The Influence of Para-Substituent on the Molecular Properties and Biological Activities of Aroylhydrazone Derivatives: DFT Analysis and Anti-Cervical Cancer Evaluation Through In Silico and In Vitro Studies
DOI:
https://doi.org/10.48048/tis.2026.12603Keywords:
Aroylhydrazone, Characterization, Density functional theory, ADMET, Molecular dockingAbstract
The synthesis of four aroylhydrazone-derived compounds was successfully conducted via the condensation reaction between benzoyl hydrazine and four different aromatic aldehydes. The resulting comppunds were identified as 4-Methoxybenzylidene ben-zo hydrazide (L1), 4-hidroksi-benzaldehid benzoyl hydrazone (L2), 4-methylbenzylidene benzohydrazide (L3) and 4-nitrobenziliden - 2 - benzoyl hydrazine (L4). Their spectroscopi cal and physicochemical characteristics were analyzed through UV-Vis, IR, 1H NMR, and 13C NMR instruments. Structural optimization was subsequently performed using density functional theory (DFT), followed by theoretical evaluations of UV-Vis and IR spectral modeling, reactivity and stability, and several quantum chemical descriptors. It was revealed that the theoretical results agreed with the experimental yields. The results of DFT analysis showed that L1 (ΔEGAP = 4.58 eV) and L3 (ΔEGAP = 4.42 eV) have high molecular stability and reactivity. These parameters indicate L1 and L3 as candidates for anticancer compounds. Besides, the antioxidant and cytotoxicity of the prepared compounds were observed to elucidate their specific mechanisms. L1 and L3 showed moderate anticancer activity with IC50 values of 82.12 and 42.64 µg/mL, respectively. In addition, insilico analysis, indicated strong binding affinities for L1 and L4, with docking binding energies of −9.37 and −10.57 kcal/mol. The Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) predictions and pharmacokinetic parameters of the samples further supported their favorable oral bioavailability.
HIGHLIGHTS
- Four para-substituted aroylhydrazones (L1 - L4) have been successfully synthesized via condensation reactions.
- DFT analysis showed that L1 (ΔEGAP = 4.58 eV) and L3 (ΔEGAP = 4.42 eV) have high molecular stability and reactivity.
