Acute Toxicity Evaluation of A Combined Extract (Mitragyna Speciosa and Lagerstroemia Speciosa) Leaves in Rats: Hematological, Biochemical and Histopathological Examinations
DOI:
https://doi.org/10.48048/tis.2026.10867Keywords:
Mitragyna speciosa, Lagerstroemia speciosa, Acute toxicity, Single dose, Rats, Hematological, Biochemical, Histopathological examinationsAbstract
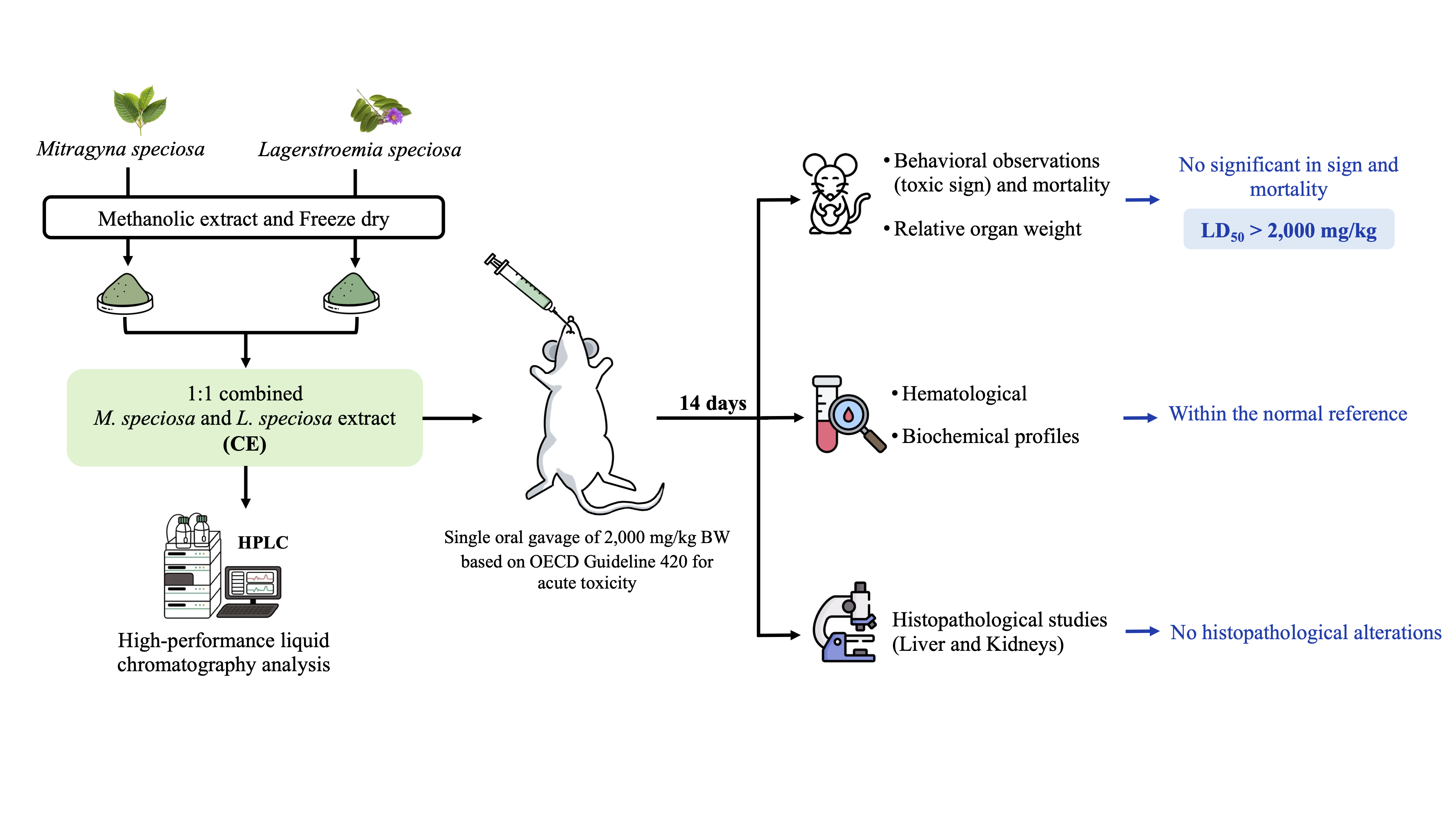
Mitragyna speciosa and Lagerstroemia speciosa are traditional herbal medicines and are claimed to possess hypoglycemic effects. The current study aimed to evaluate the acute toxicity of a 1:1 ratio combined extract (CE) of these herbal medicines, using an animal model, to provide a safety assessment for concomitant use. Methanolic extractions were performed on the dried powder of each herb, followed by quantification of the active compounds (mitragynine and corosolic acid) using high-performance liquid chromatography (HPLC) techniques. For the acute toxicity study, a total of 12 adult female Sprague-Dawley rats (5 weeks old, weighing 200 - 250 g) were randomly assigned to control and CE groups, with 6 rats in each group. A single oral dose of 2,000 mg/kg body weight of the CE was orally administered to the animals, with monitoring conducted in accordance with OECD Guideline 420. Behavioral, hematological and biochemical profiles were collected at the end of the 14-day study period, followed by a post-mortem histopathological examination to assess tissue integrity in vital organs. Statistical analyses were conducted to identify significant differences in all