Evaluation of Tyrosinase Inhibitory and Antioxidant Activity of Bis(carboxymethoxy) Curcumin Derivatives
DOI:
https://doi.org/10.48048/tis.2025.10745Keywords:
Bis(carboxymethoxy) curcumin derivatives, Tyrosinase inhibitor, Antioxidant, Molecular docking, Kinetic study, MelaninAbstract
Hyperpigmentation disorders are associated with various psychological health concerns and increased risk of skin cancer. Among potential therapeutic approaches, tyrosinase inhibitors represent promising candidates for treating these concerns. This study investigated the tyrosinase inhibitory activity and mechanisms of bis(carboxymethoxy) bismethoxycurcumin (4x), bis(carboxymethoxy) demethoxycurcumin (5v), and bis(carboxymethoxy) bisdemethoxycurcumin (6d). The results showed that 4x exhibited superior tyrosinase inhibitory activity across the tested concentrations (75 - 600 µM), decreasing enzyme activity to 78.52% - 36.72%, relative to 100% of the control, with an IC50 value of 272.85 µM. This compound also demonstrated the highest antioxidant activity, with values of 1.61 µM Trolox equivalent per µM 4x against the ABTS radical. The modification with a carboxymethoxy (-OCH2COOH) group enhanced the tyrosinase inhibitory and ABTS radical scavenging activities, while the absence of a methoxy group reduced these inhibitory activities. Molecular docking analysis indicated that these compounds interacted directly with the tyrosinase active site. Stronger interactions with copper atoms at the active site led to increased tyrosinase inhibitory activity, while the methoxy group proved significant for the tyrosinase inhibitory activity of 4x. Based on its superior inhibitory activity, 4x was selected for further kinetic and melanin inhibition studies, which revealed it to be a competitive inhibitor of tyrosinase with a Ki of 42.38 µM. Moreover, this compound, at concentrations of 75 - 150 µM, reduced melanin content in B16F10 cells to 88.02% - 70.57% compared to 100% of the control, without inducing cytotoxicity. These results suggest that 4x represents a promising candidate for development as an anti-hyperpigmentation agent in both cosmetic and medical applications.
HIGHLIGHTS
- Modification with the carboxymethoxy group significantly enhances the tyrosinase inhibitory activity of curcumin, with 4x exhibiting superior tyrosinase inhibitory and ABTS radical scavenging activities among the three modified curcuminoids, namely 4x, 5v, and 6d.
- The presence of the methoxy group is essential for the improved anti-tyrosinase and ABTS radical scavenging activities of 4x.
- 4x, functioning as a competitive inhibitor of tyrosinase and decreasing melanin production in B16F10 cells, emerges as a promising candidate for further research and development as an anti-hyperpigmentation agent.
GRAPHICAL ABSTRACT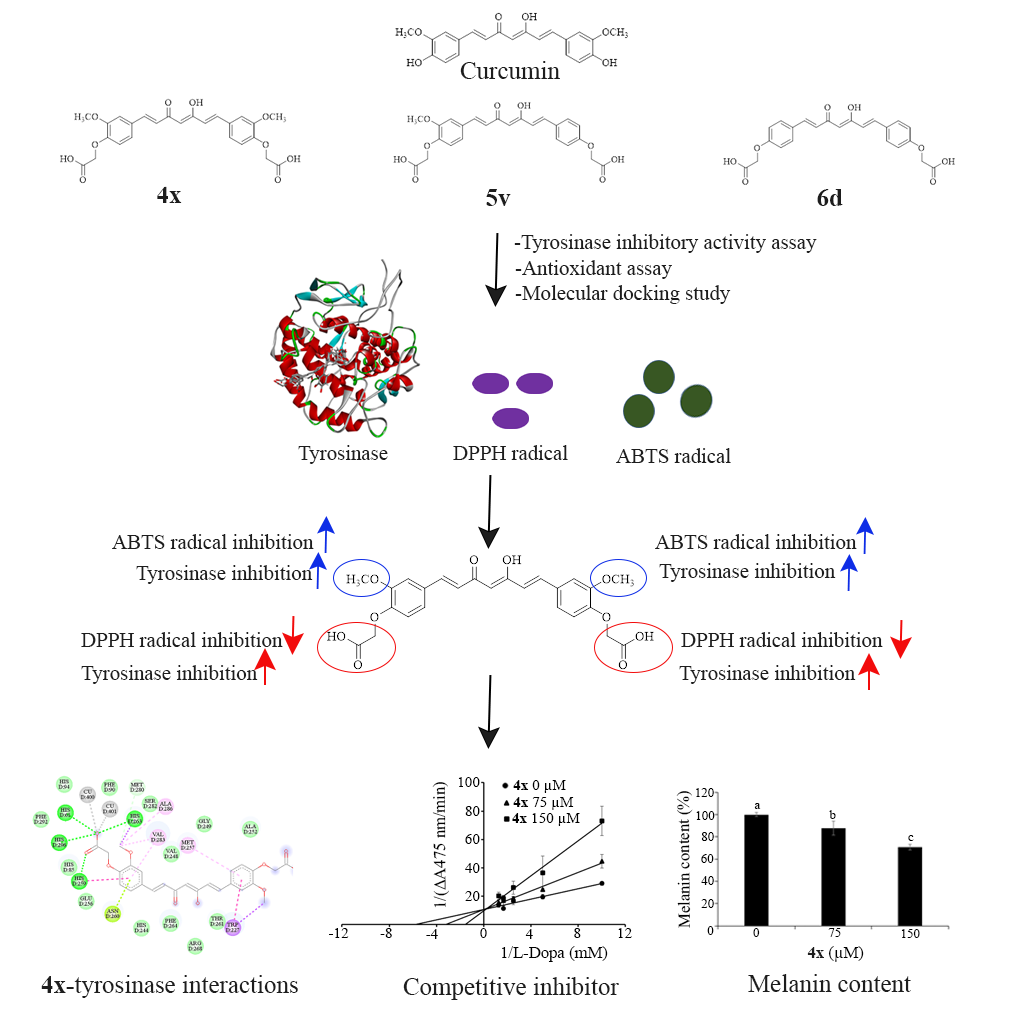
Downloads
References
M Brenner and VJ Hearing. The protective role of melanin against UV damage in human skin. Photochemistry and Photobiology 2008; 84(3), 539-549.
