Anti-migration and Anti-invasion Effects of Mitragyna Speciosa Polar Extract on A549 Cell
DOI:
https://doi.org/10.48048/tis.2025.10150Keywords:
Non-Small cell lung cancer, Mitragyna speciosa, MTT assay, Wound healing, Transwell invasion assay, Non-small cell lung cancer, Mitragyna speciosa, MTT assay, Wound healing, Transwell invasion assayAbstract
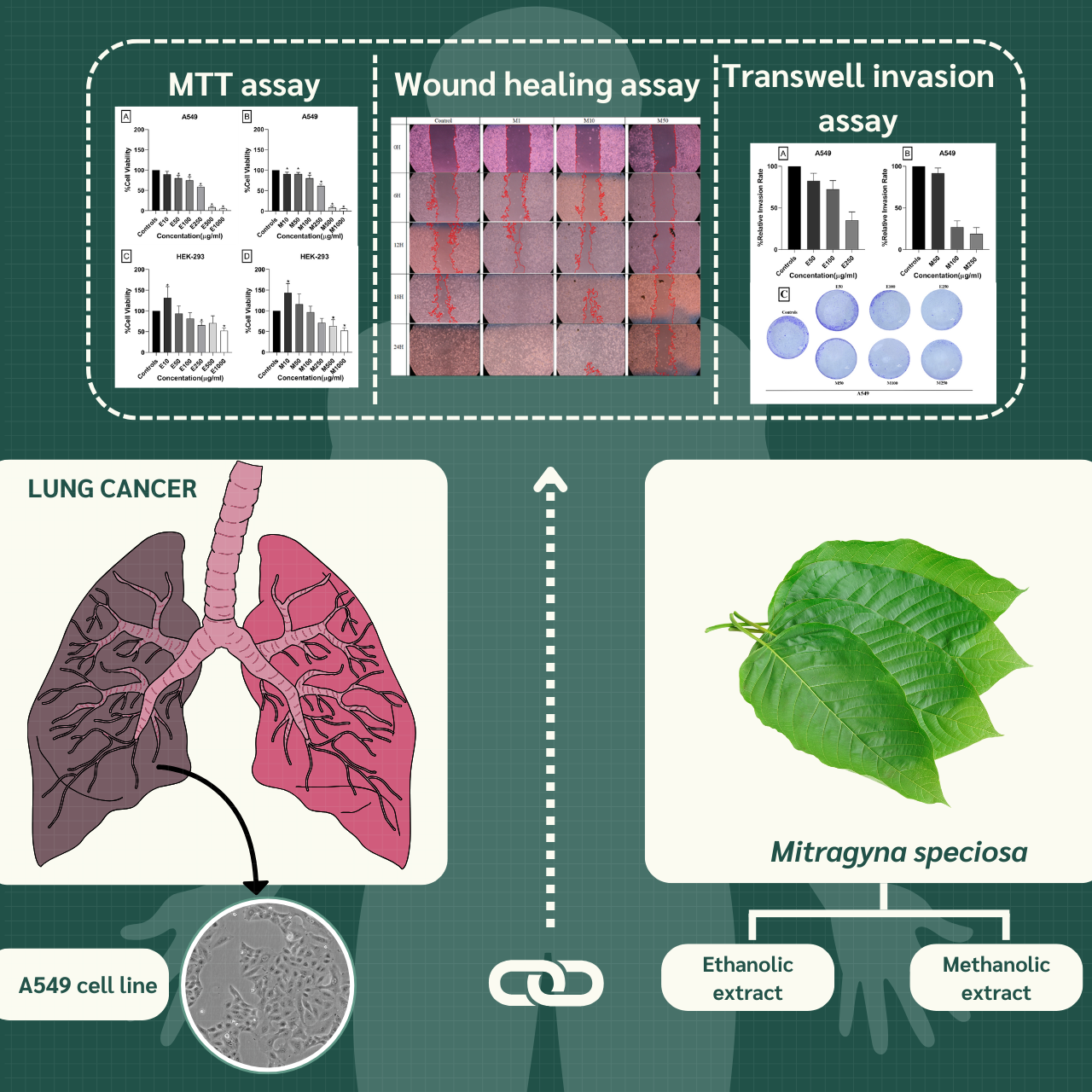
Mitragyna speciosa (commonly known as Kratom) is a tropical plant native to Southeast Asia and a member of the Rubiaceae family. This study aimed to investigate the anticancer potential of crude M. speciosa leaf extracts against non-small cell lung cancer (NSCLC) cells. Dried leaves were extracted using 95% ethanol or 95% methanol at a 1:10 w/v ratio via maceration. The cytotoxic effects of the extracts were evaluated using the MTT assay, and results were expressed as IC50 values. Cell migration and invasion were assessed using wound healing and Transwell invasion assays, respectively. All experiments were conducted in triplicate. The MTT assay revealed that both ethanolic and methanolic extracts significantly reduced the viability of A549 cells in a dose-dependent manner. The methanolic extract exhibited greater potency, with an IC50 of 283 µg/mL, compared to 295 µg/mL for the ethanolic extract. In the wound healing assay, both extracts inhibited A549 cell migration, with the methanolic extract showing stronger suppression at concentrations of 50, 100, and 250 µg/mL. The ethanolic extract also reduced migration but required higher concentrations to achieve similar effects. Transwell invasion assay results further demonstrated that both extracts suppressed cell invasion, with the methanolic extract being more effective at lower concentrations (50 and 100 µg/mL). Furthermore, real-time RT-PCR analysis revealed that the extracts upregulated E-cadherin and, in the methanolic group, downregulated MMP-2 expression, suggesting suppression of epithelial-mesenchymal transition (EMT)-associated invasion. These findings highlight the potential of M. speciosa as a source of bioactive compounds with anti-migratory and anti-invasive properties. Further investigation is warranted to elucidate the underlying mechanisms and evaluate its potential for development as a natural anticancer agent.
