Chemical Profiling and Evaluation of Antibacterial and Anticancer Potential of Endophytic Aspergillus Terreus GCD2 Isolated from Garcinia Cowa Roxb. ex Choisy: In Vitro and Silico Approaches
DOI:
https://doi.org/10.48048/tis.2026.11378Keywords:
Garcinia cowa Roxb. ex Choisy, Aspergillus terreus, Endophytic fungi, Antibacterial, Cytotoxicity, Molecular docking, Aurasperone B, Fonsecinone BAbstract
Garcinia cowa Roxb. ex Choisy, a medicinal plant rich in bioactive secondary metabolites, has long been used in traditional medicine for its diverse therapeutic properties. Endophytic fungi associated with medicinal plants have emerged as promising sources of pharmacologically active compounds with antibacterial and anticancer potential in recent years. This study investigates an endophytic fungus isolated from G. cowa leaves, identified as Aspergillus terreus GCD2 (93.56% BLAST score) through macroscopic, microscopic, and molecular characterization. Crude extracts of GCD2, obtained using ethyl acetate, were profiled via LC-MS/MS, revealing Aurasperone B and Fonsecinone B as the predominant secondary metabolites. Antibacterial activity, evaluated using the Kirby-Bauer diffusion method, showed inhibition against methicillin-resistant Staphylococcus aureus (MRSA) with a zone diameter of 12.44 mm. Cytotoxicity testing demonstrated potent bioactivity, with LC₅₀ and IC₅₀ values of 79.45 μg/mL (Brine Shrimp Lethality Test) and 14.45 μg/mL (MTT assay on MCF-7 breast cancer cells), respectively. Molecular docking simulations indicated strong binding affinities of Aurasperone B and Fonsecinone B to MRSA and breast cancer-related receptors. Against MRSA, both compounds interacted with Asn120 adjacent to the canonical binding site, yielding docking scores of –7.11 and –7.09 kcal/mol (RMSD_refine 0.98 Å and 0.91 Å). Against breast cancer receptors, they bound to Arg146 and Tyr202, with docking scores of –6.00 and –6.20 kcal/mol (RMSD_refine 1.28 Å and 1.32 Å). These findings highlight A. terreus GCD2 as a promising source of antibacterial and anticancer agents. Further research focusing on compound isolation, mechanistic studies, and in vivo validation is warranted to advance its pharmaceutical potential.
HIGHLIGHTS