AM Thawabteh, A Jibreen, D Karaman, A Thawabteh and R Karaman. Skin pigmentation types, causes and treatment-a review. Molecules 2023; 28(12), 4839.
E Platsidaki, V Efstathiou, V Markantoni, A Kouris, G Kontochristopoulos, E Nikolaidou, D Rigopoulos, A Stratigos and S Gregoriou. Self-Esteem, depression, anxiety and quality of life in patients with melasma living in a sunny mediterranean area: Results from a prospective cross-sectional study. Dermatologic Therapy (Heidelb) 2023; 13(5), 1127-1136.
K Vogan. Pigmentation and skin-cancer risk. Nature Reviews Genetics 2008; 9(7), 502.
SAN D’Mello, GJ Finlay, BC Baguley and ME Askarian-Amiri. Signaling pathways in melanogenesis. International Journal of Molecular Sciences 2016; 17(7), 1144.
C Serre, V Busuttil and JM Botto. Intrinsic and extrinsic regulation of human skin melanogenesis and pigmentation. International Journal of Cosmetic Science 2018; 40(4), 328-347.
T Pillaiyar, M Manoj and V Namasivayam. Skin whitening agents: Medicinal chemistry perspective of tyrosinase inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry 2017; 32(1), 403-425.
J Chen, Y Liu, Z Zhao and J Qiu. Oxidative stress in the skin: Impact and related protection. International Journal of Cosmetic Science 2021; 43(5), 495-509.
MN Masum, K Yamauchi and T Mitsunaga. Tyrosinase inhibitors from natural and synthetic sources as skin-lightening agents. Reviews in Agricultural Science 2019; 7, 41-58.
N Oiso, M Tatebayashi, Y Hoshiyama and A Kawada. Allergic contact dermatitis caused by arbutin and dipotassium glycyrrhizate in skin-lightening products. Contact Dermatitis 2017; 77(1), 51-53.
YJ Kim and H Uyama. Tyrosinase inhibitors from natural and synthetic sources: Structure, inhibition mechanism and perspective for the future. Cellular and Molecular Life Sciences 2005; 62(15), 1707-1723.
A Amalraj, A Pius, S Gopi and S Gopi. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives - a review. Journal of Traditional and Complementary Medicine 2017; 7(2), 205-233.
A Athipornchai, N Niyomtham, W Pabuprapap, V Ajavakom, M Duca, S Azoulay and A Suksamrarn. Potent tyrosinase inhibitory activity of curcuminoid analogues and inhibition kinetics studies. Cosmetics 2021; 8(2), 35.
D Diki, W Widowati, S Tabassum, AF Sutendi, DN Triharsiwi and DS Hadiprasetyo. Curcumin as an antitumor agent: targeting cancer stem cell markers in glioblastoma through In Silico and In Vitro approaches. Trends in Sciences 2025; 22(5), 9485.
X Zhen, D Hao, J Pei, F Zhang, H Liu, J Wang, N Bian, X Zhang, Y Li and X Bu. The study of inhibitory effects and mechanism of carboxylate chitooligomer on melanin, prepared by laccase/TEMPO system. Carbohydrate Polymers 2019; 207, 391-397.
J Chen, J Yang, L Ma, J Li, N Shahzad and CK Kim. Structure-antioxidant activity relationship of methoxy, phenolic hydroxyl and carboxylic acid groups of phenolic acids. Scientific Reports 2020; 10, 2611.
LO Somsakeesit, T Senawong, P Kumboonma, S Saenglee, A Samankul, G Senawong, C Yenjai and C Phaosiri. Influence of side-chain changes on histone deacetylase inhibitory and cytotoxicity activities of curcuminoid derivatives. Bioorganic & Medicinal Chemistry Letters 2020; 30(11), 127171.
