Bacterial Cellulose Production in Thai Red Tea Fermentation: Role of Sucrose-Based Sugar Combinations as Carbon Sources
DOI:
https://doi.org/10.48048/tis.2026.11128Keywords:
Bacterial cellulose, Kombucha fermentation, Carbon source combinations, Thai red tea, Cellulose production, Structural, Mechanical propertiesAbstract
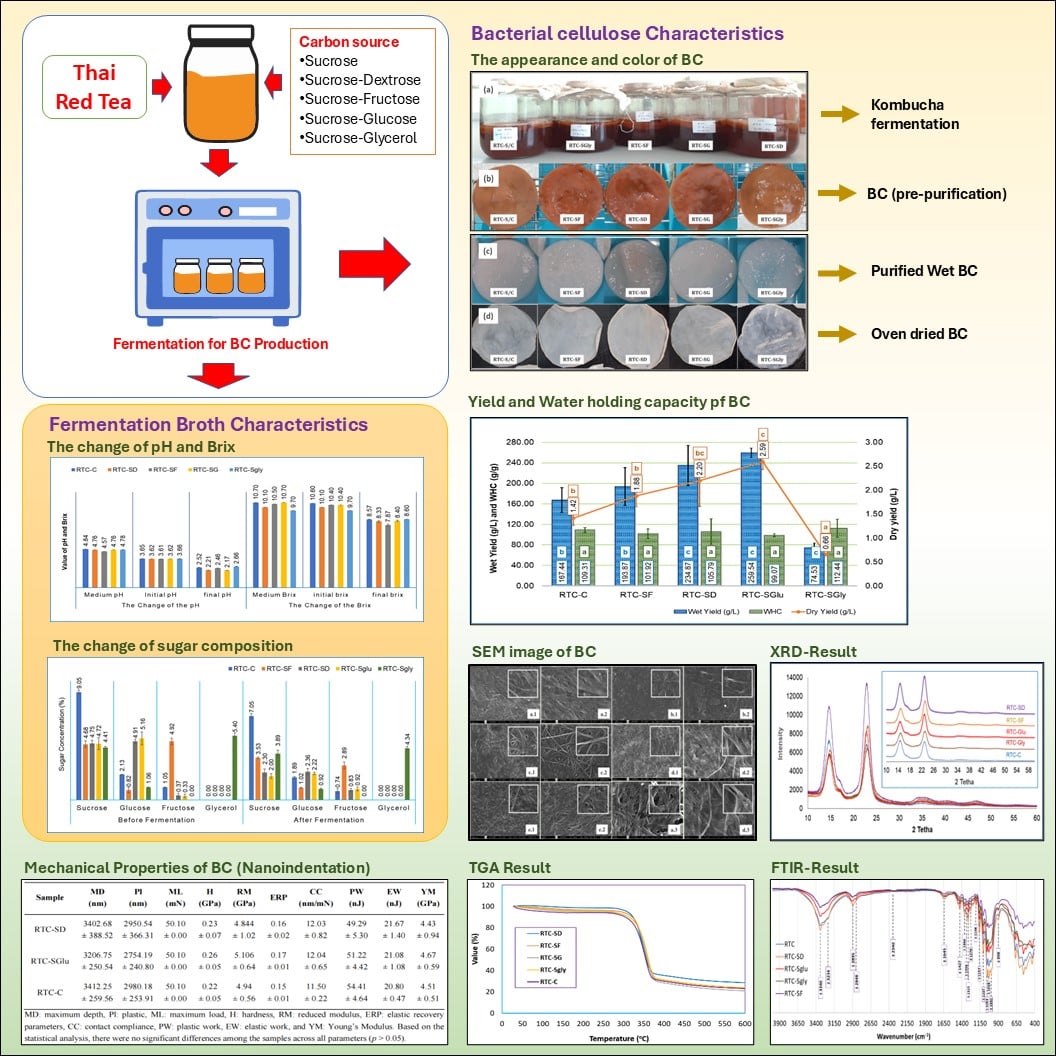
Bacterial cellulose (BC) is a versatile biopolymer produced via microbial fermentation, valued for its purity, strength, and biocompatibility. In this study, BC was synthesized using Thai red tea as the fermentation medium, combined with various carbon source combinations: Control (RTC-C), sucrose-dextrose (RTC-SD), sucrose-glucose (RTC-SGlu), sucrose-fructose (RTC-SF), and sucrose-glycerol (RTC-SGly). Fermentation was carried out for 15 days at ~30 °C using a tea solution (1% w/v), total sugar content of 10% (w/v), and 10% (v/v) kombucha culture. The resulting BC was evaluated for yield, morphology, structure, and thermal/mechanical properties using SEM, FTIR, XRD, and TGA analysis. Carbon source combinations significantly influenced BC production, with wet and dry yields ranging from 74.53 g/L and 0.66 g/L (RTC-SGly) to 259.54 g/L and 2.59 g/L (RTC-SGlu), respectively. The sucrose-glucose combination yielded the highest productivity. SEM revealed a uniform nanofiber network with fiber diameters ranging from 29.70 ± 5.17 nm to 40.39 ± 8.65 nm. FTIR and XRD confirmed the formation of cellulose type I, with crystallinity indexes ranging from 84.74% (RTC-SGly) to 88.35% (RTC-SF). Thermal