Agarwood Tea Leaves: Uncovering the Antioxidant Phytochemicals and Their Protective Role Against Hepatocyte Oxidative
DOI:
https://doi.org/10.48048/tis.2025.11129Keywords:
Agarwood tea leaves, HepG2, Reactive oxygen species, CytoprotectiveAbstract
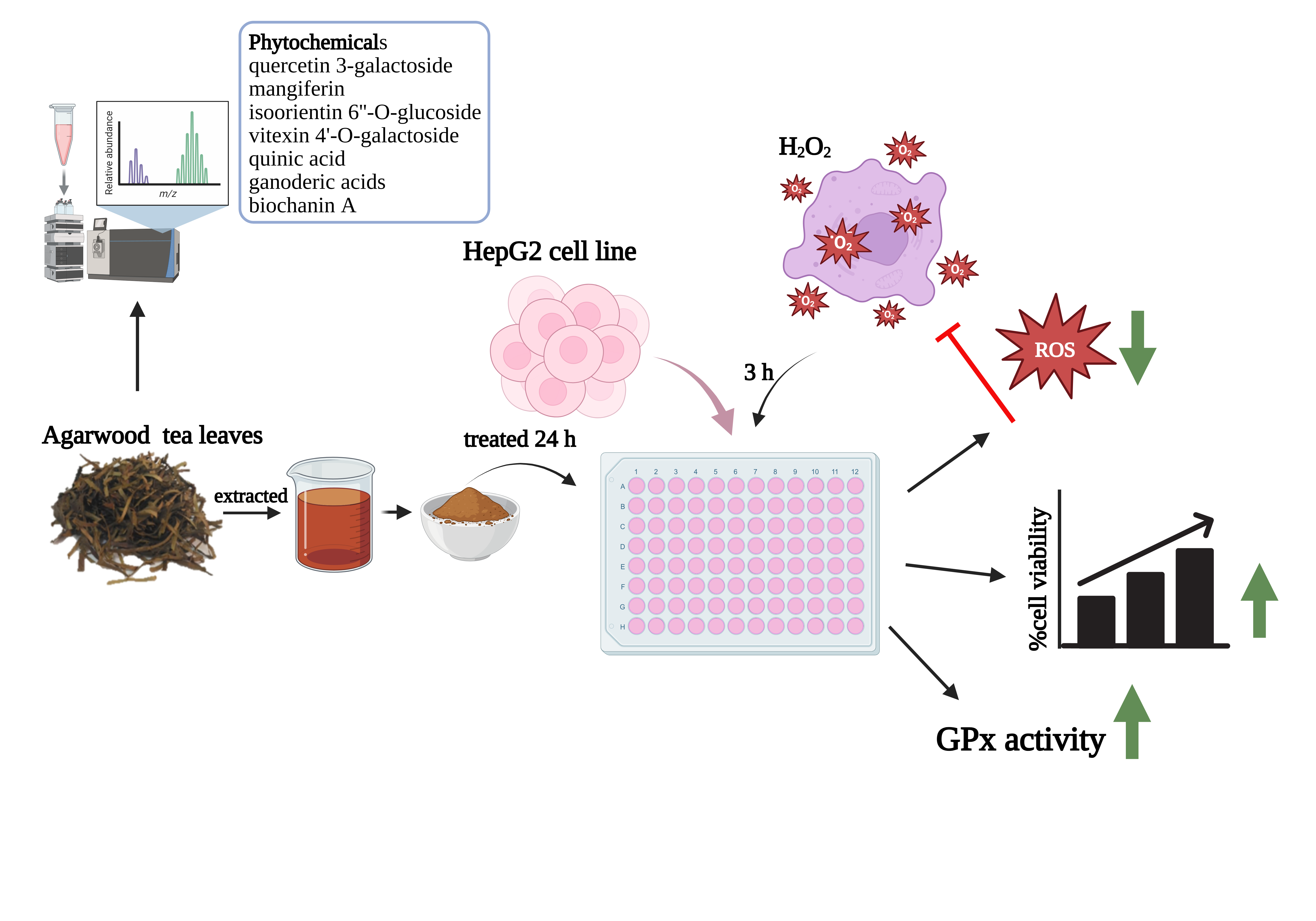
Liver cells play a key role in metabolizing nutrients, drugs, and foreign substances, during which reactive oxygen species (ROS) are generated. Excess ROS can lead to oxidative stress, contributing to liver diseases. Thus, controlling ROS is important for liver cell protection. This study aimed to investigate the chemical composition and antioxidant potential of Aquilaria subintegra (agarwood) leaves extract and its protective effects against hydrogen peroxide (H2O2)-induced oxidative stress in HepG2 cells. Phytochemical profiling was performed using LC-MS/MS QTOF. HepG2 cells were pre-treated with non-toxic concentrations of the extract for 24 h, followed by exposure to H2O2. Cell viability was assessed by MTT assay. Intracellular ROS was measured using 2',7'-dichlorofluorescein (DCF), while antioxidant enzyme activities, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), were determined using commercial kits. The results of the study showed that the phytochemicals found in the leaves of A. subintegra are diverse. It is mainly a substance in the group of flavonoids, benzopyrans, and organooxygen. The major antioxidants classified include quercetin 3-galactoside, mangiferin, isoorientin 6''-O-glucoside, vitexin 4'-O-galactoside, quinic acid, ganoderic acids and biochanin A. Agarwood tea leaves extract showed