available parameters. The results demonstrated no signs of aberrant toxicity in any of the collected parameters, which was further confirmed by histopathological examination, focusing on the liver and kidneys. The study concluded that the CE at a 1:1 ratio exhibited a favorable safety profile and could be classified as having low toxicity based on the Globally Harmonized System. However, long-term studies are recommended to further confirm their safety profile.
HIGHLIGHTS
- Oral gavage of the combined extract of Mitragyna speciosa and Lagerstroemia speciosa (1:1) demonstrated no toxicity or mortality in a single-dose acute toxicity test.
- No abnormalities were observed in animals’ behavior, food intake, or water consumption compared to the control group.
- Similarly, biochemical and hematological parameters showed no significant differences between groups, remaining within the normal reference range.
- Histopathological analysis of the liver and kidneys revealed no signs of deformities in the combined extract group, providing strong evidence of no anatomical alterations.
GRAPHICAL ABSTRACT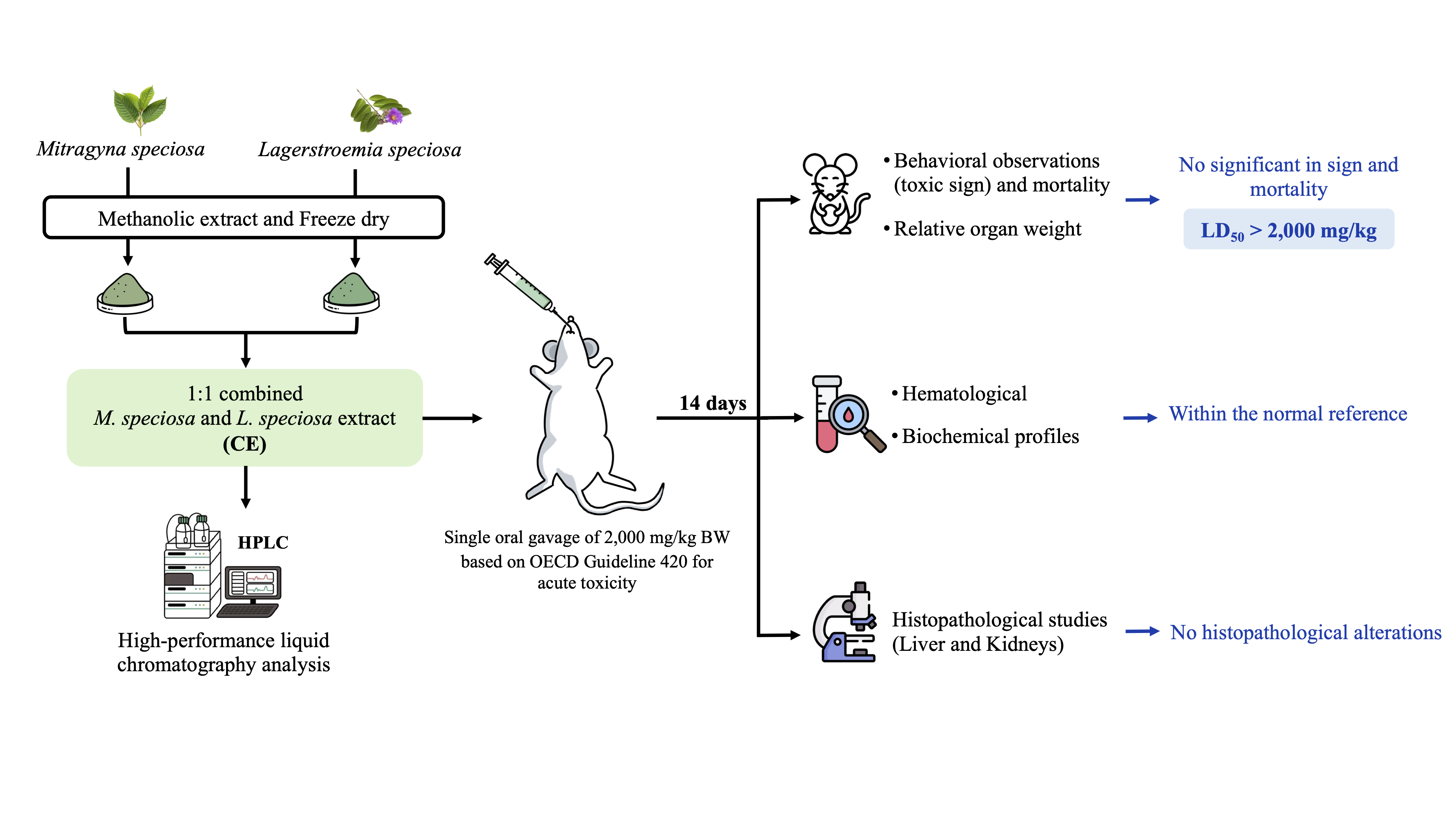
Downloads
References
H Wang, Y Song, J Ma, S Ma, L Shen, Y Huang, P Thangaraju, Z Basharat, Y Hu, Y Lin, AE Peden, SM Sawyer, H Zhang and Z Zou. Burden of non-communicable diseases among adolescents and young adults aged 10-24 years in the South-East Asia and Western Pacific regions, 1990-2019: A systematic analysis for the global burden of disease study 2019. Lancet Child Adolesc Health 2023; 7(9), 621-635.