- 4-Methoxybenzylidene benzo hydrazide (L1) and 4-methylbenzylidene benzoh ydrazide (L3) had moderate cytotoxicity toward HeLa cells.
- 4-Nitrobenzylidene-benzoyl hydrazine (L4) exhibited strong CDK2 inhibition (ΔE = −10.57 kcal/mol).
- All compounds met Lipinski rules with low predicted toxicity.
GRAPHICAL ABSTRACT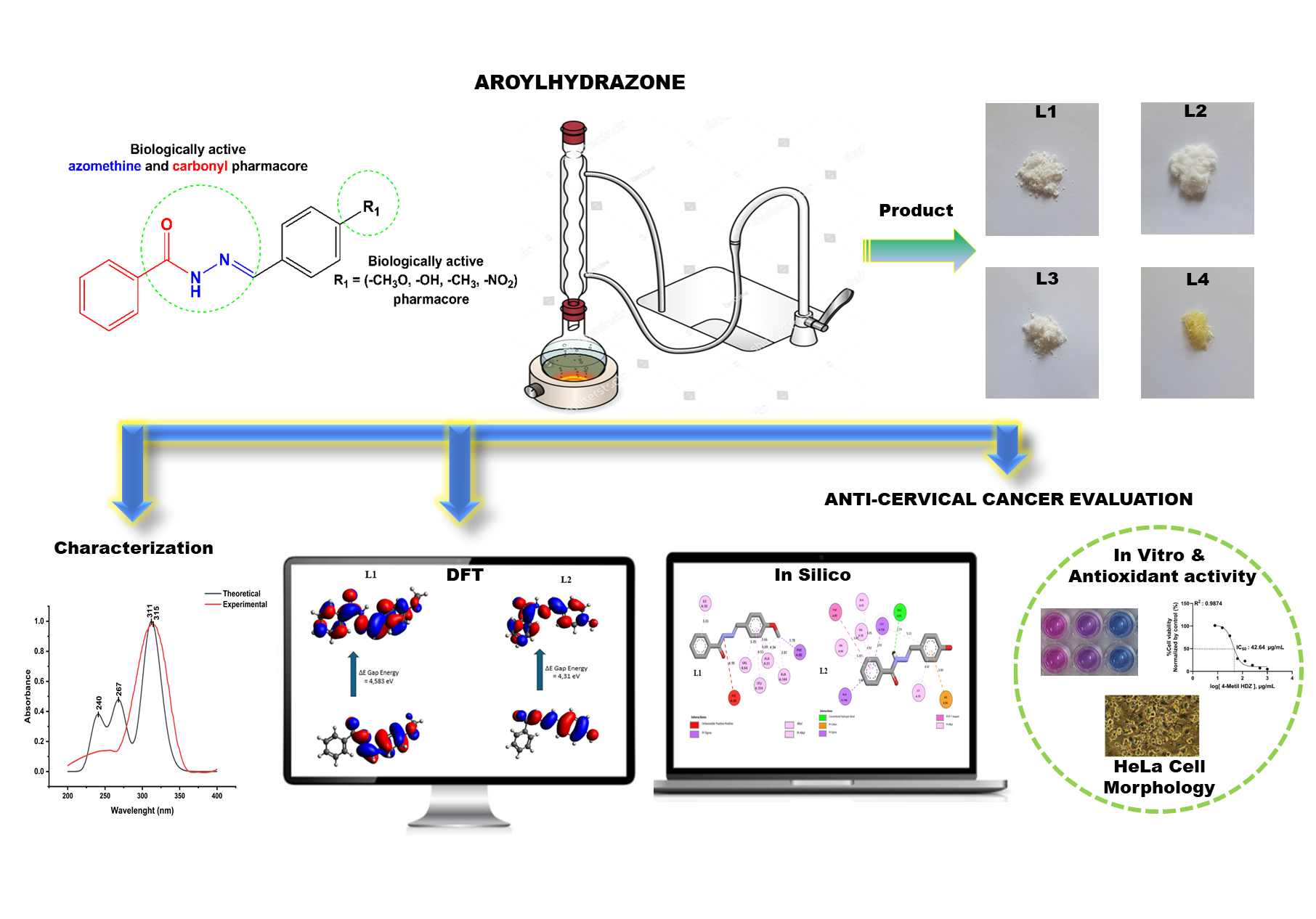
Downloads
References
J Pisk, I. Đilović, T Hrenar, D Cvijanović, G Pavlović and V Vrdoljak. Effective methods for the synthesis of hydrazones, quinazolines, and Schiff bases: Reaction monitoring using a chemometric approach. RSC Advances 2020; 10(63), 38566-38577.
C Arunagiri, AG Anitha, A Subashini and S Selvakumar. Synthesis, X-ray crystal structure, vibrational spectroscopy, DFT calculations, electronic properties and Hirshfeld analysis of (E) -4-Bromo-N’-(2,4-dihydroxy-benzylidene) benzohydrazide. Journal of Molecular Structure 2018; 1163, 368-378.
VFS Pape, S Tóth, A Füredi, K Szebényi, A Lovrics, P Szabó, M Wiese and G Szakács. Design, synthesis and biological evaluation of thiosemicarbazones, hydrazinobenzothiazoles and arylhydrazones as anticancer agents with a potential to overcome multidrug resistance. European Journal of Medicinal Chemistry 2016; 117, 335-354.
A Kajal, S Bala, N Sharma, S Kamboj and V Saini. Therapeutic potential of hydrazones as anti-inflammatory agents. International Journal of Medicinal Chemistry 2014; 2014, 761030.
SG Küçükgüzel, A Mazi, F Sahin, S Öztürk and J Stables. Synthesis and biological activities of diflunisal hydrazide-hydrazones. European Journal of Medicinal Chemistry 2003; 38(11-12), 1005-1013.
JV Ragavendran, D Sriram, SK Patel, IV Reddy, N Bharathwajan, J Stables and P Yogeeswari. Design and synthesis of anticonvulsants from a combined phthalimide-GABA-anilide and hydrazone pharmacophore. European Journal of Medicinal Chemistry 2007; 42(2), 146-151.
MA Abdelrahman, I Salama, MS Gomaa, MM Elaasser, MM Abdel-Aziz and DH Soliman. Design, synthesis and 2D QSAR study of novel pyridine and quinolone hydrazone derivatives as potential antimicrobial and antitubercular agents. European Journal of Medicinal Chemistry 2017; 138, 698-714.