an effect in preventing H2O2-induced oxidative stress in HepG2 cells at concentrations ranging from 1.95 to 15.63 μg/mL, with a significant reduction in ROS levels at concentrations ranging from 1.95 to 7.81 μg/mL compared to the control group. The extract did not influence the activity of SOD and CAT enzymes; however, it significantly enhanced the activity of GPx enzyme within the concentration range of 7.81 to 62.50 μg/mL. In conclusion, the results of the study suggest that agarwood tea leaves extract may have an antioxidative effect on liver cells. This may be due to the presence of high amounts of antioxidants, which may be a promising strategy for mitigating oxidative damage in liver-related diseases.
HIGHLIGHTS
- subintegra leaves extract demonstrates powerful antioxidant potential, effectively protecting liver cells from oxidative injury induced by hydrogen peroxide.
- Comprehensive LC-MS/MS QTOF analysis identified a unique phytochemical fingerprint, rich in flavonoids, benzopyrans, and organooxygen compounds with known bioactivities.
- The extract selectively enhances GPx activity, suggesting a targeted cellular defense mechanism without disrupting other major antioxidant enzymes.
- Significant ROS scavenging effects were observed at low micromolar concentrations, highlighting its potency and therapeutic relevance in oxidative stress models.
- This study provides novel mechanistic insight into the hepatoprotective action of agarwood tea leaves, opening new avenues for natural antioxidant-based liver therapies.
GRAPHICAL ABSTRACT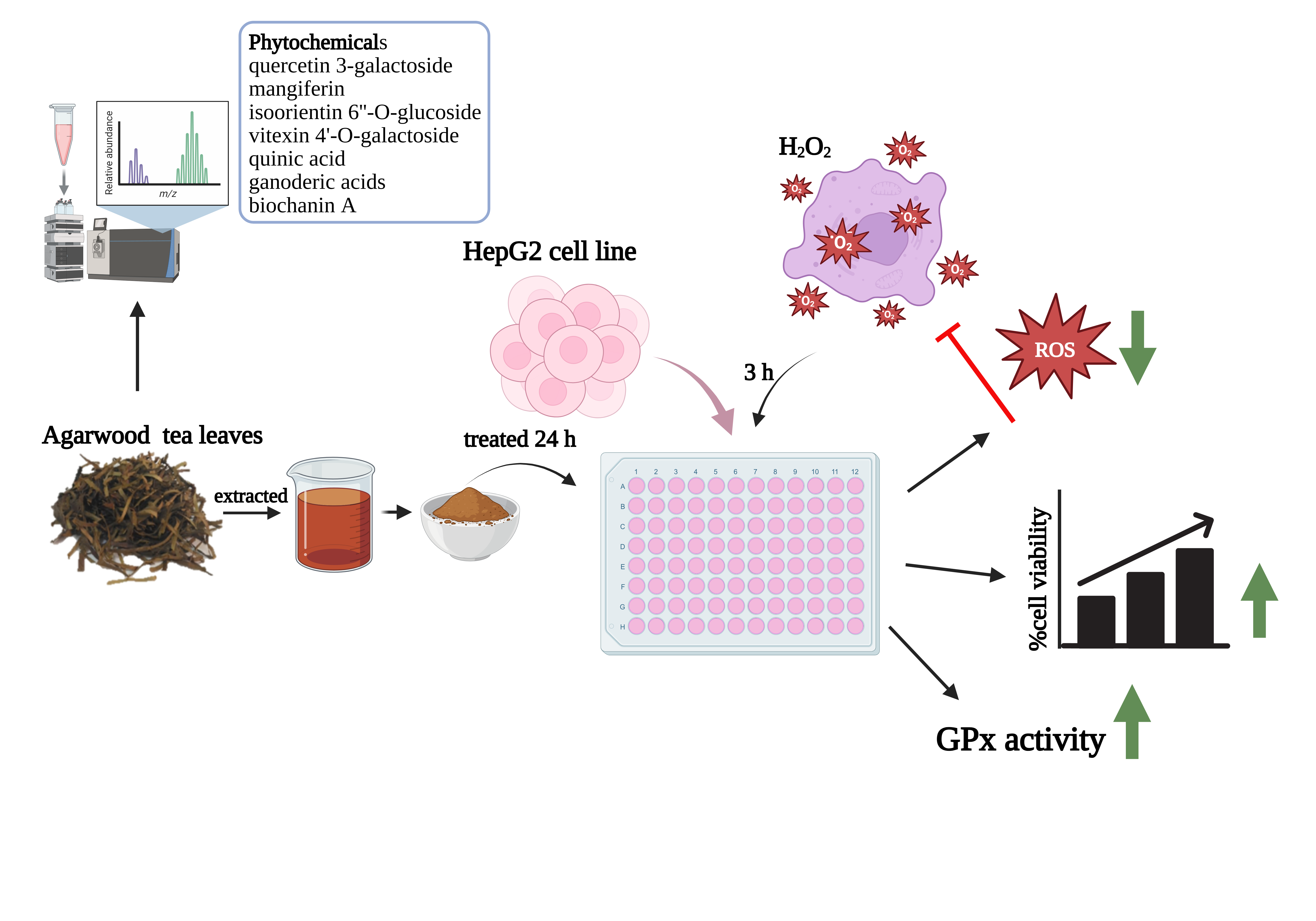
Downloads
References