J Bai, J Cui, F Shi and C Yu. Global epidemiological patterns in the burden of main non-communicable diseases, 1990-2019: Relationships with socio-demographic index. International Journal of Public Health 2023; 68, 1605502.
ED Canto, A Ceriello, L Ryden, M Ferrini, TB Hansen, O Schnell, E Standl and JW Beulens. Diabetes as a cardiovascular risk factor: An overview of global trends of macro and micro vascular complications. European Journal of Preventive Cardiology 2019; 26, 25-32.
W Fan. Epidemiology in diabetes mellitus and cardiovascular disease. Cardiovasc Endocrinol 2017; 6(1), 8-16.
G Piazzolla, A Vozza, S Volpe, A Bergamasco, V Triggiani, G Lisco, M Falconieri, C Tortorella, V Solfrizzi and C Sabba. Effectiveness and clinical benefits of new anti-diabetic drugs: A real life experience. Open Medicine (Warsaw) 2022; 17(1), 1203-1215.
AD Kinghorn, L Pan, JN Fletcher and H Chai. The relevance of higher plants in lead compound discovery programs. Journal of Natural Products 2011; 74(6), 1539-1555.
S Charoenratana, C Anukul and A Aramrattana. Attitudes towards Kratom use, decriminalization and the development of a community-based Kratom control mechanism in Southern Thailand. International Journal of Drug Policy 2021; 95, 103197.
K Peltzer and S Pengpid. The use of herbal medicines among chronic disease patients in Thailand: A cross-sectional survey. Journal of Multidisciplinary Healthcare 2019; 12, 573-582.
M Phumthum, K Srithi, A Inta, A Junsongduang, K Tangjitman, W Pongamornkul, C Trisonthi and H Balslev. Ethnomedicinal plant diversity in Thailand. Journal of Ethnopharmacology 2018; 214, 90-98.
