Transcriptomic Profiling of 6-OHDA- and Reserpine-Mediated Neurotoxicity and Neurobehavioral Change in Caenorhabditis elegans Model
DOI:
https://doi.org/10.48048/tis.2026.11920Keywords:
Transcriptome, Metascape, 6-OHDA, Reserpine, Neurotoxicity, Caenorhabditis elegansAbstract
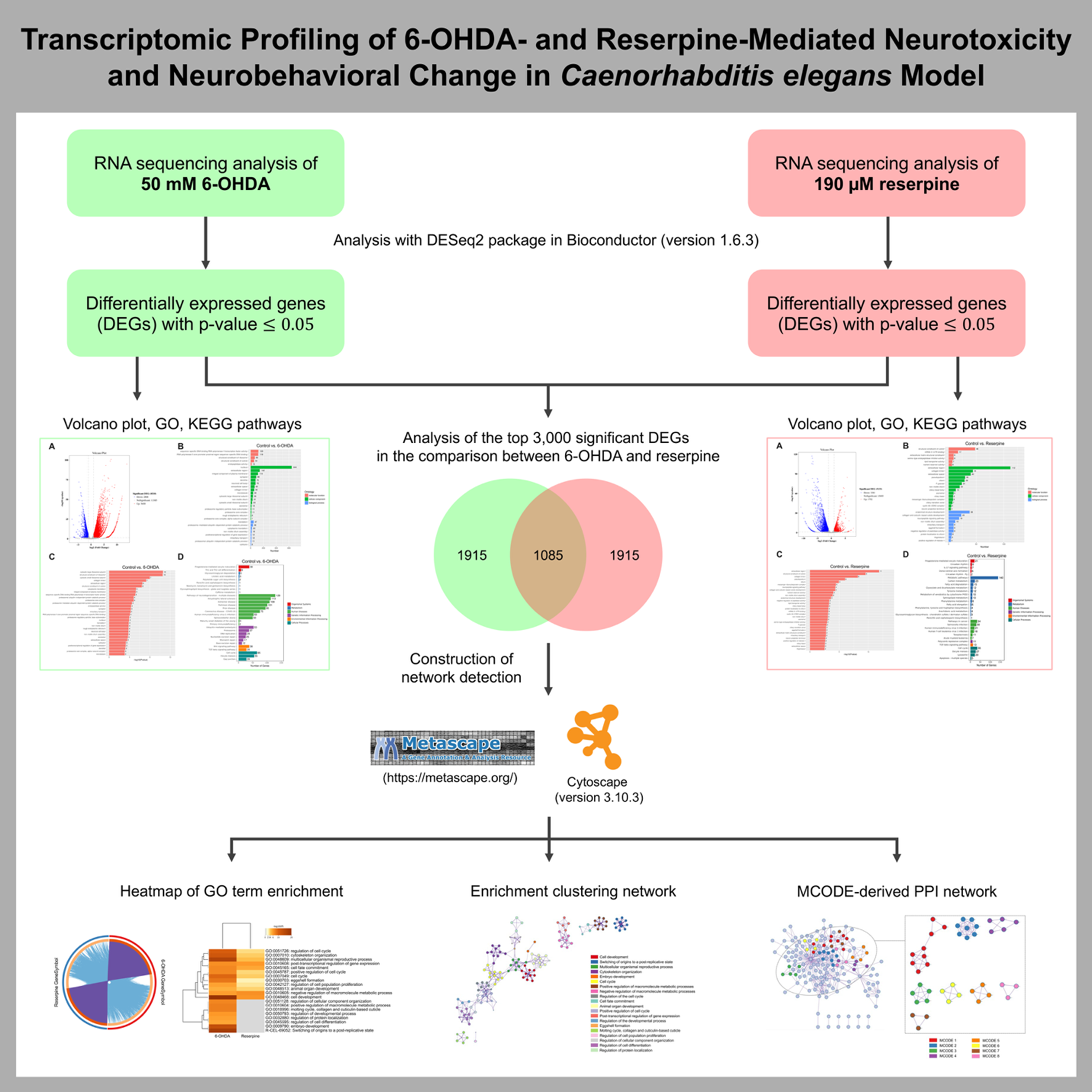
Neurotoxic agents such as 6-hydroxydopamine (6-OHDA) and reserpine are reported to cause neurodegenerative and neuroendocrine effects, respectively. Using the Caenorhabditis elegans (C. elegans) model, this study performs comparative transcriptomic profiling to elucidate molecular pathways that contribute to their neurotoxicity. Differentially expressed genes (DEGs) were identified following 6-OHDA and reserpine exposure, with subsequent analyses for Gene Ontology (GO) and KEGG pathway enrichment. Notably, 6-OHDA mainly disrupted ribosomal/proteasomal activity and neurodegeneration-related pathways, while reserpine primarily affected the extracellular compartment, neuropeptide-based signaling, and metabolic processes. Meta-analysis of the top 3,000 DEGs, combined with comparative enrichment network analysis of the 2 neurotoxicants using Metascape, identified critical regulatory processes, including cell development, reproduction, cytoskeletal organization, cell cycle control, metabolic pathways, and post-transcriptional regulation. In addition, protein-protein interaction (PPI) network analysis revealed eight distinct Molecular Complex Detection (MCODE) modules, which correspond to biological clusters such as regulation of translation, fatty acid metabolism, glutamate/glutamine metabolism, ceramide metabolism, sphingolipid metabolism, porphyrin metabolism, and