M Valko, D Leibfritz, J Moncol, MTD Cronin, M Mazur and J Telser. Free radicals and antioxidants in normal physiological functions and human disease. The International Journal of Biochemistry and Cell Biology 2007; 39(1), 44-84.
GA Engwa, FN EnNwekegwa and BN Nkeh-Chungag. Free radicals, oxidative stress-related diseases and antioxidant supplementation. Alternative Therapies in Health and Medicine 2022; 28(1), 114-128.
J Chiang. Liver physiology: Metabolism and detoxification. In: ML McManus and RN Mitchell (Eds.). Pathobiology of human disease. Elsevier, San Diego, 2014, p. 1770-1782.
H Cichoż-Lach and A Michalak. Oxidative stress as a crucial factor in liver diseases. World Journal of Gastroenterology 2014; 20(25), 8082-8091.
CF Lima, M Fernandes-Ferreira and C Pereira-Wilson. Phenolic compounds protect HepG2 cells from oxidative damage: Relevance of glutathione levels. Life Sciences 2006; 79(21), 2056-2068.
MA Martín, S Ramos, R Mateos, ABG Serrano, M Izquierdo-Pulido, L Bravo and L Goya. Protection of human HepG2 cells against oxidative stress by cocoa phenolic extract. Journal of Agricultural and Food Chemistry 2008; 56(17), 7765-7772.
T Osawa. Nephroprotective and hepatoprotective effects of curcuminoids. In: BB Aggarwal, YJ Surh and S Shishodia (Eds.). The molecular targets and therapeutic uses of curcumin in health and disease. Springer, Boston, 2007, p. 407-423.
YZHY Hashim, PG Kerr, P Abbas and HM Salleh. Aquilaria spp. (agarwood) as source of health beneficial compounds: A review of traditional use, phytochemistry and pharmacology. Journal of Ethnopharmacology 2016; 189, 331-360.