S Vogel, D Kaufmann, M Pojarová, C Müller, T Pfaller, S Kühne, PJ Bednarski and EV Angerer. Aroyl hydrazones of 2-phenylindole-3-carbaldehydes as novel antimitotic agents. Bioorganic & Medicinal Chemistry 2008; 16(12), 6436-6447.
C Vanucci-Bacqué, C Carayon, C Bernis, C Camare, A Nègre-Salvayre and F Bedos-Belval. Synthesis, antioxidant and cytoprotective evaluation of potential antiatherogenic phenolic hydrazones. A structure-activity relationship insight. Bioorganic & Medicinal Chemistry 2014; 22(15), 4269-4276.
Jasril, Y Nurulita, N Afriana, I Ikhtiarudin and N Frimayanti. Synthesis, docking, and molecular dynamic study of hydrazones compounds to search potential inhibitor for breast cancer MCF-7. Thai Journal of Pharmaceutical Sciences 2021; 45(6), 477-486.
V Balachandran, G Santhi, V Karpagam and A Lakshmi. DFT computation and spectroscopic analysis of N-(p-methoxybenzylidene) aniline, a potentially useful NLO material. Journal of Molecular Structure 2013; 1047, 249-261.
Naveen, RK Tittal, VD Ghule, N Kumar, L Kumar and K Lal. Design, synthesis, biological activity, molecular docking and computational studies on novel 1,4-disubstituted-1,2,3-Triazole-Thiosemicarbazone hybrid molecules. Journal of Molecular Structure 2020; 1209, 127951.
C James, AA Raj, R Reghunathan, VS Jayakumar and IH Joe. Structural conformation and vibrational spectroscopic studies of 2,6-bis(p-N,N-dimethyl benzylidene) cyclohexanone using density functional theory. Journal of Raman Spectroscopy 2006; 37(12), 1381-1392.
Nurlelasari, I Rahmayanti, S Salam, A Safari, D Harneti, R Maharani, AT Hidayat, M Tanjung, R Retnowati, Y Shiono and U Supratman. A new havanensin-type limonoid from Chisocheton macrophyllus. Applied Biological Chemistry 2021; 64, 35.
C Thepmalee, N Sawasdee, S Thongyim, N Poungvarin, PT Yenchitsomanus and A Panya. Enhancing T cell cytotoxicity against lung cancer with an αPD-L1 protein engager and gemcitabine combination therapy. Biomedicine & Pharmacotherapy 2025; 188, 118161.
J Branković, N Milivojevic, V Milovanovic, D Simijonovic, ZD Petrovic and Z Markovic. Evaluation of antioxidant and cytotoxic properties of phenolic N-acylhydrazones: Structure-activity relationship. Royal Society Open Science 2022; 9(6), 211853.
YP Pasaribu, A Fadlan, S Fatmawati and T Ersam. Biological activity evaluation and in silico studies of polyprenylated benzophenones from Garcinia celebica. Biomedicines 2021; 9(11), 1654.
A Ragab, YA Ammar, A Ezzat, AM Mahmoud, MBI Mohamed, AS El-Tabl and RS Farag. Synthesis, characterization, thermal properties, antimicrobial evaluation, ADMET study, and molecular docking simulation of new mono Cu (II) and Zn (II) complexes with 2-oxoindole derivatives. Computers in Biology and Medicine 2022; 145, 105473.
SA Rupa, MR Moni, MAM Patwary, MM Mahmud, MA Haque, J Uddin and SMT Abedin. Synthesis of novel tritopic hydrazone ligands: Spectroscopy, biological activity, DFT, and molecular docking studies. Molecules 2022; 27(5), 1656.
L Adjissi, N Chafai, K Benbouguerra, I Kirouani, A Hellal, H Layaida, M Elkolli, C Bensouici and S Chafaa. Synthesis, characterization, DFT, antioxidant, antibacterial, pharmacokinetics and inhibition of SARS-CoV-2 main protease of some heterocyclic hydrazones. Journal of Molecular Structure 2022; 1270, 134005.
YS Alam, P Pudjiastuti, S Malulana, NR Affifah, F Martak and A Fadlan. Synthesis and antidiabetic evaluation of N’-Benzylidenebenzohydrazide derivatives by in silico studies. Indonesian Journal of Chemistry 2023; 23(4), 1061-1070.