post-transcriptional regulation. Moreover, reserpine exposure altered neurobehavioral function in the ethanol avoidance assay without inducing dopaminergic (DAergic) neurodegeneration in C. elegans. Overall, these findings suggest a transcriptomic framework that reports both common and unique molecular signatures of neurotoxicant-mediated neurodegeneration and behavioral alterations. These results provide valuable insights for future studies on regulatory pathways involved in PD pathogenesis.
HIGHLIGHTS
- 6-OHDA predominantly modulated ribosomal/proteasomal activity and neurodegeneration-related processes in C. elegans.
- Reserpine primarily influenced the extracellular region, neuropeptide-mediated signaling, and metabolic processes in C. elegans.
- Overlapping enriched DEGs identified by Metascape between the 6-OHDA- and reserpine-treated groups were mainly associated with cell development, reproduction, cytoskeletal organization, cell cycle regulation, metabolic pathways, and post-transcriptional regulation.
- MCODE cluster analysis revealed metabolic modules encompassing regulation of translation, fatty acid metabolism, glutamate/glutamine metabolism, ceramide metabolism, sphingolipid metabolism, porphyrin metabolism, and post-transcriptional regulation.
- Reserpine toxicity altered dopamine-dependent behavioral function in the ethanol avoidance assay without inducing DAergic neurodegeneration in C. elegans.
GRAPHICAL ABSTRACT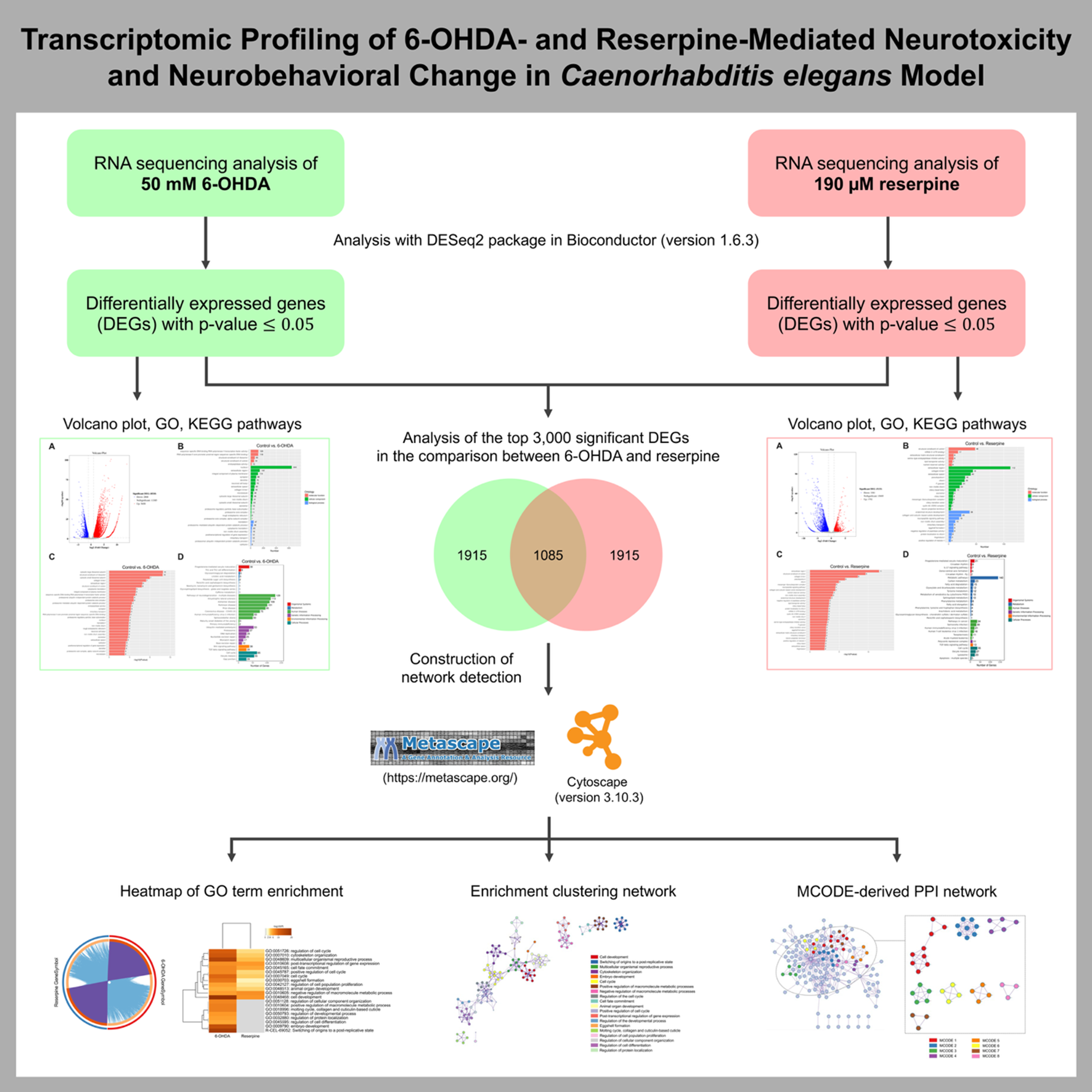
Downloads
References
DM Wilson, MR Cookson, L Van Den Bosch, H Zetterberg, DM Holtzman and I Dewachter. Hallmarks of neurodegenerative diseases. Cell 2023; 186(4), 693-714.
