Protein-Bound Polysaccharide K (PSK)-Rich Extract of Lentinus Polychrous Exhibits Anticancer Activity by Apoptosis Induction and Cell Cycle Arrest in Cholangiocarcinoma Cells
DOI:
https://doi.org/10.48048/tis.2025.8806Keywords:
Cholangiocarcinoma, Lentinus polychrous, Anticancer, Apoptosis, Cell cycle, Protein-bound polysaccharide K (PSK), AntioxidantAbstract
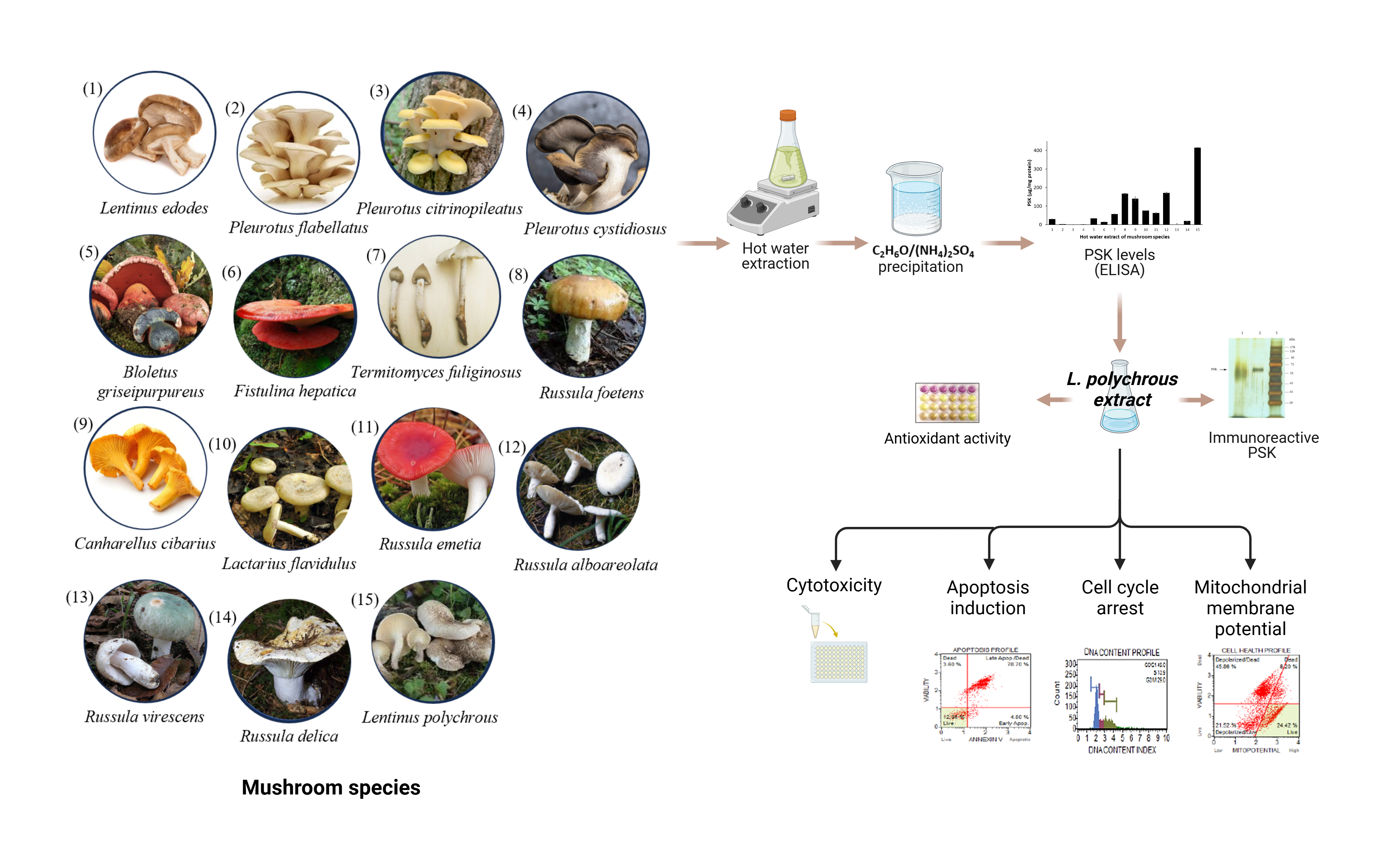
Cholangiocarcinoma (CCA) is an uncommon adenocarcinoma of the bile duct epithelial. There is no standard regimen yet for patients with adequate performance status. Therefore, effective agents are necessary. Recently, protein-bound polysaccharide K (PSK) has shown a great anticancer potential. L. polychrous is a Thai local edible mushroom that traditionally used as a folk medicine for treatments of fever and inflammation. This study aimed to identify the PSK from hot water extract of L. polychrous and examine the anticancer activity against a CCA cell line KKU-213A. The PSK were quantified by ELISA and characterized using SDS-PAGE, glycoprotein staining kit, and western blot analysis. The antioxidant activity was assessed by DPPH assay. Cytotoxic effect against KKU-213A was assessed by MTT assay. Apoptosis and cell cycle were analyzed by flow cytometry. The highest PSK levels were found in L. polychrous extract compared with the other 14 mushroom species which purified PSK is approximately 70 kDa of glycoprotein. The hot water extract of L. polychrous showed strong antioxidant activity. The cell viability of KKU-213A cells was significantly inhibited with L. polychrous extract at an IC50 value of 3.31 ± 0.09 mg/mL. In addition, the induction of cell cycle arrest at S and G2/M phases