N Kanjanahattakij, P Kwankhao, P Vathesatogkit, N Thongmung, Y Gleebbua, P Sritara and C Kitiyakara. Herbal or traditional medicine consumption in a Thai worker population: Pattern of use and therapeutic control in chronic diseases. BMC Complementary Medicine and Therapies 2019; 19(1), 258.
M Niyomdecha, T Kuttiyod and S Sanongkiet. α-glucosidase inhibition activities of crude extract and mitragynine from Mitragyna Speciosa Korth. International Journal of Health Sciences 2022; 6, 10254-10261.
D Fadly. Blood glucose response to kratom tea (Mitragyna speciosa Korth.). BIO Web of Conferences 2025; 153, 03015.
LD López-Murillo, M Gonzalez-Ortiz, E Martinez-Abundis, M Cortez-Navarrete and KG Perez-Rubio. Effect of banaba (Lagerstroemia speciosa) on metabolic syndrome, insulin sensitivity and insulin secretion. Journal of Medicinal Food 2022; 25(2), 177-182.
SJ Stohs, H Miller and GR Kaats. A review of the efficacy and safety of banaba (Lagerstroemia speciosa L.) and corosolic acid. Phytotherapy Research 2012; 26(3), 317-324.
T Nakaphan, M Teerachaisakul, S Puttum, K Sompimai and P Nootim. Traditional use of kratom (Mitragyna speciosa Korth) among folk healers in southern Thailand. Journal of Thai Traditional & Alternative Medicine 2018; 14(3), 274-285.
N Wiwatkunupakarn, C Aramrat, P Sanguanwai, Y Choksomngam, ME Gilder, W Jiraporncharoen, R McGready and C Angkurawaranon. The use of herbal medicine for hypertension in rural and urban Thailand: A cross sectional study. Journal of Herbal Medicine 2024; 47, 100916.
A Prasopthum, T Insawek and P Pouyfung. Herbal medicine use in Thai patients with type 2 diabetes mellitus and its association with glycemic control: A cross-sectional evaluation. Heliyon 2022; 8(10), e10790.
Z Hassan, D Singh, FW Suhaimi, NJY Chear, N Harun, CP See, G Kaur, NH Mat, SNS Bakar, NSM Yusof, VB Kasinather, MC Chawarski, V Murugaiyah and S Ramanathan. Evaluation of toxicity profile of kratom (Mitragyna speciosa Korth) decoction in rats. Regulatory Toxicology and Pharmacology 2023; 143, 105466.
N Thongsepee, S Amonyingcharoen, P Chamod, W Himakhun, K Sangpairoj, P Martviset, P Chantree and P Sornchuer. Modulatory effects of kratom extract on the gut microbiota of rats: Implications for health. BMC Complementary Medicine and Therapies 2025; 25(1), 85.
S Alkahtani, MS Hasnain, H Algamdy, NH Aljarba and A AlKahtane. Acute and sub-acute oral toxicity Lagerstroemia speciosa in sprague-dawley rats. Saudi Journal of Biological Sciences 2022; 29(3), 1585-1591.
AK Azad, MK Rahman and NK Sunzida. Acute oral toxicity study on Malaysian traditional herb: Lagerstroemia speciosa (Banaba). Journal of Pharmacognosy and Phytochemistry 2015; 4(4), 228-232.
S Limsuwanchote, J Wungsintaweekul, N Keawpradub, W Putalun, S Morimoto and H Tanaka. Development of indirect competitive ELISA for quantification of mitragynine in kratom (Mitragyna speciosa (Roxb.) Korth.). Forensic Science International 2014; 244, 70-77.
G Yusakul, P Saensom, N Mitsantia, C Pengdee and W Putalun. Development of a high-performance liquid chromatography for analysis of corosolic acid in Lagerstroemia species and their hypoglycemic potentials. Songklanakarin Journal of Science and Technology 2020; 42(2), 314-320.
Organisation for Economic Co-operation and Development. Test no. 420: Acute oral toxicity - fixed dose procedure. Organisation for Economic Co-operation and Development, Paris, France, 2002.