E Guatteo, N Berretta, V Monda, A Ledonne and NB Mercuri. Pathophysiological features of nigral dopaminergic neurons in animal models of Parkinson’s disease. International Journal of Molecular Sciences 2022; 23(9), 4508.
National Research Council (US) Committee on Neurotoxicology and Models for Assessing Risk. Environmental neurotoxicology. National Academies Press, Washington DC, 1992.
AY Altahrawi, AW James and ZA Shah. The role of oxidative stress and inflammation in the pathogenesis and treatment of vascular dementia. Cells 2025; 14(8), 609.
A Schober. Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell and Tissue Research 2004; 318(1), 215-224.
Y Li, Q Yin, B Wang, T Shen, W Luo and T Liu. Preclinical reserpine models recapitulating motor and non-motor features of Parkinson’s disease: Roles of epigenetic upregulation of alpha-synuclein and autophagy impairment. Frontiers in Pharmacology 2022; 13, 944376.
M Govindarajulu, T Shankar, S Patel, M Fabbrini, A Manohar, S Ramesh and P Boralingaiah. Reserpine-induced depression and other neurotoxicity: A monoaminergic hypothesis. In: Medicinal herbs and fungi: Neurotoxicity vs. Neuroprotection. Springer, Cham, Switzerland, 2021, p. 293-313.
AC Lima, VS Bioni, MS Becegato, Y Meier, DMG Cunha, NA Aguiar, N Gonçalves, FF Peres, AW Zuardi, JEC Hallak, JEC Hallak, A Crippa, JA Crippa, SS Smaili, SS Smaili, VC Abilio,VC Abilio, RH Silva and RH Silva. Preventive beneficial effects of cannabidiol in a reserpine-induced progressive model of parkinsonism. Frontiers in Pharmacology 2025; 16, 1539783.
J Lama, Y Buhidma, EJR Fletcher and S Duty. Animal models of Parkinson’s disease: A guide to selecting the optimal model for your research. Neuronal Signaling 2021; 5(4), NS20210026.