and mechanical properties were comparable across all samples. These findings demonstrate that specific carbon source combinations, particularly sucrose-glucose and sucrose-dextrose, can significantly enhance BC yield without compromising its structural integrity, highlighting their potential for industrial-scale applications.
HIGHLIGHTS
- Thai red tea fermentation serves as an effective substrate for bacterial cellulose production.
- Fermentation medium composition, especially carbon source combinations, plays a key role in influencing BC yield and characteristics.
- Sucrose combined with glucose or dextrose significantly improves BC productivity over sucrose alone.
- Structural, thermal, and mechanical properties remain stable across treatments, despite enhanced yield.
GRAPHICAL ABSTRACT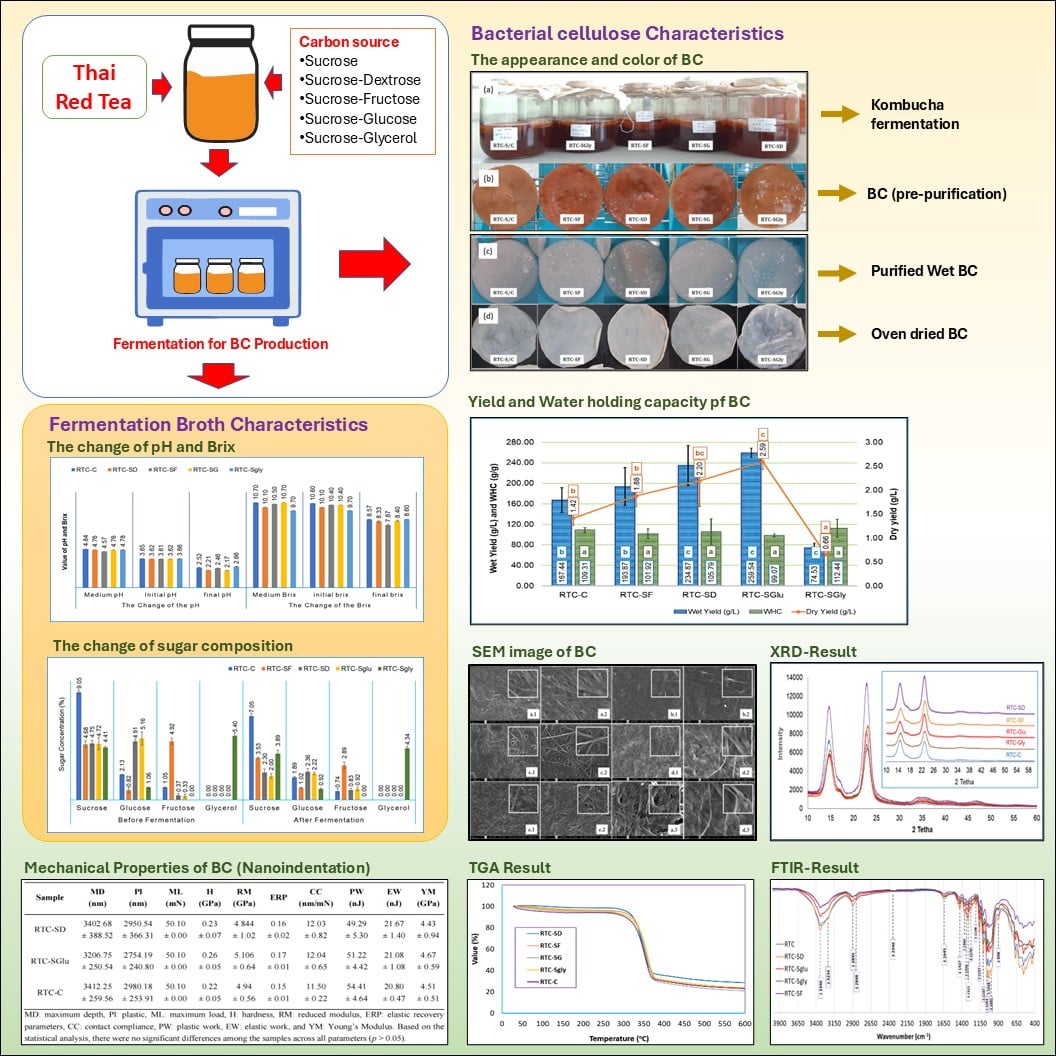
Downloads
References
D Lin, Z Liu, R. Shen, S Chen and X Yang. Bacterial cellulose in food industry: Current research and future prospects. International Journal of Bioliogical Macromolecules 2020; 158, 10007-1019.
C Zhong. Industrial-Scale Production and Applications of Bacterial Cellulose. Frontiers in Bioengineering and Biotechnology 2020; 8, 605374.
L Rosson, B Tan, W Best and N Byrne. Applications of regenerated bacterial cellulose: A review. Cellulose 2024; 31, 10165-10190.
D Absharina, M Padri, C Veres and C Vagvolgyi. Bacterial Cellulose: From biofabrication to applications in sustainable fashion and vegan leather. Fermentation 2025; 11(1), 23.