Aspergillus terreus GCD2 was isolated from Garcinia cowa Roxb. ex Choisy (Asam kandis) leaves. Novel bioactive metabolites were identified via LC-MS/MS, indicating strong antibacterial and anticancer potential. Antibacterial activity against methicillin-resistant Staphylococcus aureus (MRSA) showed measurable inhibition. Cytotoxicity was confirmed through Brine Shrimp Lethality Test (BSLT) and MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide] assays, demonstrating significant in vitro effects. Molecular docking revealed strong interactions of key compounds (e.g., Aurasperone B, Fonsecinone B) with MRSA- and breast cancer-related target proteins. Results support the potential of A. terreus GCD2 metabolites for pharmaceutical development as anti-MRSA and anticancer agents.
GRAPHICAL ABSTRACT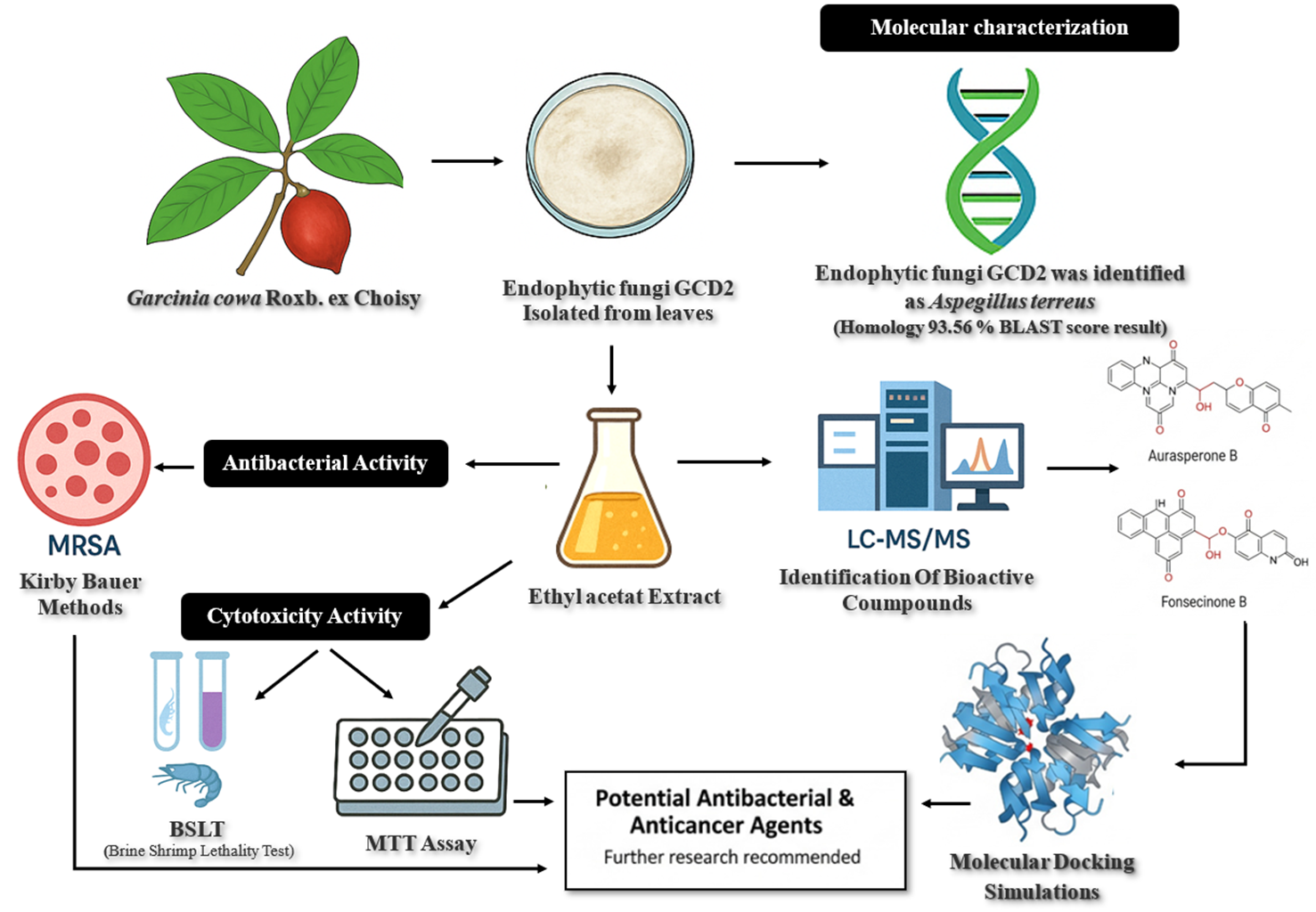
Downloads
References
K Panthong, W Pongcharoen, S Phongpaichit and WC Taylor. Tetraoxygenated xanthones from the fruits of Garcinia cowa. Phytochemistry 2006; 67(10), 999-1004.
T Ritthiwigrom, S Laphookhieo and SG Pyne. Chemical constituents and biological activities of Garcinia cowa Roxb. Maejo International Journal of Science and Technology 2013; 7(2), 212-231.
K Panthong, N Hutadilok-Towatana and A Panthong. A new tetraoxygenated xanthone, and other anti-inflammatory and antioxidant compounds from Garcinia cowa. Canadian Journal of Chemistry 2009; 87(11), 1636-1640.
A Sarma, P Sarmah, D Kashyap and A Kalita. Evaluation of nutraceutical properties and antioxidant activity of Garcinia cowa Roxb. ex Choisy fruits found in Assam (India). World Journal of Pharmacy and Pharmaceutical Sciences 2014; 3(12), 553-559.
A Chouni and S Paul. A comprehensive review of the phytochemical and pharmacological potential of an evergreen plant, Garcinia cowa. Chemistry & Biodiversity 2023; 20(2), e202200910.
NTK An, NV Hien, NT Thuy, DL Phuong, HG Bach, NT Tra, NQ Tung, PT Tham, BH Tai and TTT Thuy. Garcicowanones C-E, three new hydrated-geranylated xanthones from the roots of Garcinia cowa Roxb. ex Choisy, and their α-glucosidase inhibition activities. Natural Product Research 2023; 37(21), 3668-3676.
HL Devi, B Das, T Angami and B Kandpal. Garcinia cowa Roxb. ex Choisy (Clusiaceae). In: AA Waman and P Bohra (Eds.). Perennial underutilized horticultural species of India. Jaya Publishing House, Delhi, India, 2021, p. 131-143.
F Eshboev and D Egamberdieva. Medicinal plant-associated endophytic fungi: Metabolites and bioactivity. In: D Egamberdieva, JA Parray and K Davranov (Eds.). Plant endophytes and secondary metabolites. Academic Press, Cambridge, UK, 2024, p. 95-104.
A Yang, Y Hong, F Zhou, L Zhang, Y Zhu, C Wang, Y Hu, L Yu, L Chen and X Wang. Endophytic microbes from medicinal plants in Fenghuang mountain as a source of antibiotics. Molecules 2023; 28(17), 6301.