P Reckziegel, P Chen, S Caito, P Gubert, FAA Soares, R Fachinetto and M Aschner. Extracellular dopamine and alterations on dopamine transporter are related to reserpine toxicity in Caenorhabditis elegans. Archives of Toxicology 2016; 90(3), 633-645.
A Dovonou, C Bolduc, V Soto Linan, C Gora, MR Peralta and M Lévesque. Animal models of Parkinson’s disease: Bridging the gap between disease hallmarks and research questions. Translational Neurodegeneration 2023; 12(1), 36.
S Das, KS Saili and RL Tanguay. Nonmammalian models in toxicology screening. In: Encyclopedia of toxicology. 3rd ed. Elsevier, Amsterdam, Netherlands, 2014, p. 609-613.
J Li and W Le. Modeling neurodegenerative diseases in Caenorhabditis elegans. Experimental Neurology 2013; 250, 94-103.
X Chen, JW Barclay, RD Burgoyne and A Morgan. Using C. elegans to discover therapeutic compounds for ageing-associated neurodegenerative diseases. Chemistry Central Journal 2015; 9(1), 65.
M Maulik, S Mitra, A Bult-Ito, BE Taylor and EM Vayndorf. Behavioral phenotyping and pathological indicators of Parkinson’s disease in C. elegans models. Frontiers in Genetics 2017; 8, 77.
CH Lai, CY Chou, LY Ch’ang, CS Liu and W Lin. Identification of novel human genes evolutionarily conserved in Caenorhabditis elegans by comparative proteomics. Genome Research 2000; 10(5), 703-713.
X Chen, JW Barclay, RD Burgoyne and A Morgan. Using C. elegans to discover therapeutic compounds for ageing-associated neurodegenerative diseases. Chemistry Central Journal 2015; 9, 65.
S Promtang, T Sanguanphun, P Chalorak, D Rodma, R Sunan, LS Pe and N Niamnont. Neurorestorative properties of 2-butoxytetrahydrofuran from Holothuria scabra via activation of stress resistance and detoxification in a 6-OHDA-induced C. elegans model of Parkinson’s disease. Biomedicine & Pharmacotherapy 2025; 188, 118158.
E da Cruz Guedes, AG Erustes, AHFF Leão, CA Carneiro, VC Abílio, AW Zuardi, JEC Hallak, JA Crippa, C Bincoletto, SS Smaili, P Reckziegel and GJS Pereira. Cannabidiol recovers dopaminergic neuronal damage induced by reserpine or α-synuclein in Caenorhabditis elegans. Neurochemical Research 2023; 48(8), 2390-2405.
S Promtang, T Sanguanphun, P Chalorak, LS Pe, N Niamnont, P Sobhon and K Meemon. 2-butoxytetrahydrofuran, isolated from Holothuria scabra, attenuates aggregative and oxidative properties of α-synuclein and alleviates its toxicity in a transgenic Caenorhabditis elegans model of Parkinson’s disease. ACS Chemical Neuroscience 2024; 15(11), 2182.
Y Zhou, B Zhou, L Pache, M Chang, AH Khodabakhshi, O Tanaseichuk, C Benner and SK Chanda. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nature Communications 2019; 10(1), 1523.
J Kohen. A coefficient of agreement for nominal scale. Educational and Psychological Measurement 1960; 20, 37-46.
A Chatr-Aryamontri, R Oughtred, L Boucher, J Rust, C Chang, NK Kolas, L O’Donnell, S Oster, C Theesfeld, A Sellam, C Stark, BJ Breitkreutz, K Dolinski and M Tyers. The BioGRID interaction database: 2017 update. Nucleic Acids Research 2017; 45(D1), D369-D379.
GD Bader and CW Hogue. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics 2003; 4(1), 2.
P Shannon, A Markiel, O Ozier, NS Baliga, JT Wang, D Ramage, N Amin, B Schwikowski and T Ideker. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Research 2003; 13(11), 2498-2504.
JF Cooper, DJ Dues, KK Spielbauer, E Machiela, MM Senchuk and JM Van Raamsdonk. Delaying aging is neuroprotective in Parkinson’s disease: A genetic analysis in C. elegans models. NPJ Parkinson’s Disease 2015; 1(1), 15022.
I Alecu and SAL Bennett. Dysregulated lipid metabolism and its role in α-synucleinopathy in Parkinson’s disease. Frontiers in Neuroscience 2019; 13, 328.
