma 2018; 35, 907-17.
P Cooke, H Janowitz and SE Dougherty. Neuronal redevelopment and the regeneration of neuromodulatory axons in the adult mammalian central nervous system. Front. Cell. Neurosci. 2022; 16, 872501.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