R Mehrotra, S Sharma, N Shree and K Kaur. Bacterial cellulose: An ecological alternative as a biotextile. Biosciences Biotechnology Research Asia 2023; 20(2), 449-463.
X Feng, Z Ge, Y Wang, X Xia, B Zhao and M Dong. Production and characterization of bacterial cellulose from kombucha-fermented soy whey. Food Production, Processing and Nutrition 2024; 6, 20.
LFA Amorim, L Li, AP Gomes, R Fangueiro and IC Gouveia. Sustainable bacterial cellulose production by low-cost feedstock: Evaluation of apple and tea by-products as alternative sources of nutrients. Cellulose 2023; 30, 5589-5606.
MZ Treviño-Garza, AS Guerrero-Medina, RA Gonzalez-Sanchez, C Garcia-Gomez, A Guzman-Velasco, JG Baez-Gonzalez and JM Marquez-Reyes. Production of microbial cellulose films from green tea (Camellia Sinensis) kombucha with various carbon sources. Coating 2020; 10(11), 1132.
G Buldum and A Mantalaris. Systematic understanding of recent developments in bacterial cellulose biosynthesis at genetic bioprocess and product levels. International Journal of Molecular Sciences 2021; 22(13), 7192.
A Pandey, A Singh and MK Singh. Novel low-cost green method for production bacterial cellulose. Polymer Bulletin 2024; 81, 6721-6741.
B Hungund, S Prabhu, C Shetty, S Acharya, V Prabhu, and SG Gupta. Production of bacterial cellulose from Gluconacetobacter persimmonis GH-2 using dual and cheaper carbon sources. Journal of Microbial & Biochemical Technology 2013; 5(2), 31-33.
G Singh, P Gauba and G Mathur. Bacterial Cellulose Production by Acetobacter aceti MTCC 2623 Using Different Carbon Sources. Current Applied Science and Technology 2024; 24(6), 0260805.