was observed together with induction of apoptosis, and mitochondrial membrane dysfunction in concentration-dependent manner. Hence the L. polychrous extract is the great source of PSK with strong antioxidant activity and exhibits anti-CCA effects through cell cycle arrest and apoptotic induction.
HIGHLIGHTS
- Lentinus polychrous, a Thai local edible mushroom, is the good source of PSK and powerful antioxidant activity.
- The hot water extract of L. polychrous suppresses cell viability, induces apoptosis, cell cycle arrest, and mitochondrial dysfunction in CCA cell line.
- The anticancer effects of L. polychrous extract against CCA cells makes it a potential agent for CCA treatment.
GRAPHICAL ABSTRACT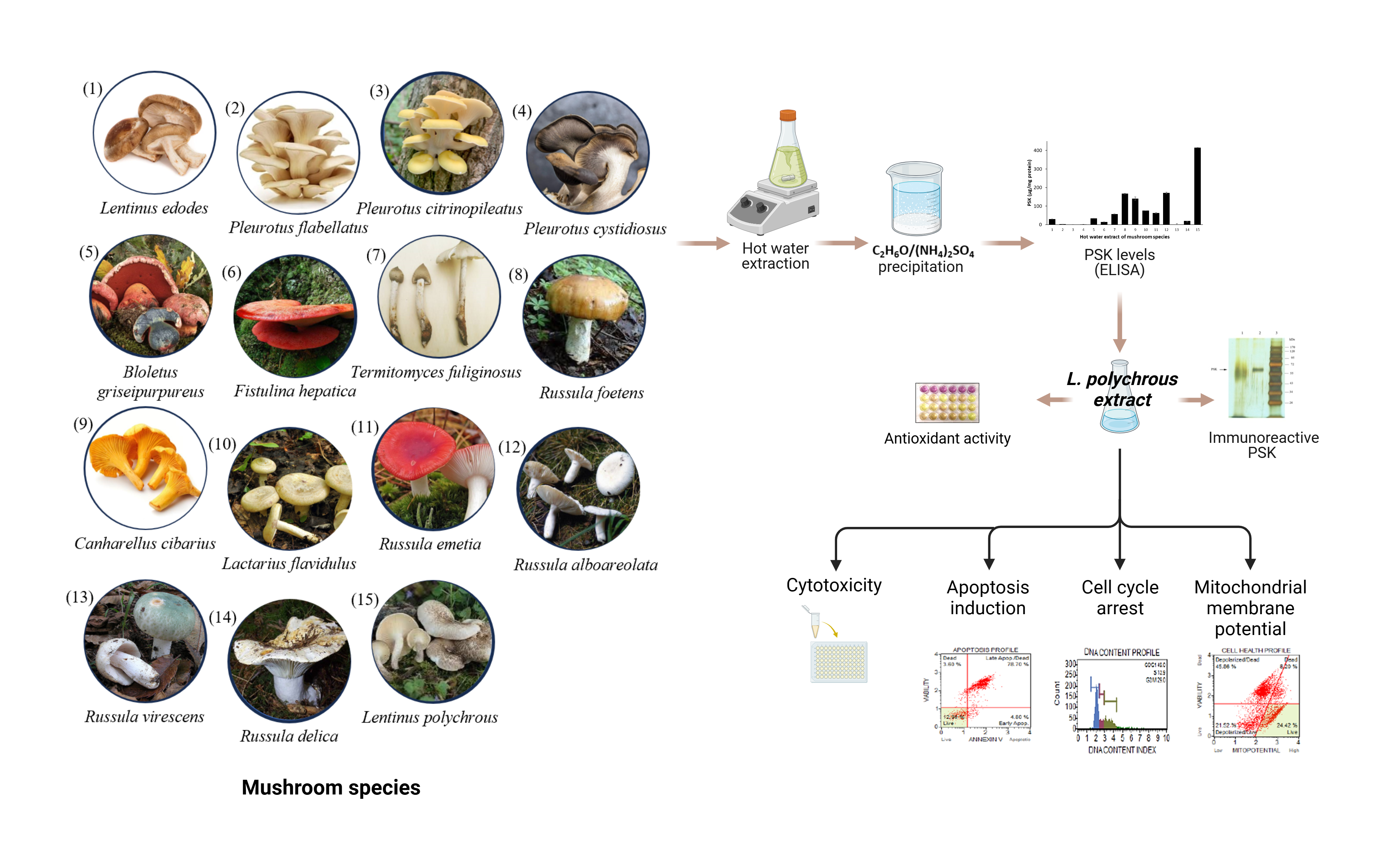
Downloads
References
B Sripa, JM Bethony, P Sithithaworn, S Kaewkes, E Mairiang, A Loukas, J Mulvenna, T Laha, PJ Hotez and PJ Brindley. Opisthoriasis and Opisthorchis-associated cholangiocarcinoma in Thailand and Laos. Acta Trop. 2011; 120, S158-S168.
S Sriamporn, P Pisani, V Pipitgool, K Suwanrungruang, S Kamsa-Ard and DM Parkin. Prevalence of Opisthorchis viverrini infection and incidence of cholangiocarcinoma in Khon Kaen, Northeast Thailand. Trop. Med. Int. Health 2004; 9, 588-94.
AE Sirica. Cholangiocarcinoma: Molecular targeting strategies for chemoprevention and therapy. Hepatology 2005; 41, 5-15.
M Hejna, M Pruckmayer and M Raderer. The role of chemotherapy and radiation in the management of biliary cancer: A review of the literature. Eur. J. Cancer 1998; 34, 977-86.
T Sasaki, T Takeda, T Okamoto, M Ozaka and N Sasahira. Chemotherapy for biliary tract cancer in 2021. J. Clin. Med. 2021; 10, 3108.