PC Agu, CA Afiukwa, OU Orji, EM Ezeh, IH Ofoke, CO Ogbu, EI Ugwuja and PM Aja . Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management. Scientific Reports 2023; 13, 13398.
I Al-Qadsy, AB Al-Odayni, WS Saeed, A Alrabie, A Al-Adhreai, LAS Al-Faqeeh, P Lama, AA Alghamdi and M Farooqui. Synthesis, characterization, single-crystal x-ray structure and biological activities of [(Z)-N -(4-Methoxybenzylidene)benzohydrazide-Nickel(II)] complex. Crystals 2021; 11(2), 110.
W Yu, G Huang,Y Zhang, H Liu, L Dong, X Yu, Y Li and J Chang. I2-mediated oxidative C−O bond formation for the synthesis of 1,3,4-Oxadiazoles from aldehydes and hydrazides. The Journal of Organic Chemistry 2013; 78(20), 10337-10343.
S Nagar, S Raizada and N Tripathee. A review on various green methods for synthesis of Schiff base ligands and their metal complexes. Results in Chemistry 2023; 6, 101153.
J Silver. Let us teach proper thin layer chromatography technique. Journal of Chemical Education 2020; 97(12), 4217-4219.
K Benbouguerra, N Chafai, S Chafaa, YI Touahria and H Tlidjane. New α-Hydrazinophosphonic acid: Synthesis, characterization, DFT study and in silico prediction of its potential inhibition of SARS-CoV-2 main protease. Journal of Molecular Structure 2021; 1239, 130480.
N Chafai, S Chafaa, K Benbouguerra, A Hellal and M Mehri. Synthesis, spectral analysis, anti-corrosive activity and theoretical study of an aromatic hydrazone derivative. Journal of Molecular Structure 2019; 1181, 83-92.
NA Mathews, A Jose and MRP Kurup. Synthesis and characterization of a new aroylhydrazone ligand and its cobalt(III) complexes: X-ray crystallography and in vitro evaluation of antibacterial and antifungal activities. Journal of Molecular Structure 2019; 1178, 544-553.
K Ahmed, M Bashir, R Bano, M Sarfraz, HU Khan, S Khan, A Sharif, A Waseem, MA Gilani, K Batool, R Idrees, A Rauf, RSZ Saleem and M Arshad. Potent heteroaromatic hydrazone based 1,2,4-triazine motifs: Synthesis, anti-oxidant activity, cholinesterase inhibition, quantum chemical and molecular docking studies. Journal of Molecular Structure 2023; 1284, 135383.
P Teli, S Soni, S Teli and S Agarwal. Unveiling the catalytic potency of a novel hydrazone-linked covalent organic framework for the highly efficient one-pot synthesis of 1,2,4-triazolidine-3-thiones. Nanoscale Advances 2024; 6(22), 5568-5578.
S Ibesh, Y Bitar and S Trefi. A New method for simultaneous qualitative and quantitative determination of amlodipine besylate and atorvastatin calcium in bulk and pharmaceutical formulations using transmission FT-IR spectroscopy. Heliyon 2023; 9(3), 14189.
EH El-Mossalamy, NF Al-Harby, SA Aal, NM Ali, M El-Desawy, MM Elewa and ME Batouti. Tenability on schiff base Hydrazone derivatives and Frontier molecular orbital. Heliyon 2024, 10(2), 24472.
R Munir, N Javid, M Zia-Ur-Rehman, M Zaheer, R Huma, A Roohi and MM Athar. Synthesis of novel N-acylhydrazones and their C-N/N-N bond conformational characterization by NMR spectroscopy. Molecules 2021; 26(16), 4908.
N Flores-Holguín, J Frau and D Glossman-Mitnik. In silico pharmacokinetics, ADMET study and conceptual DFT analysis of two plant cyclopeptides isolated from rosaceae as a computational peptidology approach. Frontiers in Chemistry 2021; 9, 708364.
NM Sabry, R Badry, FK Abdel-Gawad, H Elhaes and MA Ibrahim. Electronic structure, global reactivity descriptors and nonlinear optical properties of glycine interacted with ZnO, MgO and CaO for bacterial detection. Scientific Reports 2024; 14, 22801.