AZ Adam, SY Lee and R Mohamed. Pharmacological properties of agarwood tea derived from Aquilaria (Thymelaeaceae) leaves: An emerging contemporary herbal drink. Journal of Herbal Medicine 2017; 10, 37-44.
K Vakati, H Rahman MC Eswaraiah and AM Dutta. Evaluation of hepatoprotective activity of ethanolic extract of Aquilaria agallocha leaves (EEAA) against CCl4 induced hepatic damage in rat. Scholars Journal of Applied Medical Sciences 2013; 1(1), 9-12.
PA Kroon, MN Clifford, A Crozier, AJ Day, JL Donovan, C Manach and G Williamson. How should we assess the effects of exposure to dietary polyphenols in vitro? The American Journal of Clinical Nutrition 2004; 80(1), 15-21.
C Aguilar-Garcia, G Gavino, M Baragaño-Mosqueda, P Hevia and VC Gavino. Correlation of tocopherol, tocotrienol, γ-oryzanol and total polyphenol content in rice bran with different antioxidant capacity assays. Food Chemistry 2007; 102(4), 1228-1232.
S Adusei, JK Otchere, P Oteng, RQ Mensah and E Tei-Mensah. Phytochemical analysis, antioxidant and metal chelating capacity of Tetrapleura tetraptera. Heliyon 2019; 5(11), e02762.
T Ito, M Kakino, S Tazawa, T Watarai, M Oyama, H Maruyama, Y Araki, H Hara and M Iinuma. Quantification of polyphenols and pharmacological analysis of water and ethanol-based extracts of cultivated agarwood leaves. Journal of Nutritional Science and Vitaminology 2012; 58(2), 136-142.
M Renis, L Calandra, C Scifo, B Tomasello, V Cardile, L Vanella, R Bei, LL Fauci and F Galvano. Response of cell cycle/stress-related protein expression and DNA damage upon treatment of CaCo2 cells with anthocyanins. British Journal of Nutrition 2008; 100(1), 27-35.
ZM Rashid, NAC Zahari, WA Wan-Nadilah, AKK Yunusa and NH Mahmod. Effect of extraction solvents on chemical characteristics and DPPH scavenging activity of Aquilaria Malaccensis leaves. Bioscience Research 2019; 16, 213-222.
YL Yang, HD Sun, J Yang, CZ Liu, CZ Kang, J Liu and LP Guo. Tandem mass spectrometry (MS/MS) molecular networking guided profiling of small molecules from Aquilaria sinensis (Lour.) Gilg leaves and their bioactivity evaluation. Phytochemical Analysis 2024; 35(1), 135-145.
Y Li, N Sheng, L Wang, S Li, J Chen and X Lai. Analysis of 2-(2-Phenylethyl)chromones by UPLC-ESI-QTOF-MS and multivariate statistical methods in wild and cultivated agarwood. International Journal of Molecular Sciences 2016; 17(5), 771.
C Tian, A Wu, C Yao, Z Song, L Shen and L He. UHPLC-QTOF-MS based metabolite profiling analysis and the correlation with biological properties of wild and artificial agarwood. Journal of Pharmaceutical and Biomedical Analysis 2021; 194, 113782.
S Chen, X Wang, Y Cheng, H Gao and X Chen. A review of classification, biosynthesis, biological activities and potential applications of flavonoids. Molecules 2023; 28(13), 4982.
RJ Nijveldt, EV Nood, DEV Hoorn, PG Boelens, KV Norren and PAV Leeuwen. Flavonoids: A review of probable mechanisms of action and potential applications. The American Journal of Clinical Nutrition 2001; 74(4), 418-425.
PS Sellamuthu, P Arulselvan, BP Muniappan, S Fakurazi and M Kandasamy. Mangiferin from Salacia chinensis prevents oxidative stress and protects pancreatic β-cells in streptozotocin-induced diabetic rats. Journal of Medical Food 2013; 16(8), 719-727.
M Imran, MS Arshad, MS Butt, JH Kwon, MU Arshad and MT Sultan. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids in Health and Disease 2017; 16(1), 84.
