Potential Effects of Pseuderanthemum palatiferum Extract on Inhibiting Adipogenesis and Promoting Lipolysis in 3T3-L1 Cell Line
DOI:
https://doi.org/10.48048/tis.2026.11840Keywords:
Pseuderanthemum palatiferum, Adipogenesis, Lipolysis, Adipocyte, 3T3-L1, Reactive oxygen species, Liquid chromatography-mass spectrometry, Gas chromatography-mass spectrometryAbstract
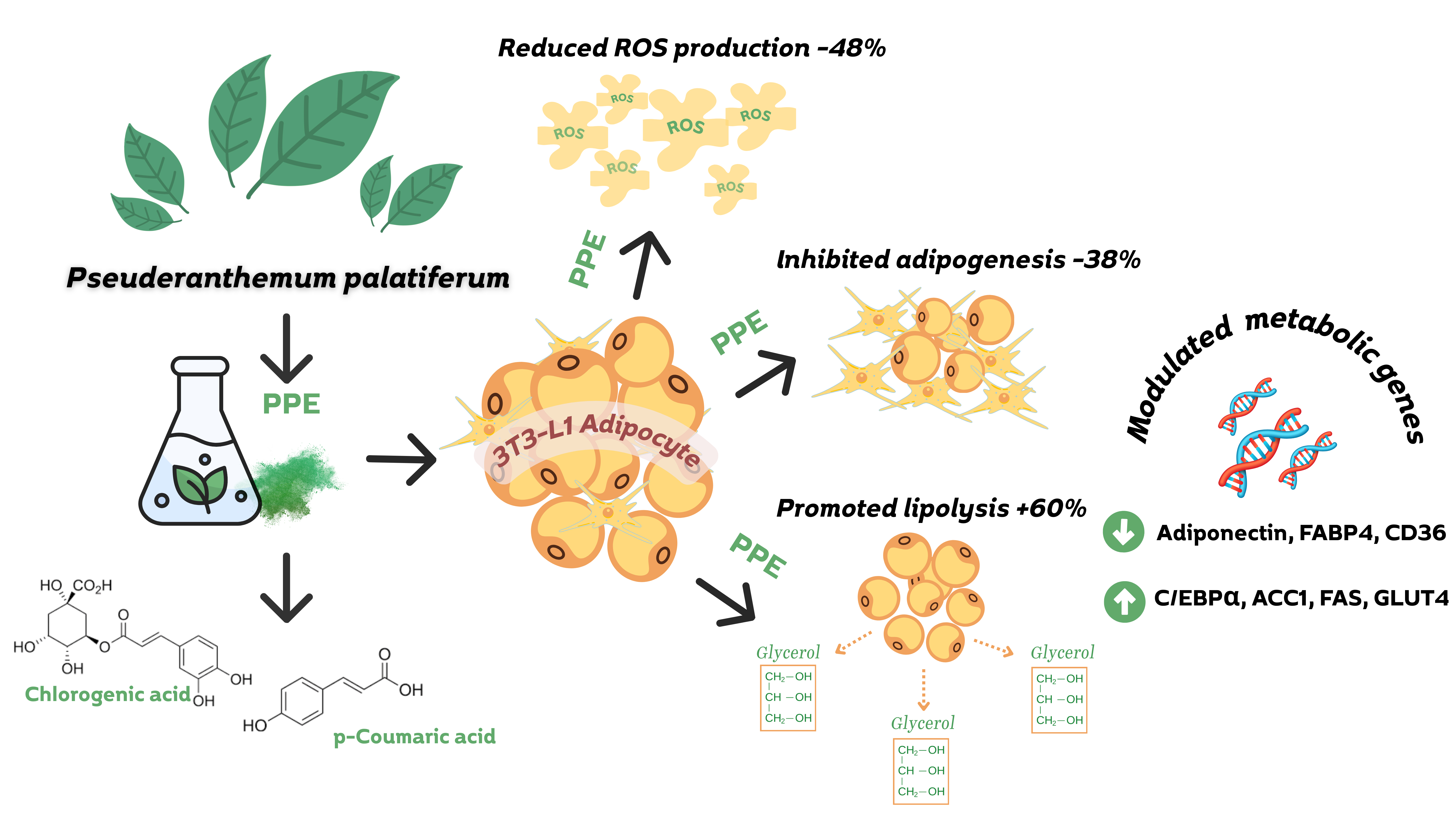
Obesity is a global health concern driven by excessive lipid accumulation and adipocyte dysfunction, requires therapeutic strategies that both inhibit fat formation and promote lipid degradation. This study investigated the dual anti-adipogenic and pro-lipolytic effects of Pseuderanthemum palatiferum leaf extract (PPE) in 3T3-L1 adipocytes. The phytochemical analysis of PPE performed by using gas chromatography-mass spectrometry (GC-MS) and Liquid chromatography-mass spectrometry (LC-MS). The findings revealed that GC-MS analysis of PPE identified 35 volatile compounds, with benzofuran (29.94%) as the major constituent. LC-MS detected 27 non-volatiles phytochemicals, including chlorogenic acid and p-coumaric acid. PPE exhibited no significant cytotoxic effects on 3T3-L1 preadipocytes at doses ranging from 50 - 150 μg/mL. At 150 μg/mL, PPE reduced lipid accumulation by 37.84% and increased glycerol release by 60.46%, indicating both an anti-adipogenic and a lipolytic properties. Gene expression analysis revealed that pre-treatment with 150 μg/mL of PPE promoted the upregulation of early adipogenic markers (C/EBPα, ACC1, FAS, and GLUT4) while downregulating markers associated with mature adipocytes (Adiponectin, AP2, and CD36), indicating interference with adipocyte differentiation and lipid synthesis. Additionally, PPE lowered intracellular reactive oxygen species, suggesting an antioxidative