Fine-Tuning the Degree of Negative Charge and Carboxylate Grafting Density of Polymer-Coated Magnetite Nanoparticle
DOI:
https://doi.org/10.48048/tis.2023.6384Keywords:
Nanoparticle, Magnetite, ATRP, Polyelectrolyte, PAA, PEGMA, pH ResponsiveAbstract
The surface of magnetite nanoparticle (MNP) was modified with poly(acrylic acid)(PAA)-poly(ethylene glycol) methyl methacrylate (PEGMA) (co)polymer to obtain water dispersible MNP with pH sensitive surfaces via atom transfer radical polymerization (ATRP) of tert-butyl acrylate (t-BA), followed by the t-BA group deprotection. Molar compositions of the (co)polymer were systematically varied, namely 100:0, 75:25, 50:50 and 25:75 of P(t-BA)/PEGMA, respectively, such that the grafting density of carboxylate groups on the MNP surface after the deprotection can be fine-tuned. Transmission electron microscopy (TEM) indicated that the MNP were spherical (ca.5-12 nm in diameter) with some nanoclustering observed. The roles of PAA and PEGMA affecting the hydrodynamic size and zeta potential of the nanocomposites observed via photocorrelation spectroscopy (PCS) were discussed. The percentages of the polymeric composition in the nanocomposites in each step of the reaction were determined via thermogravimetric analysis (TGA) and their magnetic properties were studied via vibrating sample magnetometry (VSM). These novel nanocomposites with pH sensitive surface and magnetically guidable properties might be advantageous for further conjugations either by covalent or ionic bonding with bioentities.
HIGHLIGHTS
- Magnetite nanoparticle (MNP) coated poly(acrylic acid)(PAA)-poly(ethylene glycol) methyl methacrylate (PEGMA) (co)polymer were fabricated via ATRP
- The ratio of t-BA to PEGMA used in the copolymerization was studied
- The nanocomposites showed pH sensitive surface and magnetically guidable properties
GRAPHICAL ABSTRACT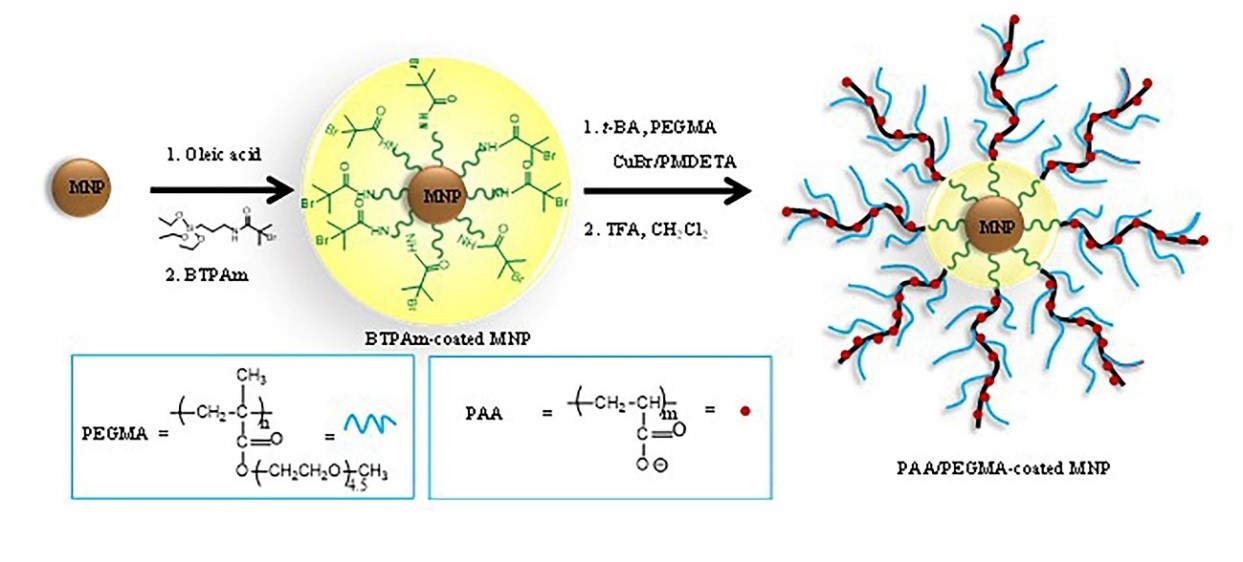
Downloads
References
T Theppaleak, B Rutnakornpituk, U Wichai, T Vilaivan and M Rutnakornpituk. Magnetite nanoparticle with positively charged surface for immobilization of peptide nucleic acid and deoxyribonucleic acid. J. Biomed. Nanotechnology 2013; 9, 1509-20.