R Zhou, ZK Liu, YN Zhang, JH Wong, TB Ng and F Liu. Research progress of bioactive proteins from the edible and medicinal mushrooms. Curr. Protein Pept. Sci. 2019; 20, 196-219.
S Tsukagoshi, Y Hashimoto, G Fujii, H Kobayashi, K Nomoto and K Orita. Krestin (PSK). Cancer Treat. Rev. 1984; 11, 131-55.
H Nakazato, A Koike, S Saji, N Ogawa and J Sakamoto. Efficacy of immunochemotherapy as adjuvant treatment after curative resection of gastric cancer. Lancet 1994; 343, 1122-6.
TS Hattori, N Komatsu, S Shichijo and K Toh. Protein-bound polysaccharide K induced apoptosis of the human Burkitt lymphoma cell line, Namalwa. Biomed. Pharmacother. 2004; 58, 226-30.
E Jiménez-Medina, E Berruguilla, I Romero, I Algarra, A Collado, F Garrido and A Garcia-Loracorresponding. The immunomodulator PSK induces in vitro cytotoxic activity in tumor cell lines via arrest of cell cycle and induction of apoptosis. BMC Cancer 2008; 8, 78.
N Hirahara, M Fujioka, T Edamatsu, A Fujieda, F Sekine, T Wada and T Tanaka. Protein-bound polysaccharide-K (PSK) induces apoptosis and inhibits proliferation of promyelocytic leukemia HL-60 cells. Anticancer Res. 2011; 31, 2733-8.
Y Maehara, S Tsujitani, H Saeki, E Oki, K Yoshinaga, Y Emi, M Morita, S Kohnoe, Y Kakeji, T Yano and H Baba. Biological mechanism and clinical effect of protein-bound polysaccharide K (KRESTIN(®): Review of development and future perspectives. Surg. Today 2012; 42, 8-28.
J Sakamoto, S Morita, K Oba, T Matsui, M Kobayashi, H Nakazato and Y Ohashi. Efficacy of adjuvant immunochemotherapy with polysaccharide K for patients with curatively resected colorectal cancer: A meta-analysis of centrally randomized controlled clinical trials. Cancer Immunol. Immunother. 2006; 55, 404-11.