J Amorim, K Liao, A Mandal, AF de S Costa, E Roumeli and LA Sarubbo. Impact of carbon source on bacterial cellulose network architecture and prolonged lidocaine release. Polymers 2024; 16(21), 3021.
K Aswini, NO Gopal and S Uthandi. Optimized culture conditions for bacterial cellulose production by Acetobacter senegalensis MA1. BMC Biotechnology 2020; 20(1), 46.
ST Schrecker and PA Gostomski. Determining the water holding capacity of microbial cellulose. Biotechnology letters 2005; 27, 1435-1438.
TG Volova, SV Prudnikova, EG Kiselev, IV Nemtsev, AD Vasiliev, AP Kuzmin and EI Shishatskaya. Bacterial Cellulose (BC) and BC Composites: Production and Properties. Nanomaterials 2022; 12(2), 192.
SA Sardjono, H Suryanto, Aminnudin and M Muhajir. Crystallinity and morphology of the bacterial nanocellulose membrane extracted from pineapple peel waste using high-pressure homogenizer. AIP Conference Proceedings 2019; 2120(1), 080015.
RJ Roberts, RC Rowe and P York. The Poisson’s ratio of microcrystalline cellulose. International Journal of Pharmaceutics 1994; 105(2), 177-180.
FA Rabbani, S Yasin, T Iqbal and U Farooq. Experimental Study of mechanical properties of polypropylene random copolymer and Rice-Husk-Based biocomposite by using nanoindentation. Materials (Basel) 2022; 15(5), 1956.
KR Lee, K Jo, KS Ra, HJ Suh and K Hong. Kombucha fermentation using commercial kombucha pellicle and culture broth as starter. Food Science and Technology 2022; 42, 70020.
K Neffe-Skocińska, B Sionek, I Ścibisz and D Kołożyn-Krajewska. Acid contents and the effect of fermentation condition of Kombucha tea beverages on physicochemical, microbiological and sensory properties. CyTA - Journal of Food 2017; 15(4), 1321588.
B Vohra, S Fazry, F Sairi and BA Othman. Effects of medium variation and fermentation time towards the pH level and ethanol content of Kombucha. AIP Conference Proceedings 2019; 2111, 040008.
AQ Chong, NL Chin, RA Talib and RK Basha. Modelling pH dynamics, SCOBY biomass formation, and acetic acid production of kombucha fermentation using black, green, and oolong teas. Processes 2024; 12(7), 1301.
M Muzaifa, S Rohaya, C Nilda and KR Harahap. Kombucha fermentation from cascara with addition of red dragon fruit (Hylocereus polyrhizus): Analysis of alcohol content and total soluble solid. In: Proceedings of the International Conference on Tropical Agrifood, Feed and Fuel, Samarinda East Kalimantan, Indonesia.
E Zubaidah, RA Ifadah and CA Afgani. Changes in chemichal characteristics of kombucha from various cultivars of snake fruit during fermentation. International Conference on Green Agro-industry and Bioeconomy 2019; 230, 012098.
KN Sinamo, S Ginting and S Pratama. Effect of sugar concentration and fermentation time on secang kombucha drink. IOP Conference Series: Earth and Environmental Science 2022; 977, 012080.
E Ball. Hydrolysis of sucrose by autoclaving media, a neglected aspect in the technique of culture of plant tissues. Bulletin of the Torrey Botanical Club 1953; 80(5), 409-411.
JH de Lange. Significance of autoclaving-induced toxicity from and hydrolysis of carbohydrates in in vitro studies of pollen germination and tube growth. South African Journal of Botany 1989; 55(1), 1-5.
M Gao, D Ploessl and Z Shao. Enhancing the co-utilization of biomass-derived mixed sugars by yeasts. Frontiers in Microbiology 2018; 9, 03264.
J Lee. Exploring sucrose fermentation: Microorganisms, Biochemical Pathways, and Applications. Fermentation Technology 2023; 12(1), 1000166.