D Handayani, RI Muslim, N Syafni, MA Artasasta and R Riga. Endophytic fungi from medicinal plant Garcinia cowa Roxb. ex Choisy and their antibacterial activity. Journal of Applied Pharmaceutical Science 2024; 14(9), 182-188.
N Sandrawati, SP Hati, F Yunita, AE Putra, F Ismed, TE Tallei, T Hertiani and D Handayani. Antimicrobial and cytotoxic activities of marine sponge-derived fungal extracts isolated from Dactylospongia sp. Journal of Applied Pharmaceutical Science 2020; 10(4), 028-033.
J Mani, J Johnson, H Hosking, BE Hoyos, KB Walsh, P Neilsen and M Naiker. Bioassay-guided fractionation protocol for determining novel active compounds in selected Australian flora. Plants 2022; 11(21), 2886.
B Meyer, N Ferrigni, J Putnam, L Jacobsen, D Nichols and J McLaughlin. Brine shrimp: A convenient general bioassay for active plant constituents. Planta Medica 1982; 45(5), 31-34.
P Kumar, A Nagarajan and PD Uchil. Analysis of cell viability by the MTT assay. Cold Spring Harbor Protocols 2018; 2018(6), 469-471.
JV Meerloo, GJL Kaspers and J Cloos. Cell sensitivity assays: The MTT assay. In: IA Cree (Ed.). Cancer cell culture. Humana press, New Jersey, USA, 2011, p. 237-245.
D Handayani, W Rasyid, EN Zainudin and T Hertiani. Cytotoxic activity screening of fungal extracts derived from the West Sumatran marine sponge Haliclona fascigera to several human cell lines: HeLa, WiDr, T47D, and Vero. Journal of Applied Pharmaceutical Science 2018; 8, 055-058.
GS Guide, MassLynx 4.1, Available at: http://argenta2.chem.unr.edu/downloads/LCMS%20Waters %20MassLynx4.1%20 Manual.pdf, accessed January 2025.
W Zhang, X Li, X Ji and M Gouda. Use of LC-Q-TOF-MS for elucidating the flavonoids. In: M Gouda, X Li and Y He (Eds.). Plant chemical compositions and bioactivities. Humana Press, New York, 2024, p. 165-191.
W Li, J Zhang and LS Francis. Handbook of LC-MS bioanalysis: Best practices, experimental protocols, and regulations. Wiley, New Jersey, USA, 2013, p. 704.
B Zhou, JF Xiao, L Tuli and HW Ressom. LC-MS-based metabolomics. Molecular BioSystems 2012; 8(2), 470-481.
TE Tallei and BJ Kolondam. DNA barcoding of Sangihe Nutmeg (Myristica fragrans) using matK gene. HAYATI Journal of Biosciences 2015; 22(1), 41-47.
K Tamura, G Stecher and S Kumar. MEGA11: Molecular evolutionary genetics analysis version 11. Molecular Biology and Evolution 2021; 38(7), 3022-3027.
RW Birkinshaw, J Gong, CS Luo, D Lio, CA White, MA Anderson, P Blombery, G Lessene, IJ Majewski, R Thijssen, AW Roberts, DCS Huang, PM Colman and PE Czabotar. Structures of BCL-2 in complex with venetoclax reveal the molecular basis of resistance mutations. Nature Communications 2019; 10(1), 2385.
RCSB PDB - 6O0K, Crystal structure of BCL-2 with venetoclax, Available at: https://www.rcsb.org/structure/6O0K, accessed June 2025.
RCSB PDB - 6Q9N, Crystal structure of PBP2a from MRSA in complex with piperacillin and quinazolinone, Available at: https://www.rcsb.org/structure/6Q9N, accessed June 2025.
J Janardhanan, R Bouley, S Martínez-Caballero, Z Peng, M Batuecas-Mordillo, JE Meisel, D Ding, VA Schroeder, WR Wolter, KV Mahasenan, JA Hermoso, S Mobashery and M Chang. The quinazolinone allosteric inhibitor of PBP 2a synergizes with piperacillin and tazobactam against methicillin-resistant staphylococcus aureus. Antimicrobial Agents and Chemotherapy 2019; 63(5), e02637.
MC Nagtilak. Molecular docking: A highly efficient method for structure-based drug designing. In: VA Chavan, PS Sagar, R Shukla, E Laxminarayana, A Debnath, S Supriya, U Das and S Vijayakumar (Eds.). Futuristic trends in chemical material sciences & nano technology. Iterative International Publishers, Michigan, USA, 2024, p. 97-106.
Molecular Docking With Molecular Operating Environment (MOE), Available at: https://www.chemcomp.com/ moe/help/latest/index_tut.html, accessed November 2024.
BD Systèmes. Discovery Studio Visualizer Software. Accelrys, San Diego, 2021.