HIGHLIGHTS
- This study investigates anti-metastatic effects of Mitragyna speciosa extract on NSCLC cell.
- The methanolic extract demonstrated higher efficacy compared to the ethanolic extract.
- The extract modulates key signaling pathways related to cancer metastasis.
- Findings suggest potential therapeutic implications for lung cancer treatment.
GRAPHICAL ABSTRACT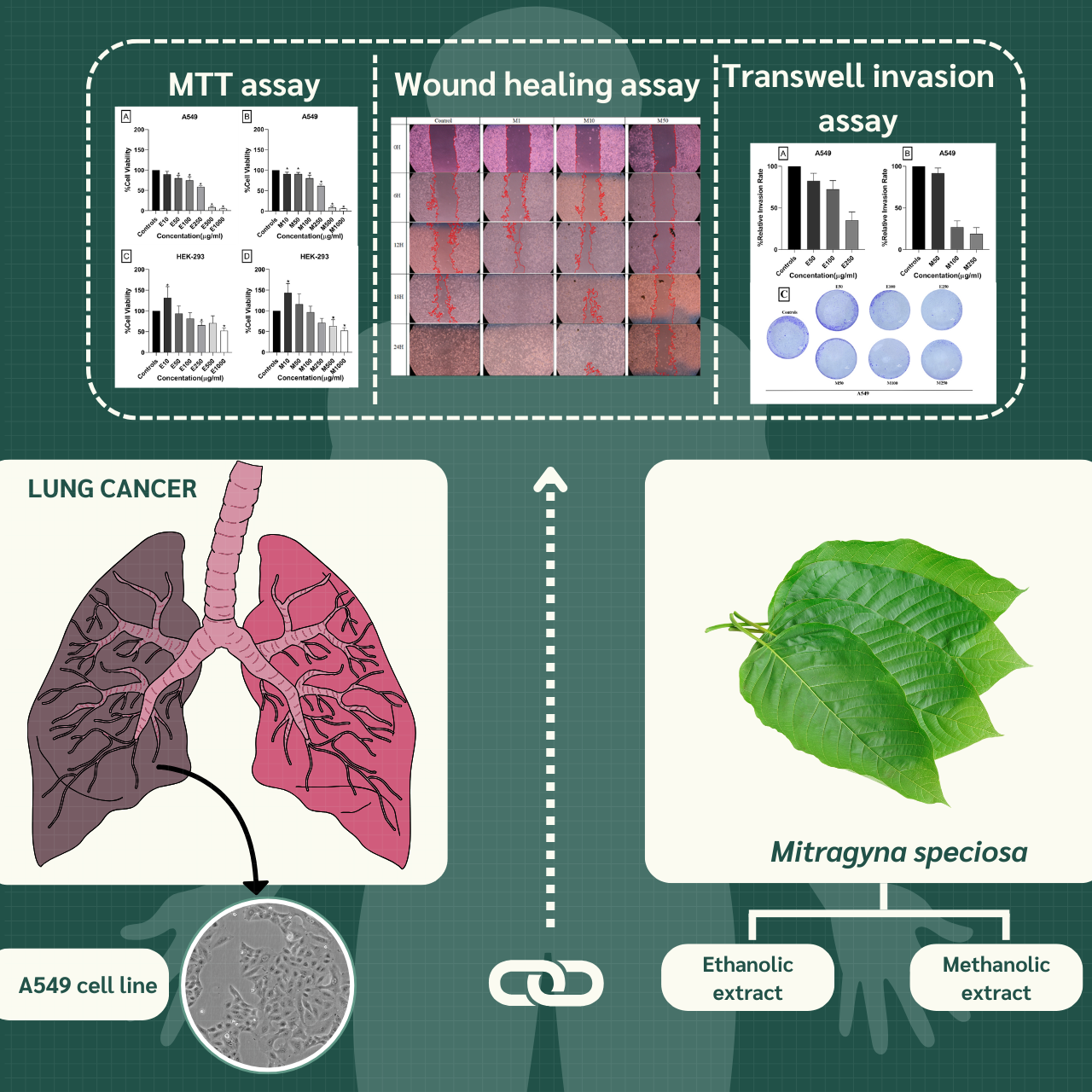
Downloads
References
J Rojanamatin, W Ukranun, P Supaattagorn, I Chiawiriyabunya, M Wongsena, A Chaiwerawattana, P Laowahutanont, I Chitapanarux, P Vatanasapt, S L Greater, S Sangrajrang and R Buasom. Cancer in Thailand Vol. X, 2016–2018. National Cancer Institute, Bangkok, Thailand, 2021.