B Wang, K Rutherfurd-Markwick, XX Zhang and AN Mutukumira. Kombucha: Production and microbiological research. Foods 2022; 11(21), 3456.
H Deka, PP Sarmah, A Devi, P Tamuly and T Karak. Changes in major catechins, caffeine, and antioxidant activity during CTC processing of black tea from North East India. RSC Advances 2021; 11(19), 11457-11467.
A Ito and E Yanase. Study into the chemical changes of tea leaf polyphenols during japanese black tea processing. Food Research International 2022; 160, 111731.
K Izawa, Y Amino, M Kohmura, Y Ueda and M Kuroda. Human-environment interactions – taste. Comprehensive Natural Products II: Chemistry and Biology 2010; 4, 631-671.
AS Amarasekara, D Wang, and TL Grady. A comparison of kombucha SCOBY bacterial cellulose purification methods. SN Applied Sciences 2020; 2, 240.
ASM Kamal, MI Misnon and F Fadil. The effect of sodium hydroxide concentration on yield and properties of Bacterial Cellulose membranes. IOP Conference Series: Materials Science and Engineering 2020; 732, 012064.
SMAS Keshk and K Sameshima. Evaluation of different carbon sources for bacterial cellulose production. African Journal of Biotechnology 2005; 6(4), 478-482.
JDP Amorim, AFS Costa, CJS Galdino, GM Vinhas, EMS Santos and LA Sarubbo. Bacterial cellulose production using fruit residues as substract to industrial application. Chemical Engineering Transactions 2019; 74, 1165-1170.
YH Jin, T Lee, JR Kim, YE Choi and C Park. Improved production of bacterial cellulose from waste glycerol through investigation of inhibitory effects of crude glycerol-derived compounds by Gluconacetobacter xylinus. Journal of Industrial and Engineering Chemistry 2019; 75, 158-163.
MN Thorat and SG Dastager. High yield production of cellulose by a: Komagataeibacter rhaeticus PG2 strain isolated from pomegranate as a new host. RSC Advances 2018; 52, 29797-29805.
MJ Tabaii and G Emtiazi. Comparison of bacterial cellulose production among different strains and fermented media. Applied Food Biotechnology 2016; 3(1), 35-41.
YE Agustin, KS Padmawijaya, HF Rixwari and VAS Yuniharto. Glycerol as an additional carbon source for bacterial cellulose synthesis. IOP Conference Series: Earth and Environmental Science 2018; 141, 012001.
A Adnan, GR Nair, MC Lay, JE Swan, and R Umar. Glycerol as a cheaper carbon source in Bacterial Cellulose (BC) production by Gluconacetobacter xylinus Dsm46604 in Batch Fermentation System. Malaysian Journal of Analytical Sciences 2015; 19(5), 1131-1136.
JM Wu and RH Liu. Cost-effective production of bacterial cellulose in static cultures using distillery wastewater. Journal of Bioscience and Bioengineering 2013; 115(3), 284-290.
NH Avcioglu, M Birben and IS Bilkay. Optimization and physicochemical characterization of enhanced microbial cellulose production with a new Kombucha consortium. Process Biochemistry 2021; 108, 60-68.
E Tsouko, C Kourmentza, D Ladakis, N Kopsahelis, I Mandala, S Papanikolaou, F Paloukis, V Alves and A Koutinas. Bacterial cellulose production from industrial waste and by-product streams. International Journal of Molecular Sciences 2015; 16(7), 14832-14849.
RAH Almihyawi, E Musazade, N Alhussany, S Zhang and H Chen. Production and characterization of bacterial cellulose by Rhizobium sp. isolated from bean root. Scientific Reports 2024; 14, 10848.
C Ugurel and H Ogut. Optimization of bacterial cellulose production by Komagataeibacter rhaeticus K23. Fibers 2024; 12(3), 29.
MP Illa, M Khandelwal and CS Sharma. Modulated dehydration for enhanced anodic performance of bacterial cellulose derived carbon nanofibers. ChemistrySelect 2019; 21(4), 6642-6650.