K Oba, S Teramukai, M Kobayashi, T Matsui, Y Kodera and J Sakamoto. Efficacy of adjuvant immunochemotherapy with polysaccharide K for patients with curative resections of gastric cancer. Cancer Immunol. Immunother. 2007; 56, 905-11.
H Hoshi, H Saito, H Iijima, M Uchida, T Wada, G Ito, H Tanaka, T Sawada and K Hirakawa. Anti-protein-bound polysaccharide-K monoclonal antibody binds the active structure and neutralizes direct antitumor action of the compound. Oncol. Rep. 2011; 25, 905-13.
U Klinhom and W Klinhom. Traditional mushrooms. Thai Health Foundation, Bangkok, Thailand, 2006.
N Fangkrathok, J Junlatat and B Sripanidkulchai. In vivo and in vitro anti-inflammatory activity of Lentinus polychrous extract. J. Ethnopharmacol. 2013; 147, 631-7.
N Fangkrathok, J Junlatat, K Umehara, H Noguchi and B Sripanidkulchai. Cytotoxic and immunomodulatory effects of polyhydroxyoctane isolated from Lentinus polychrous mycelia. J. Nat. Med. 2014; 68, 302-9.
C Thetsrimuang, S Khammuang and R Sarnthima. Antioxidant activity of crude polysaccharides from edible fresh and dry mushroom fruiting bodies of Lentinus sp. Strain RJ-2. Int. J. Pharmacol. 2011; 7, 58-65.
N Armassa, O Poungchompu, S Rayan, S Leethong, N Weerapreeyakul and S Machana. Antioxidant activity and cytotoxicity in breast cancer cells line of mushrooms extracts; Lentinus polychrous Lev. compared to Ganoderma lucidum (Fr.) Karst. Isan J. Pharmaceut. Sci. 2009; 5, 243-50.
C Thetsrimuang, S Khammuang, K Chiablaem, C Srisomsap and R Sarnthima. Antioxidant properties and cytotoxicity of crude polysaccharides from Lentinus polychrous Lév. Food Chem. 2011; 128, 634-9.
N Fangkrathok. Chemical constituents and pharmacological activities of Lentinus polychrous Lév. Isan J. Pharmaceut. Sci. 2018; 15, 49-57.
J Attarat and T Phermthai. Bioactive compounds in three edible Lentinus mushrooms. Walailak J. Sci. Tech. 2015; 12, 491-504.
JH Choi, YB Kim, HY Lim, JS Park, HC Kim, YK Cho, SW Han, MW Kim and HJ Joo. 5-fluorouracil, mitomycin-C, and polysaccharide-K adjuvant chemoimmunotherapy for locally advanced gastric cancer: The prognostic significance of frequent perineural invasion. Hepato Gastroenterology 2007; 54, 290-7.
R Katoh and M Ooshiro. Enhancement of antitumor effect of tegafur/uracil (UFT) plus leucovorin by combined treatment with protein-bound polysaccharide, PSK, in mouse models. Cell. Mol. Immunol. 2007; 4, 295-9.
C Mekjaruskul, N Fangkrathok and B Sripanidkulchai. Acute toxicity investigation of polysaccharide extracts of Lentinus polychrous in rats. Songklanakarin J. Sci. Tech. 2015; 34, 433-9.
AH Rosendahl, C Sun, D Wu and R Andersson. Polysaccharide-K (PSK) increases p21(WAF/Cip1) and promotes apoptosis in pancreatic cancer cells. Pancreatology 2012; 12, 467-74.
C Chen and ANT Kong. Dietary cancer-chemopreventive compounds: From signaling and gene expression to pharmacological effects. Trends Pharmacol. Sci. 2005; 26, 318-26.
BB Aggarwal, Y Takada and OV Oommen. From chemoprevention to chemotherapy: Common targets and common goals. Expert Opin. Invest. Drugs 2004; 13, 1327-38.
HK Matthews, C Bertoli and RAMD Bruin. Cell cycle control in cancer. Nat. Rev. Mol. Cell Biol. 2022; 23, 74-88.
MO Hengartner. The biochemistry of apoptosis. Nature 2000; 407, 770-6.
V Gogvadze, S Orrenius and B Zhivotovsky. Mitochondria in cancer cells: What is so special about them? Trends Cell. Biol. 2008; 18, 165-73.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