E Udayan and JJ Gnanadoss. Potential of endophytic fungi as therapeutics: Antibiotics, antiviral and anticancer properties. Research Journal of Biotechnology 2023; 18(6), 132-145.
SK Deshmukh, L Dufossé, H Chhipa, S Saxena, GB Mahajan and MK Gupta. Fungal endophytes: A Potential source of antibacterial compounds. Journal of Fungi 2022; 8(2), 164.
SJ Li, X Zhang, XH Wang and CQ Zhao. Novel natural compounds from endophytic fungi with anticancer activity. European Journal of Medicinal Chemistry 2018; 156, 316-343.
D Handayani, MA Artasasta, D Mutia, N Atikah, Rustini and TE Tallei. Antimicrobial and cytotoxic activities screening of fungal secondary metabolites isolated from marine sponge Callyspongia sp. AACL Bioflux 2021; 14(1), 249-258.
MA Artasasta, Y Yanwirasti, M Taher, A Djamaan, NP Ariantari, RA Edrada-Ebel and D Handayani. Apoptotic activity of new oxisterigmatocystin derivatives from the marine-derived fungus Aspergillus nomius NC06. Marine Drugs 2021; 19(11), 631.
Aspergillus terreus, Doctor Fungus, Available at: https://drfungus.org/knowledge-base/aspergillus-terreus/, accessed June 2025.
C Lass-Flörl, AM Dietl, DP Kontoyiannis and M Brock. Aspergillus terreus species complex. Clinical Microbiology Reviews 2021; 34(4), e0031120.
P Kapli, Z Yang and MJ Telford. Phylogenetic tree building in the genomic age. Nature Reviews Genetics 2020; 21(7), 428-444.
MS Lestari, T Himawan, AL Abadi and R Retnowati. Toxicity and phytochemistry test of methanol extract of several plants from papua using Brine Shrimp Lethality Test (BSLT). Journal of Chemical and Pharmaceutical Research 2015; 7(4), 866-872.
H Niksic, F Becic, E Koric, I Gusic, E Omeragic, S Muratovic, B Miladinovic and K Duric. Cytotoxicity screening of Thymus vulgaris L. essential oil in brine shrimp nauplii and cancer cell lines. Scientific Reports 2021; 11(1), 13178.
S Ghosal, K Biswas and DK Chakrabarti. Toxic naphtho-.gamma.-pyrones from Aspergillus niger, Journal of Agricultural and Food Chemistry 1979; 27(6), 1347-1351.
S Padhi, M Masi, SK Panda, W Luyten, A Cimmino, K Tayung and A Evidente. Evidente, antimicrobial secondary metabolites of an endolichenic Aspergillus niger isolated from lichen thallus of Parmotrema ravum. Natural Product Research 2020; 34(18), 2573-2580.
Dulcioic acid | C7H12O8, Available at: https://www.chemspider.com/Chemical-Structure.29365152.html, accessed July 2025.
8-Benzyl-3-(4-biphenylyl)-4,9-dimethyl-7H-furo[2,3-f]chromen-7-one,C32H24O3, https://www.chemspider.com/Chemical-Structure.1385615.html, accessed July 2025.
PubChem, Malformin A, Available at: https://pubchem.ncbi.nlm.nih.gov/compound/4005, accessed June 2025.
PubChem, Aurasperone B, Available at: https://pubchem.ncbi.nlm.nih.gov/compound/179522, accessed June 2025).
PubChem, Fonsecinone B, Available at: https://pubchem.ncbi.nlm.nih.gov/compound/101301306, accessed June 2025.
Aurasperone A, C32H26O10, Available at: https://www.chemspider.com/Chemical-Structure.2341316.html, accessed July 2025.
K Palaniveloo, KH Ong, H Satriawan, SA Razak, S Suciati, HY Hung, S Hirayama, M Rizman-Idid, JK Tan, YS Yong and SM Phang. In vitro and in silico cholinesterase inhibitory potential of metabolites from Laurencia snackeyi (Weber-van Bosse) M. Masuda. 3 Biotech 2023; 13(10), 337.
SA Attique, M Hassan, M Usman, RM Atif, S Mahboob, KA Al-Ghanim, M Bilal and MZ Nawaz. A molecular d ocking approach to evaluate the pharmacological properties of natural and synthetic treatment candidates for use against hypertension. International Journal of Environmental Research and Public Health 2019; 16(6), 923.
H Purnomo. Kimia Komputasi: Molecular docking plants-penambatan molekul plants (protein ligand ant system). Pustaka Pelajar, Yogyakarta, 2011.
S Mukherjee, TE Balius and RC Rizzo. Docking validation resources: Protein family and ligand flexibility experiments. Journal of Chemical Information and Modeling 2010; 50(11), 1986-2000.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