BW Chen, YC He, SY Sung, TTH Le, CL Hsieh, JY Chen, ZH Wei and DJ Yao. Synthesis and characterization of magnetic nanoparticles coated with polystyrene sulfonic acid for biomedical applications. Sci. Tech. Adv. Mater. 2020; 21, 471-81.
X Guan, S Yan, Q Zeng, Z Xu, Y Chen and H Fan. Polyacrylic acid-grafted magnetite nanoparticles for remediation of Pb(II)-contained water. Fibers Polymers 2016; 17, 1131-9.
S Meerod, B Rutnakornpituk, U Wichai and M Rutnakornpituk. Hydrophilic magnetic nanoclusters with thermo-responsive properties and their drug controlled release. J. Magnetism Magnetic Mater. 2015; 392, 83-90.
N Deepuppha, A Thongsaw, B Rutnakornpituk, WC Chaiyasith and M Rutnakornpituk. Alginate-based magnetic nanosorbent immobilized with aptamer for selective and high adsorption of Hg2+ in water samples. Environ. Sci. Pollut. Res. 2020; 27, 12030-8.
N Deepuppha, S Khadsai, B Rutnakornpituk, F Kielar and M Rutnakornpituk. Reusable pectin‐coated magnetic nanosorbent functionalized with an aptamer for highly selective Hg2+ detection. Polym. Adv. Technol. 2021; 32, 2207-17.
B Thong-On, B Rutnakornpituk, U Wichai and M Rutnakornpituk. Magnetite nanoparticle coated with amphiphilic bilayer surfactant of polysiloxane and poly (poly (ethylene glycol) methacrylate). J. Nanoparticle Res. 2012; 14, 953.
U Mahanitipong and M Rutnakornpituk. Palladium‐immobilized polymer‐coated magnetic nanocomposites as reusable catalysts for the reduction of 4‐nitrophenol. Polym. Int. 2022; 71, 1119-26.
S Stafford, C Garnier and YK Gun’ko. Polyelectrolyte-stabilised magnetic-plasmonic nanocomposites. Nanomaterials 2018; 8, 1044.
D Patiño-Ruiz, L Sanchez-Botero, J Hinestroza and A Herrera. Modification of cotton fibers with magnetite and magnetic core-shell mesoporous silica nanoparticles. Phys. Status Solidi Appl. Mater. 2018; 215, 1800266.
D Chełminiak, M Ziegler-Borowska and H Kaczmarek. Synthesis of magnetite nanoparticles coated with poly(acrylic acid) by photopolymerization. Mater. Lett. 2016; 164, 464-7.
R Wang, J lin, SH Huang, QY Wang, Q Hu, S Peng, LN Wu and QH Zhou. Disulfide cross-linked poly(methacrylic acid) iron oxide nanoparticles for efficiently selective adsorption of Pb(II) from aqueous solutions. ACS Omega 2021; 6, 976-87.
AV Dobrynin and M Rubinstein. Theory of polyelectrolytes in solutions and at surfaces. Prog. Polym. Sci. 2005; 30, 1049-118.
Z Mozafari, B Massoumi and M Jaymand. A novel stimuli-responsive magnetite nanocomposite as de novo drug delivery system. Polym. Plast. Technol. Mater. 2019; 58, 405-18.
D Niu, X Liu, Y Li, Z Ma, W Dong, S Chang, W Zhao, J Gu, S Zhang and J Shi. Fabrication of uniform, biocompatible and multifunctional PCL-b-PAA copolymer-based hybrid micelles for magnetic resonance imaging. J. Mater. Chem. 2011; 21, 13825-31.
A Dolatkhah and LD Wilson. Magnetite/polymer brush nanocomposites with switchable uptake behavior toward methylene blue. ACS Appl. Mater. Inter. 2016; 8, 5595-607.
S Wan, Y Zheng, Y Liu, H Yan and K Liu. Fe3O4 nanoparticles coated with homopolymers of glycerol mono (meth) acrylate and their block copolymers. J. Mater. Chem. 2005; 15, 3424-30.
E Illés, E Tombácz, M Szekeres, IY Tóth, Á Szabó and B Iván. Novel carboxylated PEG-coating on magnetite nanoparticles designed for biomedical applications. J. Magnetism Magnetic Mater. 2015; 380, 132-9.