TA Nguyen and XC Nguyen. Bacterial Cellulose-Based biofilm forming agent extracted from vietnamese nata-de-coco tree by ultrasonic vibration method: Structure and properties. Journal of Chemistry 2022; 2022(1), 7502796.
R Brandes, L De Souza, C Carminatti and D Recouvreux. Production with a high yield of bacterial cellulose nanocrystals by enzymatic hydrolysis. International Journal of Nanoscience 2020; 19(3), 1950015.
D Abol-Fotouh, MA Hassan, H Shokry, A Roig, MS Azab and AEHB Kashyout. Bacterial nanocellulose from agro-industrial wastes: Low-cost and enhanced production by Komagataeibacter saccharivorans MD1. Scientific Reports 2020; 10, 3491.
M Yilmaz and Y Goksungur. Optimization of bacterial cellulose production from waste figs by Komagataeibacter xylinus. Fermentation 2024; 10(9), 466.
Y Raiszadeh-Jahromi, M Rezazadeh-Bari, H Almasi and S Amiri. Optimization of bacterial cellulose production by Komagataeibacter xylinus PTCC 1734 in a low-cost medium using optimal combined design. Journal of Food Science and Technology 2020; 57(7), 2524-2533.
SS Wang, YH Han, JL Chen, DC Zhang, XX Shi, YX Ye, DL Chen and M Li. Insights into bacterial cellulose biosynthesis from different carbon sources and the associated biochemical transformation pathways in Komagataeibacter sp. W1. Polymers (Basel) 2018; 10(9), 963.
NAM Razali, RM Sohaimi, RNIR Othman, N Abdullah, SZN Demon, L Jasmani, WMZW Yunus, WMHW Ya’acob, EM Salleh, MN Norizan and NA Halim. Comparative study on extraction of cellulose fiber from rice straw waste from chemo-mechanical and pulping method. Polymers (Basel) 2022; 14(3), 387.
MA Adekoya, S Liu, SS Oluyamo, OT Oyeleye and RT Ogundare. Influence of size classifications on the crystallinity index of Albizia gummifera cellulose. Heliyon 2022; 8(12), 12019.
X Liu, L Cao, S Wang, L Huang, Y Zhang, M Tian, X Li and J Zhang. Isolation and characterization of bacterial cellulose produced from soybean whey and soybean hydrolyzate. Scientific Reports 2023; 13, 16024.
S Srivastava and G Mathur. Komagataeibacter saccharivorans strain BC-G1: An alternative strain for production of bacterial cellulose. Biologia (Bratisl) 2022; 77, 3657-3668.
E Leonarski, K Cesca, E Zanella, BU Stambuk, D de Oliveira and P Poletto. Production of kombucha-like beverage and bacterial cellulose by acerola byproduct as raw material. LWT 2021; 135, 110075.
A Fatima, P Ortiz-Albo, LA Neves, FX Nascimento and JG Crespo. Biosynthesis and characterization of bacterial cellulose membranes presenting relevant characteristics for air/gas filtration. Journal of Membrane Science 2023; 674, 121509.
D Ciolacu, F Ciolacu and VI Popa. Amorphous cellulose - structure and characterization. Cellulose Chemistry and Technology 2011; 45(1-2), 13-21.
D Gaspar, SN Fernandes, AG De Oliveira, JG Fernandes, P Grey, RV Pontes, L Pereira, R Martins, MH Godinho and E Fortunato. Nanocrystalline cellulose applied simultaneously as the gate dielectric and the substrate in flexible field effect transistors. Nanotechnology 2014; 25(9), 094008.
SNNS Azmi, Z’Asyiqin Samsu, ASFM Asnawi, H Ariffin and SS Syed Abdullah. The production and characterization of bacterial cellulose pellicles obtained from oil palm frond juice and their conversion to nanofibrillated cellulose. Carbohydrate Polymer Technologies and Applications 2023; 5, 100327.
T Hossen, CK Kundu, BRR Pranto, MS Rahi, R Chanda, S Mollick, AB Siddique and HA Begum. Synthesis, characterization, and cytotoxicity studies of nanocellulose extracted from okra (Abelmoschus esculentus) fiber. Heliyon 2024; 10(3), 25270.