contribution to its anti-obesity action. Collectively, this study provides the first evidence that P. palatiferum exerts complementary regulation of adipogenesis and lipolysis in 3T3-L1 cells, offering new insight into its multi-target mechanisms and potential as a natural therapeutic candidate for obesity management.
HIGHLIGHTS
- Abnormal lipid metabolism contributes to obesity, accompanied by oxidative stress and inflammation.
- PPE modulates key pathways regulating fat storage and breakdown, leading to reduced lipid accumulation.
- PPE may influence adipose cells to reduce the release of inflammatory factors and subsequently to diminish oxidative stress.
- PPE may serve as a natural candidate for dietary or therapeutic strategies targeting obesity.
GRAPHICAL ABSTRACT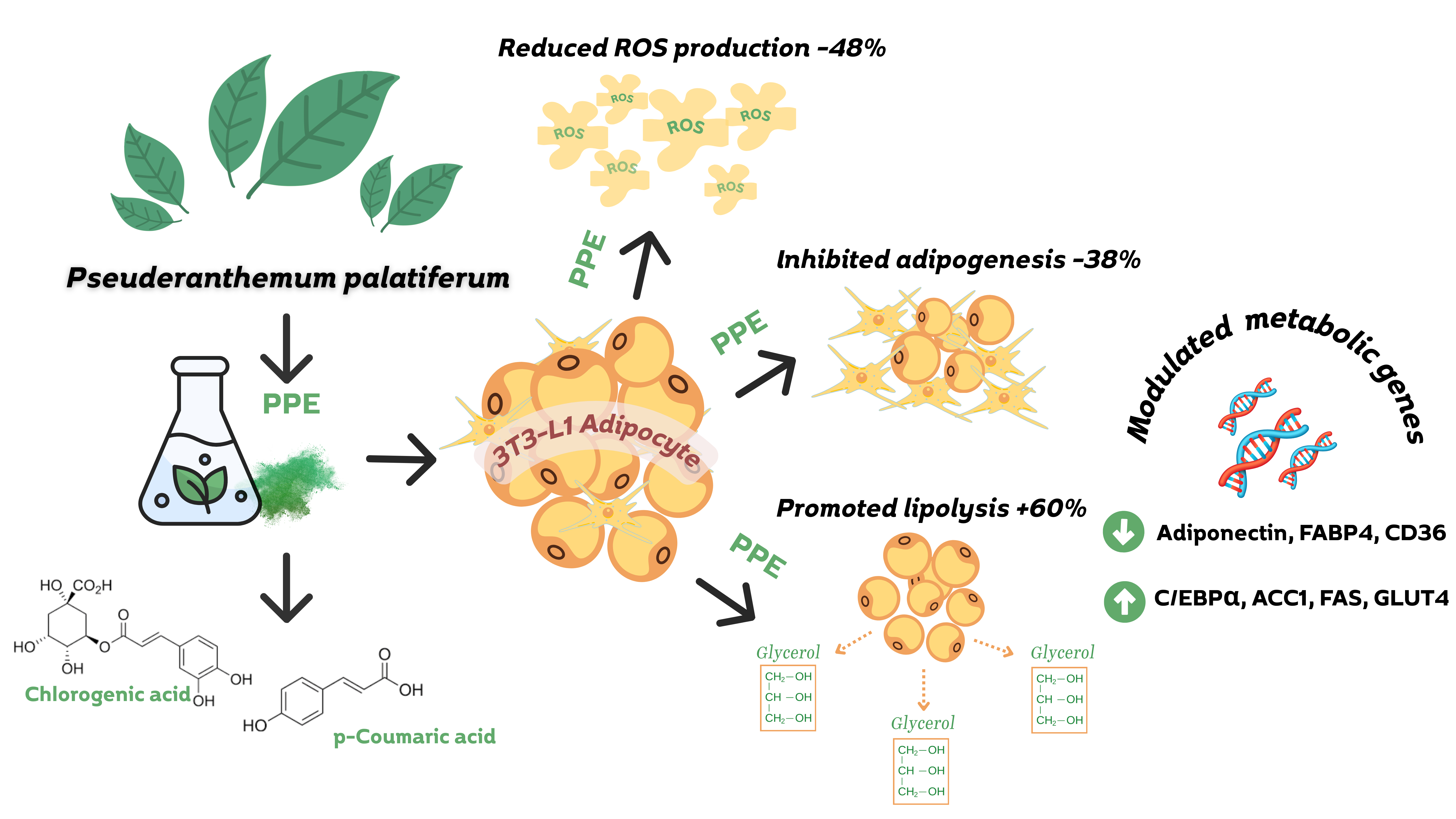
Downloads
References
Y Zhang, J Liu, J Yao, G Ji, L Qian, J Wang, G Zhang, J Tian, Y Nie, YE Zhang, MS Gold and Y Liu. Obesity: Pathophysiology and intervention. Nutrients 2014; 6(11), 5153-5183.
X Jin, T Qiu, L Li, R Yu, X Chen, C Li, CG Proud and T Jiang. Pathophysiology of obesity and its associated diseases. Acta Pharmaceutica Sinica B 2023; 13(6), 2403-2424.
K Lee, L Kruper, CM Dieli-Conwright and JE Mortimer. The impact of obesity on breast cancer diagnosis and treatment. Current Oncology Reports 2019; 21, 41.
KA Orringer, RV Harrison, SS Nichani, MA Riley, AE Rothberg, LE Trudeau and Y White. Obesity prevention and management. Michigan Medicine University of Michigan, Michigan, 2020.
ED Rosen and BM Spiegelman. What we talk about when we talk about fat. Cell 2014; 165(1-2), 20-44.
J Jakab, B Miskic, S Miksic, B Juranic, V Cosic, D Schwarz and A Vcev. Adipogenesis as a potential anti-obesity target: A review of pharmacological treatment and natural products. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 2021; 14, 67-83.