W Imsamran, A Chaiwerawattana, S Wiangnon, D Pongnikorn, K Suwanrungrung, S Sangrajrang and R Buasom. Cancer in Thailand Vol. VIII, 2010-2012. National Cancer Institute, Bangkok, Thailand, 2015.
C Holohan, SV Schaeybroeck, DB Longley and PG Johnston. Cancer drug resistance: An evolving paradigm. Nature Reviews Cancer 2013; 13(10), 714-726.
R Sullivan, J Peppercorn, K Sikora, J Zalcberg, NJ Meropol, E Amir, D Khayat, P Boyle, P Autier, IF Tannock, T Fojo, J Siderov, S Williamson, S Camporesi, JG McVie, AD Purushotham, P Naredi, A Eggermont, MF Brennan, ML Steinberg, …, M Aapro. Delivering affordable cancer care in high-income countries. The Lancet Oncology 2011; 12(10), 933-980.
A Montazeri. Quality of life data as prognostic indicators of survival in cancer patients: An overview of the literature from 1982 to 2008. Health and Quality of Life Outcomes 2009; 7, 102.
J Poofery, P Khaw-on, S Subhawa, B Sripanidkulchai, A Tanstrawarasin, S Saetang, S Siwachat, N Lertprasertsuke, R Banjerdpongchai. Potential of Thai Herbal Extracts on Lung Cancer Treatment by Inducing Apoptosis and Synergizing Chemotherapy. Molecules. 2020; 25(1):231.
K Srichana, B Janchawee, S Prutipanlai, P Raungrut and N Keawpradub. Effects of Mitragynine and a Crude Alkaloid Extract Derived from Mitragyna speciosa Korth. on Permethrin Elimination in Rats. Pharmaceutics. 2015; 7(2):10-26.
G Domnic, NJY Chear, SFA Rahman, S Ramanathan, KW Lo, D Singh and N Mohana-Kumaran. Combinations of indole-based alkaloids from Mitragyna speciosa (Kratom) and cisplatin inhibit cell proliferation and migration of nasopharyngeal carcinoma cell lines. Journal of Ethnopharmacology 2021; 279, 114391.
SH Goh, TK Yian, MN Mordi and SM Mansor. Mitragynine exhibits selective cytotoxicity and apoptosis induction in cancer cells. Journal of Ethnopharmacology 2014; 153(1), 50-56.
PA Priatna, IM Puspitasari and R Abdulah. Cytotoxic potential of Mitragyna speciosa as anticancer - A review. Pharmacognosy Journal 2024; 16(6), 1418-1423.
NA Saidin, H Takayama, E Holmes and NJ Gooderham. Cytotoxicity of mitragynine and analogues on human cancer cells. Phytotherapy Research 2022; 36(1), 508-518.
LG Rodriguez, X Wu and JL Guan. Wound-healing assay. Methods in Molecular Biology 2005; 294, 23-29.
CR Justus, MA Marie, EJ Sanderlin and LV Yang. Transwell in vitro cell migration and invasion assays. Methods in Molecular Biology 2023; 2644, 349-359.
A Bayu, SI Rahmawati, F Karim, JA Panggabean, DP Nuswantari, DW Indriani, P Ahmadi, R Witular, NLPI Dharmayanti and MY Putra. An in vitro examination of whether kratom extracts enhance the cytotoxicity of low-dose doxorubicin against A549 human lung cancer cells. Molecules 2024; 29(6), 1404.