OS Samuel and AM Adefusika. Influence of size classifications on the structural and solid-state characterization of cellulose materials. In: R Pascual and MEE Martin (Eds.). Cellulose. IntechOpen, London, 2019.
X Zeng, J Liu, J Chen, Q Wang, Z Li and H Wang. Screening of the common culture conditions affecting crystallinity of bacterial cellulose. Journal of Industrial Microbiology and Biotechnology 2011; 38(12), 1993-1999.
K Thielemans, YD Bondt, L Comer, J Raes, N Everaert, BF Sels and CM Courtin. Decreasing the crystallinity and degree of polymerization of cellulose increases its susceptibility to enzymatic hydrolysis and fermentation by colon microbiota. Foods 2023; 12(5), 1100.
C Molina-Ramirez, C Enciso, M Torres-Taborda, R Zuluaga, P Ganan, OJ Rojas and C Castro. Effects of alternative energy sources on bacterial cellulose characteristics produced by Komagataeibacter medellinensis. International Journal of Biological Macromolecules 2018; 117, 735-741.
S Agustin, ET Wahyuni, Suparmo, Supriyadi and MN Cahyanto. Production and characterization of bacterial cellulose-alginate biocomposites as food packaging material. Food Research 2021; 5(6), 204-210.
P Singhsa, R Narain and H Manuspiya. Physical structure variations of bacterial cellulose produced by different Komagataeibacter xylinus strains and carbon sources in static and agitated conditions. Cellulose 2018; 25, 1571-1581.
S Charoenrak, S Charumanee, P Sirisa-ard, S Bovonsombut, L Kumdhitiahutsawakul, S Kiatkarun, W Pathom-Aree, T Chitov and S Bovonsombut. Nanobacterial cellulose from kombucha fermentation as a potential protective carrier of Lactobacillus plantarum under simulated gastrointestinal tract conditions. Polymers (Basel) 2023; 15(6), 1356.
GN Balistreri, IR Campbell, X Li, J Amorim, S Zhang, E Nance and E Roumeli. Bacterial cellulose nanoparticles as a sustainable drug delivery platform for protein-based therapeutics. RSC Applied Polymers 2024; 2(2), 172-183.
Y Jia, X Wang, M Huo, X Zhai, F Li and C Zhong. Preparation and characterization of a novel bacterial cellulose/chitosan bio-hydrogel. Nanomaterials and Nanotechnology 2017; 7(4), 184798041770717.
G Gayathri and G Srinikethan. Bacterial cellulose production by K. saccharivorans BC1 strain using crude distillery effluent as cheap and cost-effective nutrient medium. International Journal of Biological Macromolecules 2019; 138, 950-957.
NM Nurazzi, MRM Asyraf, M Rayung, MNF Norrrahim, SS Shazleen, MSA Rani, AR Shafi, HA Aisyah, MHM Radzi, FA Sabaruddin, RA Ilyas, ES Zainudin and K Abdan. Thermogravimetric analysis properties of cellulosic natural fiber polymer composites: A review on influence of chemical treatments. Polymers (Basel) 2021; 13(16), 2710.
S Mohamad, LC Abdullah, SS Jamari, SSOA Edrus, MM Aung and SFS Mohamad. Production and characterization of bacterial cellulose nanofiber by Acetobacter xylinum 0416 using only oil palm frond juice as fermentation medium. Journal of Natural Fibers 2022; 19(17), 16005-16016.
SRZ Teixeira, EMD Reis, GP Apati, MM Meier, AL Nogueira, MCF Garcia, ALDS Schneider, APT Pezzin and LM Porto. Biosynthesis and functionalization of bacterial cellulose membranes with cerium nitrate and silver nanoparticles. Materials Research 2019; 22(1), 0054.
YA Gismatulina and VV Budaeva. Cellulose nitrates-blended composites from bacterial and plant-based celluloses. Polymers (Basel) 2024; 16(9), 1183.