R Morad, M Akbari and M Maaza. Theoretical study of chemical reactivity descriptors of some repurposed drugs for COVID-19. MRS Advances 2023; 8, 656-660.
MA Mumit, TK Pal, MA Alam, MAAAA Islam, S Paul and MC Sheikh. DFT studies on vibrational and electronic spectra, HOMO-LUMO, MEP, HOMA, NBO and molecular docking analysis of benzyl-3-N-(2,4,5-trimethoxyphenylmethylene)hydrazinecarbodithioate. Journal of Molecular Structure 2020; 1220, 128715.
YT Male, IW Sutapa, IB Kapelle and M Lopulalan. QSAR modeling and design of a new model of anti- HIV drug 1-aryl-tetrahydroisoquinoline derived using the PM3 semiempirical method. RASAYAN Journal of Chemistry 2022; 15(1), 359-368.
HA Arjun, GNA Kumar, R Elancheran and S Kabilan. Crystal structure, DFT and Hirshfeld surface analysis of (E)-N′-[(1-chloro-3,4-dihydronaphthalen-2-yl)methylidene]benzohydrazide monohydrate. Acta Crystallographica Section E Crystallographic Communications 2020; 76(2), 132-136.
U Vanitha, R Elancheran, V Manikandan, S Kabilan and K Krishnasamy. Design, synthesis, characterization, molecular docking and computational studies of 3-phenyl-2-thioxoimidazolidin-4-one derivatives. Journal of Molecular Structure 2021; 1246, 131212.
N Chafai, S Chafaa, K Benbouguerra, D Daoud, A Hellal and M Mehri. Synthesis, characterization and the inhibition activity of a new α-aminophosphonic derivative on the corrosion of XC48 carbon steel in 0.5 M H2SO4: Experimental and theoretical studies. Journal of the Taiwan Institute of Chemical Engineers 2017; 70, 331-344.
H Kang and SG Lee. Scalable analysis of dipole moment fluctuations for characterizing intermolecular interactions and structural stability. Journal of Chemical Information and Modeling 2024; 64(11), 4518-4529.
TK Chaitra, KN Mohana, DM Gurudatt and HC Tandon. Inhibition activity of new thiazole hydrazones towards mild steel corrosion in acid media by thermodynamic, electrochemical and quantum chemical methods. Journal of the Taiwan Institute of Chemical Engineers 2016; 67, 521-531.
M Rafiq, M Khalid, MN Tahir, MU Ahmad, MU Khan, MM Naseer, AAC Braga, S Muhammad and Z Shafiq. Synthesis, XRD, spectral (IR, UV-Vis, NMR) characterization and quantum chemical exploration of benzoimidazole-based hydrazones: A synergistic experimental-computational analysis. Applied Organometallic Chemistry 2019; 33(11), 5182.
A Itam, MS Wati, V Agustin, N Sabri, RA Jumanah and M Efdi. Comparative study of phytochemical, antioxidant, and cytotoxic activities and phenolic content of Syzygium aqueum (Burm. f. Alston f.) extracts growing in West Sumatera Indonesia. The Scientific World Journal 2021; 2021, 5537597.
GU Anywar, E Kakudidi, H Oryem-Origa, A Schubert and C Jassoy. Cytotoxicity of medicinal plant species used by traditional healers in treating people suffering from HIV/AIDS in uganda. Frontiers in Toxicology 2022; 4, 832780.
Mİ Han and N İmamoğlu. Design, synthesis, and anticancer evaluation of novel tetracaine hydrazide-hydrazones. ACS Omega 2023; 8(10), 9198-9211.
L Fu , S Shi , J Yi , N Wang , Y He , Z Wu , J Peng , Y Deng , W Wang , C Wu , A Lyu , X Zeng , W Zhao , T Hou and D Cao. ADMETlab 3.0: An updated comprehensive online ADMET prediction platform enhanced with broader coverage, improved performance, API functionality and decision support. Nucleic Acids Research 2024; 52(1), 422-431.
J Zhang, Y Gan, H Li, J Yin, X He, L Lin, S Xu, Z Fang, B Kim, L Gao, L Ding, E Zhang, X Ma, J Li, L Li, Y Xu, D Horne, R Xu, H Yu, Y Gu and W Huang. Inhibition of the CDK2 and cyclin a complex leads to autophagic degradation of CDK2 in cancer cells. Nature Communications 2022; 13, 2835.