K Johnson, K Matsumoto, T Mori and A Saitoh. Comprehensive toxicity studies of Mitragyna speciosa extracts: Implications for therapeutic use. Toxicology Letters 2025; 355, 128-140.
K Viwatpinyo, S Mukda and S Warinhomhoun. Effects of mitragynine on viability, proliferation, and migration of C6 rat glioma, SH-SY5Y human neuroblastoma, and HT22 immortalized mouse hippocampal neuron cell lines. Biomedicine and Pharmacotherapy 2023; 166, 115364.
R Sudmoon, T Tanee, V Wongpanich, K Saenphet and S Saenphet. Comparative analysis of Mitragyna species: Implications for anticancer potential. Phytochemistry 2025; 190, 113645.
F Zakaria, NNM Anuar, NSN Hisam, JK Tan, F Zakaria, SMM Fauzi, MBA Rahman and SE Ashari. An investigation of the in vitro wound healing potential of Mitragyna speciosa (Korth.) Havil leaf ultrasound-assisted methanol crude extract and fractions. Biocatalysis and Agricultural Biotechnology 2023; 50, 102707.
SY Kim, MS Shin, GJ Kim, H Kwon, MJ Lee, AR Han, JW Nam, CH Jung, KS Kang and H Choi. Inhibition of A549 lung cancer cell migration and invasion by ent-caprolactin C via the suppression of transforming growth factor-β-induced epithelial-mesenchymal transition. Marine Drugs 2021; 19(8), 465.
Y Ran, L Yang, X Jia, H Zhao, Q Hu, B Yang, D Tang and M Tian. Phytochemical composition and anticancer effect of Akebia trifoliata seed in non-small cell lung cancer A549 cells. Arabian Journal of Chemistry 2024; 17(12), 106020.
N Yang, X Jia, Y Yang, J Niu, X Wu, F Ding, M Tian and D Tang. Premna puberula root petroleum ether extract inhibits proliferation, migration, and invasion, and induces apoptosis through mitochondrial pathway in non-small cell lung cancer A549 cells. Arabian Journal of Chemistry 2023; 17, 105409.
P Friedl and S Alexander. Cancer invasion and the microenvironment: Plasticity and reciprocity. Cell 2011; 147(5), 992-1009.
J Xu, S Lamouille and R Derynck. TGF-β-induced epithelial to mesenchymal transition. Cell Research 2020; 19, 156-172.
J Brábek, CT Mierke, D Rösel, P Veselý and B Fabry. The role of the tissue microenvironment in the regulation of cancer cell motility and invasion. Cell Communication and Signaling 2010; 8, 22.
S Jiang, Y Gao, W Hou, R Liu, X Qi, X Xu, J Li, Y Bao, H Zheng and B Hua. Sinomenine inhibits A549 human lung cancer cell invasion by mediating the STAT3 signaling pathway. Oncology Letters 2016; 12(2), 1380-1386.
X Zhang, J Wang, Q Wang, C Xiong, L Zhao, Y Gu and Y Zhang. Knockdown of HOXA5 inhibits the proliferation and invasion of A549 cells through the inhibition of the Wnt/β-catenin signaling pathway. Cell Cycle 2016; 15(8), 1075-1082.
T Akimoto, N Mitsuhashi, Y Saito, T Ebara and H Niibe. Effect of radiation on the expression of E-cadherin and α-catenin and invasive capacity in human lung cancer cell line in vitro. International of Journal of Radiation Oncology, Biology, Physics 1998; 41(5), 1171-1176.
C Chuang, W Yang, J Lin, C Hsieh, T Pan and Y Lin. Hyperoside inhibits migration and invasion of human non-small cell lung cancer A549 cells via regulation of the FAK-ERK signaling pathway. Journal of Nutritional Biochemistry 2016; 33, 118-126.
H He, L Zheng, YP Sun, GW Zhang and ZG Yue. Steroidal saponins from Paris polyphylla suppress malignancy of human lung cancer A549 cells. Asian Pacific Journal of Cancer Prevention 2014; 15(24), 10911-10916.
NS Holden, MC Catley, LM Cambridge, PJ Barnes and R Newton. ICAM-1 expression is highly NF-κB-dependent in A549 cells. European Journal of Biochemistry 2004; 271(4), 785-791.
P Srikoon, R Kariya, E Kudo, H Goto, K Vaeteewoottacharn, M Taura, S Wongkham and S Okada. Diethyldithiocarbamate suppresses an NF-κB dependent metastatic pathway in cholangiocarcinoma cells. Asian Pacific Journal of Cancer Prevention 2013; 14(7), 4441-4446.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