XD Yang, XC Ge, SY Jiang and YY Yang. Potential lipolytic regulators derived from natural products as effective approaches to treat obesity. Frontiers in Endocrinology 2022; 13, 1000739.
A Yang and EP Mottillo. Adipocyte lipolysis: From molecular mechanisms of regulation to disease and therapeutics. Biochemical Journal 2020; 477(5), 985-1008.
M Rajendran, S Palani, TG Singh, B Oliver, K Dua, V Subramaniyan and K Narayanan. The dual role of conjugated linoleic acid in obesity and metabolic disorders. Food Science & Nutrition 2025; 13(7), 70582.
K Chayarop, R Temsiririrkkul, P Peungvicha, Y Wongkrajang, W Chuakul, S Amnuoypol and N Ruangwises. Antidiabetic effects and in vitro antioxidant activity of Pseuderanthemum palatiferum (Nees) Radlk. ex Lindau leaf aqueous extract. Mahidol University Journal of Pharmaceutical Science 2011; 38(3-4), 13-22.
P Komonrit and R Banjerdpongchai. Effect of Pseuderanthemum palatiferum (Nees) Radlk fresh leaf ethanolic extract on human breast cancer MDA-MB-231 regulated cell death. Tumor Biology 2018; 40(9), 1010428318800182.
P Padee, S Nualkaew, C Talubmook and S Sakuljaitrong. Hypoglycemic effect of a leaf extract of Pseuderanthemum palatiferum (Nees) Radlk. in normal and streptozotocin-induced diabetic rats. Journal of Ethnopharmacology 2010; 132(2), 491-496.
T Khumpook, S Chomdej, S Saenphet, D Amornlerdpison and K Saenphet. Anti-inflammatory activity of ethanol extract from the leaves of Pseuderanthemum palatiferum (Nees) radlk. Chiang Mai Journal of Science 2013; 40(3), 321-331.
K Chayarop, P Peungvicha, Y Wongkrajang, W Chuakul, S Amnuoypol and R Temsiririrkkul. Pharmacognostic and phytochemical investigations of Pseuderanthemum palatiferum (Nees) radlk. ex Lindau Leaves. Pharmacognosy Journal 2011; 3(23), 18-23.