M Rutnakornpituk, N Puangsin, P Theamdee, B Rutnakornpituk and U Wichai. Poly (acrylic acid)-grafted magnetic nanoparticle for conjugation with folic acid. Polymer 2011; 52, 987-95.
H Derakhshankhah, B Haghshenas, M Eskandani, R Jahanban-Esfahlan, S Abbasi-Maleki and M Jaymand. Folate-conjugated thermal-and pH-responsive magnetic hydrogel as a drug delivery nano-system for “smart” chemo/hyperthermia therapy of solid tumors. Mater. Today Commun. 2022; 30, 103148.
Á Szabó, I Szanka, G Tolnai, G Szark and B Iván. LCST-type thermoresponsive behaviour of interpolymer complexes of well-defined poly (poly(ethylene glycol) methacrylate)s and poly(acrylic acid) synthesized by ATRP. Polymer 2017; 111, 61-6.
M Guo, Y Yan, H Zhang, H Yan, Y Cao, K Liu, S Wan, J Huang and W Yue. Magnetic and pH-responsive nanocarriers with multilayer core-shell architecture for anticancer drug delivery. J. Mater. Chem. 2008; 18, 5104-12.
E Illés, M Szekeres, IY Tóth, Á Szabó, B Iván, R Turcu, L Vékás, I Zupkó, G Jaics and E Tombácz. Multifunctional PEG-carboxylate copolymer coated superparamagnetic iron oxide nanoparticles for biomedical application. J. Magnetism Magnetic Mater. 2018; 451, 710-20.
E Illés, M Szekeres, IY Tóth, K Farkas, I Földesi, Á Szabó, B Iván and E Tombácz. PEGylation of superparamagnetic iron oxide nanoparticles with self-organizing polyacrylate-PEG brushes for contrast enhancement in MRI diagnosis. Nanomaterials 2018; 8, 776.
M Guo, C Que, C Wang, X Liu, H Yan and K Liu. Multifunctional superparamagnetic nanocarriers with folate-mediated and pH-responsive targeting properties for anticancer drug delivery. Biomaterials 2011; 32, 185-94.
P Kanhakeaw, B Rutnakornpituk, U Wichai and M Rutnakornpituk. Surface-initiated atom transfer radical polymerization of magnetite nanoparticles with statistical poly(tert-butyl acrylate)-poly(poly (ethylene glycol) methyl ether methacrylate) Copolymers. J. Nanomaterials 2015; 2015, 1-10.
G Gratzl, C Paulik, S Hild, JP Guggenbichler and M Lackner. Antimicrobial activity of poly(acrylic acid) block copolymers. Mater. Sci. Eng. C 2014; 38, 94-100.
SD Sütekin and O Güven. Preparation of poly(tert-butyl acrylate)-poly(acrylic acid) amphiphilic copolymers via radiation-induced reversible addition-fragmentation chain transfer mediated polymerization of tert-butyl acrylate. Polym. Int. 2020; 69, 693-701.
A Musyanovych, R Rossmanith, C Tontsch and K Landfester. Effect of hydrophilic comonomer and surfactant type on the colloidal stability and size distribution of carboxyl-and amino-functionalized polystyrene particles prepared by miniemulsion polymerization. Langmuir 2007; 23, 5367-76.
S Berger, ASynytska, L Ionov, KJ Eichhorn and M Stamm. Stimuli-responsive bicomponent polymer janus particles by “grafting from”/“grafting to” approaches. Macromolecules 2008; 41, 9669-76.
K Chen, Y Zhu, Y Zhang, L Li, Y Lu and X Guo. Synthesis of magnetic spherical polyelectrolyte brushes. Macromolecules 2011; 44, 632-9.
J Hang, L Shi, X Feng and L Xiao. Electrostatic and electrosteric stabilization of aqueous suspensions of barite nanoparticles. Pow. Technol. 2009; 192, 166-70.
Z Sui, JA Jaber and JB Schlenoff. Polyelectrolyte complexes with pH-tunable solubility. Macromolecules 2006; 39, 8145-52.
H Kong, C Gao and D Yan. Constructing amphiphilic polymer brushes on the convex surfaces of multi-walled carbon nanotubes by in situ atom transfer radical polymerization. J. Mater. Chem. 2004; 14, 1401-5.
RD Braun. Introduction to chemical analysis. Britannica, Illinois, 1982.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