F Mohammadkazemi, M Azin and A Ashori. Production of bacterial cellulose using different carbon sources and culture media. Carbohydrate Polymers 2015; 117, 518-523.
K Potivara and M Phisalaphong. Development and characterization of bacterial cellulose reinforced with natural rubber. Materials (Basel) 2019; 12(14), 2323.
R Jenkhongkarn and M Phisalaphong. Effect of reduction methods on the properties of composite films of bacterial cellulose-silver nanoparticles. Polymers (Basel) 2023; 15(14), 2996.
BC Tureck, HG Hackbarth, EZ Neves, MCF Garcia, GP Apati, DDOS Recouvreux, APT Pezzin and ALDS Schneider. Obtaining and characterization of bacterial cellulose synthesized by Komagataeibacter hansenii from alternative sources of nitrogen and carbon. Matéria (Rio de Janeiro) 2021; 26(4), 1392.
NF Lima, GM Maciel, IDAA Fernandes and CWI Haminiuk. Optimising the production process of bacterial nanocellulose: Impact on growth and bioactive compounds. Food Technology and Biotechnology 2023; 61(4), 494-504.
VM Vasconcellos and CS Farinas. The effect of the drying process on the properties of bacterial cellulose films from Gluconacetobacter hansenii. Chemical Engineering Transactions 2018; 64, 145-150
C Molina-Ramirez, C Castro, R Zuluaga and P Ganan. Physical characterization of bacterial cellulose produced by Komagataeibacter medellinensis using food supply chain waste and agricultural by-products as alternative low-cost feedstocks. Journal of Polymers and the Environment 2018; 26, 830-837.
V Chibrikov, PM Pieczywek, J Cybulska and A Zdunek. Evaluation of elasto-plastic properties of bacterial cellulose-hemicellulose composite films. Industrial Crops and Products 2023; 205, 117578.
I Betlej, R Salerno-Kochan, KJ Krajewski, J Zawadzki and P Boruszewski. The influence of culture medium components on the physical and mechanical properties of cellulose synthesized by kombucha microorganisms. BioResources 2020; 15(2), 3125-3135.
A Krystynowicz, W Czaja, A Wiktorowska-Jezierska, M Gonçalves-Miśkiewicz, M Turkiewicz and S Bielecki. Factors affecting the yield and properties of bacterial cellulose. Journal of Industrial Microbiology and Biotechnolog 2002; 29(4), 189-195.
M Zeng, A Laromaine and A Roig. Bacterial cellulose films: Influence of bacterial strain and drying route on film properties. Cellulose 2014; 21, 4455-4469.
SQ Chen, P Lopez-Sanchez, D Wang, D Mikkelsen and MJ Gidley. Mechanical properties of bacterial cellulose synthesised by diverse strains of the genus Komagataeibacter. Food Hydrocolloids 2018; 81, 87-95.
H Suryanto, M Muhajir, TA Sutrisno, Mudjiono, N Zakia and U Yanuhar. The mechanical strength and morphology of bacterial cellulose films: The effect of NaOH concentration. IOP Conference Series: Materials Science and Engineering 2019; 515, 012053.
SQ Chen, OW Meldrum, Q Liao, Z Li, X Cao, L Guo, S Zhang, J Zhu and L Li. The influence of alkaline treatment on the mechanical and structural properties of bacterial cellulose. Carbohydrate Polymers 2021; 271, 118431.
YY Wang, XQ Zhao, DM Li, YM Wu, F Wahid, YY Xie and C Zhong. Review on the strategies for enhancing mechanical properties of bacterial cellulose. Journal of Materials Science 2023; 58, 15265-15293.
N Dusunceli and OU Colak. Modelling effects of degree of crystallinity on mechanical behavior of semicrystalline polymers. International Journal of Plasticity 2008; 24(7), 1224-1242.
H Oliver-Ortega, S Geng, FX Espinach, K Oksman and F Vilaseca. Bacterial cellulose network from kombucha fermentation impregnated with emulsion-polymerized poly(methyl methacrylate) to form nanocomposite. Polymers (Basel) 2021; 13(4), 664.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