P Sittisart, B Chitsomboon and NE Kaminski. Pseuderanthemum palatiferum leaf extract inhibits the proinflammatory cytokines, TNF-α and IL-6 expression in LPS-activated macrophages. Food and Chemical Toxicology 2016; 97, 11-22.
P Sittisart and B Chitsomboon. Intracellular ROS scavenging activity and downregulation of inflammatory mediators in RAW264.7 macrophage by fresh leaf extracts of Pseuderanthemum palatiferum. Evidence-Based Complementary and Alternative Medicine 2014; 2014, 309095.
KJ Livak and TD Schmittgen. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001; 25(4), 402-408.
B Dunkhunthod, C Talabnin, M Murphy, K Thumanu, P Sittisart and G Eumkeb. Gymnema inodorum (Lour.) Decne. extract alleviates oxidative stress and inflammatory mediators produced by RAW264.7 macrophages. Oxidative Medicine and Cellular Longevity 2021; 2021, 8658314.
JJ Fuster, N Ouchi, N Gokce and K Walsh. Obesity-Induced changes in adipose tissue microenvironment and their impact on cardiovascular disease. Circulation Research 2016; 118(11), 1786-1807.
UFSM Sayed, S Moshawih, HP Goh, N Kifli, G Gupta, SK Singh, DK Chellappan, K Dua, A Hermansyah, HL Ser, LC Ming and BH Goh. Natural products as novel anti-obesity agents: Insights into mechanisms of action and potential for therapeutic management. Frontiers in Pharmacology 2023; 14, 1182937.
P Padee, S Nualkaew, C Talubmook and S Sakuljaitrong. Hypoglycemic effect of a leaf extract of Pseuderanthemum palatiferum (Nees) radlk. in normal and streptozotocin-induced diabetic rats. Journal of Ethnopharmacology 2010; 132(2), 491-496.
P Padee, S Nualkaew, C Talubmook and S Sakuljaitrong. Acute toxicity and sub-acute toxicity of Pseuderanthemun palatiferum (Nees) radlk. Leaf Extract. Isan Journal of Pharmaceutical Sciences 2009; 5(1), 74-81.
YH Kim, DH Kim, H Lim, DY Baek, HK Shin and JK Kim. The anti-inflammatory effects of methylsulfonylmethane on lipopolysaccharide-induced inflammatory responses in murine macrophages. Biological and Pharmaceutical Bulletin 2009; 32(4), 651-656.
I Sousa-Lima, SY Park, M Chung, HJ Jung, MC Kang, JM Gaspar, JA Seo, MP Macedo, KS Park, C Mantzoros, SH Lee and YB Kim. Methylsulfonylmethane (MSM), an organosulfur compound, is effective against obesity-induced metabolic disorders in mice. Metabolism 2016; 65(10), 1508-1521.
JB Jeong, SC Hong, HJ Jeong and JS Koo. Anti-inflammatory effect of 2-methoxy-4-vinylphenol via the suppression of NF-κB and MAPK activation, and acetylation of histone H3. Archives of Pharmacal Research 2011; 34(12), 2109-2106.
V Danh, N Trang and XT Vo. Influence of green solvent extraction on phytochemicals, potential antidiabetic and in vitro anti-inflammatory activities of Pseuderanthemum palatiferum (Nees.) Radlk. Leaves. The Tropical Journal of Natural Product Research 2023; 7(1), 2215-2219.
LV Vasileva, MS Savova, KM Amirova, Z Balcheva-Sivenova, C Ferrante, G Orlando, M Wabitsch and MI Georgiev. Caffeic and chlorogenic acids synergistically activate browning program in human adipocytes: Implications of AMPK- and PPAR-Mediated pathways. International Journal of Molecular Sciences 2020; 21(24), 9740.
SW Kang, SI Kang, HS Shin, SA Yoon, JH Kim, HC Ko and SJ Kim. Sasa quelpaertensis Nakai extract and its constituent p-coumaric acid inhibit adipogenesis in 3T3-L1 cells through activation of the AMPK pathway. Food and Chemical Toxicology 2013; 59, 380-385.
B Ahmad, CJ Serpell, IL Fong and EH Wong. Molecular mechanisms of adipogenesis: The anti-adipogenic role of AMP-Activated protein kinase. Frontiers in Molecular Biosciences 2020; 7, 76.
P Jimenez-Quevedo, A Serrador, AP de Prado, L Nombela-Franco, C Biagioni and M Pan. Selection of the best of 2016 in interventional cardiology: Expansion of TAVI indications to intermediate-risk patients. Revista Española de Cardiología 2017; 70(3), 218-219.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