T Begum, MH Arzmi, MN Sarian, SAA Shah, SNH Azmi, ABMH Uddin, A Khatib and QU Ahmed. A review on multi-therapeutic potential of the (kratom) alkaloids mitragynine and 7-hydroxymitragynine: Experimental evidence and future perspectives. Kuwait Journal of Science 2025; 52(2), 100381.
BC An, SH Kwak, JY Ahn, HY Won, TH Kim, Y Ryu and MJ Chung. Identification of bioactive substances derived from the probiotic-induced bioconversion of Lagerstroemia speciosa Pers. leaf extract that have beneficial effects on diabetes and obesity. Microorganisms 2024; 12(9), 1848.
N Sengnon, P Vonghirundecha, W Chaichan, T Juengwatanatrakul, J Onthong, P Kitprasong, S Sriwiriyajan, S Chittrakarn, S Limsuwanchote and J Wungsintaweekul. Seasonal and geographic variation in alkaloid content of kratom (Korth.) Havil. from Thailand. Plants-Basel 2023; 12(4), 949.
W Thitikornpong, T Phadungcharoen and S Sukrong. Pharmacognostic evaluations of leaves. Journal of Medicinal Plants Research 2011; 5(8), 1330-1337.
R Ballesteros-Ramirez, P Lasso, C Uruena, J Saturno and S Fiorentino. Assessment of acute and chronic toxicity in Wistar rats (Rattus norvegicus) and New Zealand rabbits (Oryctolagus cuniculus) of an enriched polyphenol extract obtained from Caesalpinia spinosa. Journal of Toxicology 2024; 2024, 3769933.
A Bemidinezhad, SA Zojaji, S Taraz Jamshidi, M Mohammadi, MS Alavi and A Ghorbani. Evaluation of acute, subacute and subchronic toxicity of a hepatoprotective herbal formulation. Toxicology Reports 2023; 11, 452-459.
C Patel, P Shukla, S Pande, R Punamiya, K Ranch and SHS Boddu. Acute and sub-acute toxicity study of anti-obesity herbal granules in Sprague Dawley rats. Brazilian Journal of Biology 2022; 84, e264320.
SN Harizal, SM Mansor, J Hasnan, JKJ Tharakan and J Abdullah. Acute toxicity study of the standardized methanolic extract of Mitragyna speciosa Korth in rodent. Journal of Ethnopharmacology 2010; 131(2), 404-409.
MU Ilmie, H Jaafar, SM Mansor and JM Abdullah. Subchronic toxicity study of standardized methanolic extract of Korth in Sprague-Dawley Rats. Frontiers in Neuroscience 2015; 9, 189.
I Rafter, T Graberg, A Kotronen, L Strommer, CM Mattson, RW Kim, E Ehrenborg, HB Andersson, H Yki-Jarvinen, I Schuppe-Koistinen, B Ekblom, I Cotgreave and B Glinghammar. Isoform-specific alanine aminotransferase measurement can distinguish hepatic from extrahepatic injury in humans. International Journal of Molecular Medicine 2012; 30(5), 1241-1249.
MU Ilmie, H Jaafar, SM Mansor and JM Abdullah. Subchronic toxicity study of standardized methanolic extract of Mitragyna speciosa Korth in Sprague-Dawley Rats. Frontiers in Neuroscience 2015; 9, 189.
J Yang, YL Yu, Y Jin, Y Zhang and CQ Zheng. Clinical characteristics of drug-induced liver injury and primary biliary cirrhosis. World Journal of Gastroenterology 2016; 22(33), 7579-7586.
R Li, W Yang, Y Yin, X Ma, P Zhang and K Tao. 4-OI attenuates carbon tetrachloride-induced hepatic injury via regulating oxidative stress and the inflammatory response. Frontiers in Pharmacology 2021; 12, 651444.
L Zhang, C Liu, L Yin, C Huang and S Fan. Mangiferin relieves CCl4-induced liver fibrosis in mice. Scientific Reports 2023; 13(1), 4172.
X Xu, R Zhu, J Ying, M Zhao, X Wu, G Cao and K Wang. Nephrotoxicity of herbal medicine and its prevention. Frontiers in Pharmacology 2020; 11, 569551.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