S Yamasaki and P Anderson. Reprogramming mRNA translation during stress. Current Opinion in Cell Biology 2008; 20(2), 222-226.
JV Andersen, KH Markussen, E Jakobsen, A Schousboe, HS Waagepetersen, PA Rosenberg and BI Aldana. Glutamate metabolism and recycling at the excitatory synapse in health and neurodegeneration. Neuropharmacology 2021; 196, 108719.
A Vaglio-Garro, AV Kozlov, YD Smirnova and A Weidinger. Pathological interplay between inflammation and mitochondria aggravates glutamate toxicity. International Journal of Molecular Sciences 2024; 25(4), 2276.
K Czubowicz, H Jęśko, P Wencel, WJ Lukiw and RP Strosznajder. The role of ceramide and sphingosine-1-phosphate in Alzheimer’s disease and other neurodegenerative disorders. Molecular Neurobiology 2019; 56(8), 5436-5455.
M Hernández-Elvira and P Sunnerhagen. Post-transcriptional regulation during stress. FEMS Yeast Research 2022; 22(1), foac025.
D Maitra, JB Cunha, JS Elenbaas, HL Bonkovsky, JA Shavit and MB Omary. Porphyrin-induced protein oxidation and aggregation as a mechanism of porphyria-associated cell injury. Cellular and Molecular Gastroenterology and Hepatology 2019; 8(4), 535-548.
JC Vickers, AE King, A Woodhouse, MT Kirkcaldie, JA Staal, GH McCormack, CA Blizzard, REJ Musgrove, S Mitew, Y Liu, JA Chuckowree, O Bibari a TC Dickson Axonopathy and cytoskeletal disruption in degenerative diseases of the central nervous system. Brain Research Bulletin 2009; 80(4-5), 217-223.
JF Cooper and JM Van Raamsdonk. Modeling Parkinson’s disease in C. elegans. Journal Of Parkinson’s Disease 2018; 8(1), 17-32.
AI Bernstein, SP Garrison, GP Zambetti and KL O’Malley. 6-OHDA generated ROS induces DNA damage and p53-and PUMA-dependent cell death. Molecular Neurodegeneration 2011; 6(1), 2.
M Li. The role of P53 up-regulated modulator of apoptosis (PUMA) in ovarian development, cardiovascular and neurodegenerative diseases. Apoptosis 2021; 26(5), 235-247.
R Kandel, J Jung and S Neal. Proteotoxic stress and the ubiquitin proteasome system. Seminars in Cell & Developmental Biology 2024; 156, 107-120.
KB Magalingam, SD Somanath, P Ramdas, N Haleagrahara and AK Radhakrishnan. 6-hydroxydopamine induces neurodegeneration in terminally differentiated SH-SY5Y neuroblastoma cells via enrichment of the nucleosomal degradation pathway: A global proteomics approach. Journal of Molecular Neuroscience 2022; 72(5), 1026-1046.
N Pathak, SK Vimal, I Tandon, L Agrawal, C Hongyi and S Bhattacharyya. Neurodegenerative disorders of alzheimer, parkinsonism, amyotrophic lateral sclerosis and multiple sclerosis: An early diagnostic approach for precision treatment. Metabolic Brain Disease 2022; 37(1), 67-104.
Y Luo, M Zheng, Z Su, C Cai and X Li. Transcriptome profile of reserpine-induced locomotor behavioral changes in zebrafish (Danio rerio). Progress in Neuro-Psychopharmacology and Biological Psychiatry 2024; 129, 110874.
S Duty and P Jenner. Animal models of Parkinson’s disease: A source of novel treatments and clues to the cause of the disease. British Journal of Pharmacology 2011; 164(4), 1357-1391.
F Sun, L Xia, B Wang, Y Liu, X Cui, H Kang, R Stoika, K Liu and M Jin. Reserpine causes neuroendocrine toxicity, inducing impairments in cognition via disturbing hypothalamic-pituitary-thyroid axis in zebrafish. NeuroSci 2025; 6(2), 28.
AHFF Leão, AJ Sarmento‐Silva, JR Santos, AM Ribeiro and RH Silva. Molecular, neurochemical, and behavioral hallmarks of reserpine as a model for Parkinson’s disease: New perspectives to a long‐standing model. Brain Pathology 2015; 25(4), 377-390.