T Topal, Y Zorlu and N Karapınar. Synthesis, X-ray crystal structure, IR and Raman spectroscopic analysis, quantum chemical computational and molecular docking studies on hydrazone-pyridine compound: As an insight into the inhibitor capacity of main protease of SARS-CoV2. Journal of Molecular Structure 2021; 1239, 130514.
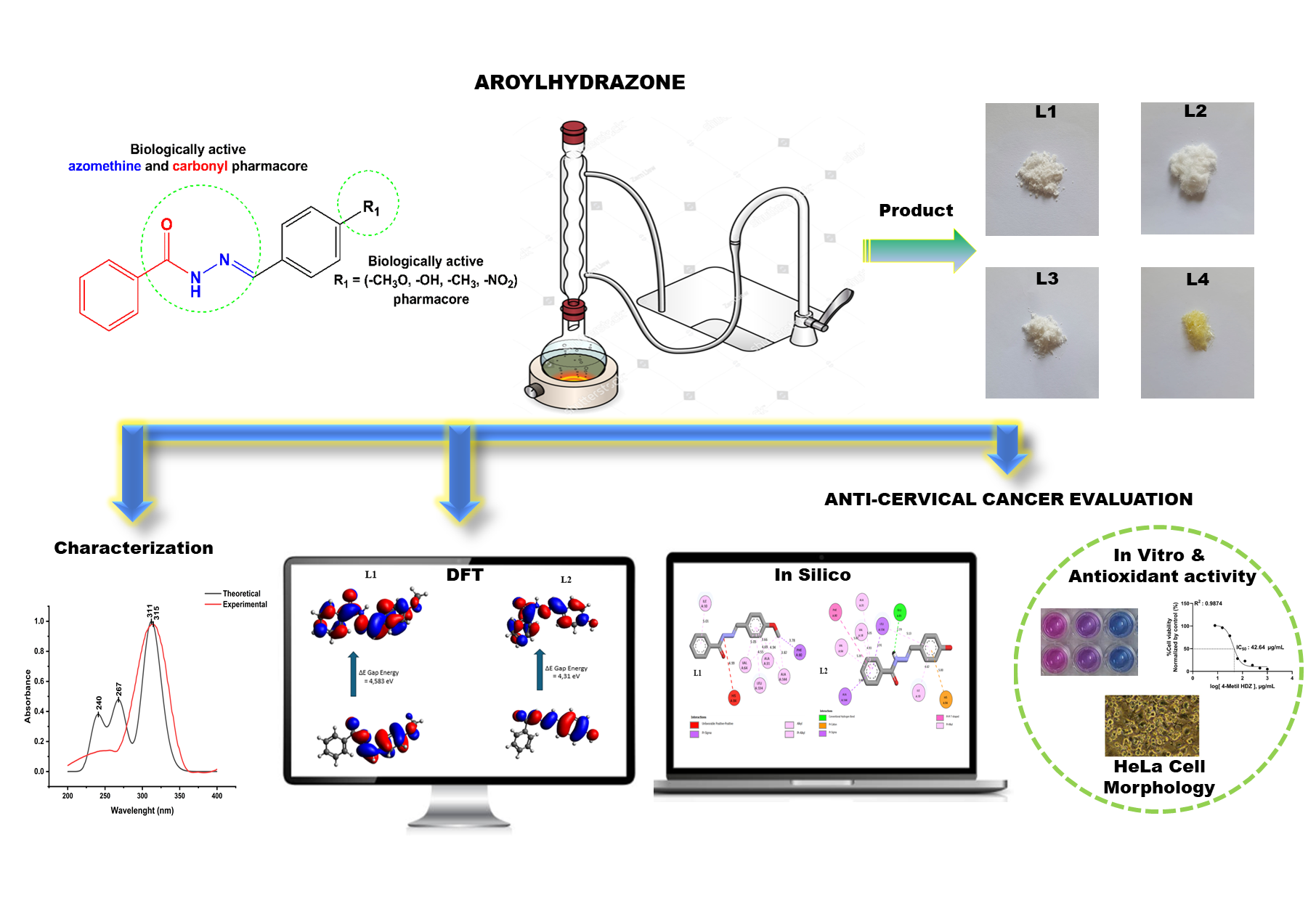
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