J Das, J Ghosh, A Roy and PC Sil. Mangiferin exerts hepatoprotective activity against D-galactosamine induced acute toxicity and oxidative/nitrosative stress via Nrf2-NFκB pathways. Toxicology and Applied Pharmacology 2012; 260(1), 35-47.
MAA Ela, AM El-Lakany, MS Abdel-Kader, SI Alqasoumi, SM Shams-El-Din and HM Hammoda. New quinic acid derivatives from hepatoprotective Inula crithmoides root extract. Helvetica Chimica Acta 2012; 95(1), 61-66.
T Benali, S Bakrim, R Ghchime, N Benkhaira, NE Omari, A Balahbib, D Taha, G Zengin, MM Hasan, S Bibi and A Bouyahya. Pharmacological insights into the multifaceted biological properties of quinic acid. Biotechnology and Genetic Engineering Reviews 2024; 40(4), 3408-3437.
J Kolniak-Ostek, J Oszmiański, A Szyjka, H Moreira and E Barg. Anticancer and antioxidant activities in Ganoderma lucidum Wild mushrooms in Poland, as well as their phenolic and triterpenoid compounds. International Journal of Molecular Sciences 2022; 23(16), 9359.
ZM Jiang, HB Qiu, SQ Wang, J Guo, ZW Yang and SB Zhou. Ganoderic acid A potentiates the antioxidant effect and protection of mitochondrial membranes and reduces the apoptosis rate in primary hippocampal neurons in magnesium free medium. Pharmazie 2018; 73(2), 87-91.
PV Anuranjana, F Beegum, KP Divya, KT George, GL Viswanatha, PG Nayak, A Kanwal, A Kishore, RR Shenoy and K Nandakumar. Mechanisms behind the pharmacological application of biochanin-A: A review. F1000Research 2023; 12, 107.
S Kamonwannasit, N Nantapong, P Kumkrai, P Luecha, S Kupittayanant and N Chudapongse. Antibacterial activity of Aquilaria crassna leaf extract against Staphylococcus epidermidis by disruption of cell wall. Annals of Clinical Microbiology and Antimicrobials 2013; 12, 20.
AS Khalil, AA Rahim, KK Taha and KB Abdallah. Characterization of methanolic extracts of agarwood leaves. Journal of Applied and Industrial Sciences 2013; 1(3), 78-88.
NNAN Wil, NAM Omar, NA Ibrahim and SN Tajuddin. In vitro antioxidant activity and phytochemical screening of Aquilaria malaccensis leaf extracts. Journal of Chemical and Pharmaceutical Research 2014; 6(12), 688-693.
KW Kong, S Mat-Junit, N Aminudin, FA Hassan, A Ismail and AA Aziz. Protective effects of the extracts of Barringtonia racemosa shoots against oxidative damage in HepG2 cells. PeerJ 2016; 4, e1628.
B Halliwell. Role of free radicals in the neurodegenerative diseases: Therapeutic implications for antioxidant treatment. Drugs and Aging 2001; 18(9), 685-716.
HY Ju, SC Chen, KJ Wu, HC Kuo, YC Hseu, H Ching and CR Wu. Antioxidant phenolic profile from ethyl acetate fraction of Fructus Ligustri Lucidi with protection against hydrogen peroxide-induced oxidative damage in SH-SY5Y cells. Food and Chemical Toxicology 2012; 50(3-4), 492-502.
SY Park, JY Je and CB Ahn. Phenolic composition and hepatoprotective activities of Allium hookeri against hydrogen-peroxide-induced oxidative stress in cultured hepatocytes. Journal of Food Biochemistry 2016; 40(3), 284-293.
S Salla, R Sunkara, S Ogutu, LT Walker and M Verghese. Antioxidant activity of papaya seed extracts against H2O2 induced oxidative stress in HepG2 cells. LWT 2016; 66, 293-297.
OM Ighodaro and OA Akinloye. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria Journal of Medicine 2018; 54(4), 287-293.
M Bošković, T Vovk, M Saje, K Goričar, V Dolžan, BK Plesničar and I Grabnar. Association of SOD2, GPX1, CAT, and TNF genetic polymorphisms with oxidative stress, neurochemistry, psychopathology, and extrapyramidal symptoms in schizophrenia. Neurochemical Research 2013; 38(2), 433-442.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