Y Cong, H Yang, P Zhang, Y Xie, X Cao and L Zhang. Transcriptome analysis of the nematode Caenorhabditis elegans in acidic stress environments. Frontiers in Physiology 2020; 11, 1107.
LK Ong, L Sominsky, PW Dickson, DM Hodgson and PR Dunkley. The sustained phase of tyrosine hydroxylase activation in vivo. Neurochemical Research 2012; 37(9), 1938-1943.
SC Daubner, T Le and S Wang. Tyrosine hydroxylase and regulation of dopamine synthesis. Archives of Biochemistry and Biophysics 2011; 508(1), 1-12.
S Zou, T Lang, B Zhang, K Huang, L Gong, H Luo, W Xu and X He. Fatty acid oxidation alleviates the energy deficiency caused by the loss of MPC1 in MPC1+/− mice. Biochemical and Biophysical Research Communications 2018; 495(1), 1008-1013.
BH Choi, S Hyun and SH Koo. The role of BCAA metabolism in metabolic health and disease. Experimental & Molecular Medicine 2024; 56(7), 1552-1559.
AA Anand, M Khan, MV and D Kar. The molecular basis of Wnt/β‐catenin signaling pathways in neurodegenerative diseases. International Journal of Cell Biology 2023; 2023(1), 9296092.
PD Lu, HP Harding and D Ron. Translation reinitiation at alternative open reading frames regulates gene expression in an integrated stress response. The Journal of Cell Biology 2004; 167(1), 27-33.
MA Bravo-Jimenez, S Sharma and S Karimi-Abdolrezaee. The integrated stress response in neurodegenerative diseases. Molecular Neurodegeneration 2025; 20(1), 20.
JM Frade and MC Ovejero-Benito. Neuronal cell cycle: The neuron itself and its circumstances. Cell Cycle 2015; 14(5), 712-720.
LW Hu, JH Yen, YT Shen, KY Wu and MJ Wu. Luteolin modulates 6-hydroxydopamine-induced transcriptional changes of stress response pathways in PC12 cells. PloS One 2014; 9(5), e97880.
R Park, KII Lee, H Kim, M Jang, TKQ Ha, WK Oh and J Park. Reserpine treatment activates AMP activated protein kinase (AMPK). Natural Product Sciences 2017; 23(3), 157-161.
C Volbracht, J Van Beek, C Zhu, K Blomgren and M Leist. Neuroprotective properties of memantine in different in vitro and in vivo models of excitotoxicity. European Journal of Neuroscience 2006; 23(10), 2611-2622.
J Qiu, G Peng, Y Tang, S Li, Z Liu, J Zheng, Y Wang, H Liu, H Liu, L Wei, Y Su, Y Lin, W Dai, Z Zhang, X Chen, L Ding, W Guo, X Zhu, P Xu and M Mo. Lipid profiles in the cerebrospinal fluid of rats with 6-hydroxydopamine-induced lesions as a model of Parkinson’s disease. Frontiers in Aging Neuroscience 2023; 14, 1077738.
F Zakaria, MT Akhtar, WN Wan Ibrahim, N Abu Bakar, A Muhamad, S Shohaimi, M Maulidiani, H Ahmad, IS Ismail and K Shaari. Perturbations in amino acid metabolism in reserpine-treated zebrafish brain detected by 1H nuclear magnetic resonance-based metabolomics. Zebrafish 2021; 18(1), 42-54.
C Weinhouse, L Truong, JN Meyer and P Allard. Caenorhabditis elegans as an emerging model system in environmental epigenetics. Environmental and Molecular Mutagenesis 2018; 59(7), 560-575.
G Khanjari, I Mohammadzadeh, Y Khakpour, M Nikoohemmat, M Norouzian, S Parvardeh, A Beirami, M Hasanzadeh, NB Amini, Z Niakan, GH Meftahi, A Seraj, AH Tajik, H Hemmatparast, R Bahar, MH Hajali, H Karbalaei-Musa, AH Bayat, MH Moghaddam, M Sani and A Aliaghae. Neuroprotective effects of the elderberry diet on the methamphetamine-induced toxicity in rats: A behavioral, electrophysiological, and histopathological study. 3 Biotech 2025; 15(5), 122.
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