A Joompang, N Jangpromma, K Choowongkomon, W Payoungkiattikun, A Tankrathok, J Viyoch, K Luangpraditkun and S Klaynongsruang. Evaluation of tyrosinase inhibitory activity and mechanism of Leucrocin I and its modified peptides. Journal of Bioscience and Bioengineering 2020; 130(3), 239-246.
G Jones, P Willett, RC Glen, AR Leach and R Taylor. Development and validation of a genetic algorithm for flexible docking11Edited by F. E. Cohen. Journal of Molecular Biology 1997; 267(3), 727-748.
MZ Abdullah, JM Ali, Mitra Abolmaesoomi, PS Abdul-Rahman and OH Hashim. Anti-proliferative, in vitro antioxidant and cellular antioxidant activities of the leaf extracts from Polygonum minus Huds: Effects of solvent polarity. International Journal of Food Properties 2017; 20(s1), 846-862.
S Phosri, P Mahakunakorn, J Lueangsakulthai, N Jangpromma, P Swatsitang, S Daduang, A Dhiravisit and S Thammasirirak. An investigation of antioxidant and anti-inflammatory activities from blood components of crocodile (Crocodylus siamensis). The Protein Journal 2014; 33(5), 484-492.
A Joompang, P Anwised, S Klaynongsruang, L Taemaitree, A Wanthong, K Choowongkomon, S Daduang, S Katekaew and N Jangpromma. Rational design of an N-terminal cysteine-containing tetrapeptide that inhibits tyrosinase and evaluation of its mechanism of action. Current Research in Food Science 2023; 7, 100598.
M Hassan, S Shahzadi and A Kloczkowski. Tyrosinase inhibitors naturally present in plants and synthetic modifications of these natural products as anti-melanogenic agents: A review. Molecules 2023; 28(1), 378.
J Kuzminska, P Szyk, DT Mlynarczyk, P Bakun, I Muszalska-Kolos, K Dettlaff, A Sobczak, T Goslinski and A Jelinska. Curcumin derivatives in medicinal chemistry: Potential applications in cancer treatment. Molecules 2024; 29(22), 5321.
D Nokinsee, L Shank, VS Lee and P Nimmanpipug. Estimation of inhibitory effect against tyrosinase activity through homology modeling and molecular docking. Enzyme Research 2015; 2015, 262364.
A Mermer and S Demirci. Recent advances in triazoles as tyrosinase inhibitors. European Journal of Medicinal Chemistry 2023; 259, 115655.
W Wang, Y Gao, W Wang, J Zhang, J Yin, T Le, J Xue, UH Engelhardt and H Jiang. Kojic acid showed consistent inhibitory activity on tyrosinase from mushroom and in cultured B16F10 Cells compared with arbutins. Antioxidants (Basel) 2022; 11(3). 502.
P Saentao, FT Schevenels, J Yahuafai, A Joompang, T Suebrasri, S Boonlue, S Tontapha and R Lekphrom. Curculigosides J–K and curcorchidihydrobenzofuran A, a dihydrobenzofuran with anti-proliferative properties from Curculigo orchioides. Natural Product Research 2024. https://doi.org/10.1080/14786419.2024.2426064
YP Chen, M Li, Z Liu, J Wu, F Chen and S Zhang. Inhibition of tyrosinase and melanogenesis by carboxylic acids: Mechanistic insights and safety evaluation. Molecules 2025; 30(7), 1642.
İ Gulcin and SH Alwasel. DPPH radical scavenging assay. Processes 2023; 11(8), 2248.
JY Feng and ZQ Liu. Phenolic and enolic hydroxyl groups in curcumin: Which plays the major role in scavenging radicals? Journal of Agricultural and Food Chemistry 2009; 57(22), 11041-11046.
İ Gulcin. Antioxidants: A comprehensive review. Archives of Toxicology 2025; 99(5), 1893-1997.
CY Lee, C Anamoah, J Semenya, KN Chapman, AN Knoll, HF Brinkman, JI Malone and A Sharma. Electronic (donating or withdrawing) effects of ortho-phenolic substituents in dendritic antioxidants. Tetrahedron Letters 2020; 61(11), 151607.
G Rocchitta, C Rozzo, M Pisano, D Fabbri, MA Dettori, P Ruzza, C Honisch, R Dallocchio, A Dessì, R Migheli, P Serra and G Delogu. Inhibitory effect of curcumin-inspired derivatives on tyrosinase activity and melanogenesis. Molecules 2022; 27(22), 7942.
MB Hay and SCB Myneni. Structural environments of carboxyl groups in natural organic molecules from terrestrial systems. Part 1: Infrared spectroscopy. Geochimica et Cosmochimica Acta 2007; 71(14), 3518-3532.
S Goenka. Comparative study of the effects of curcuminoids and tetrahydrocurcuminoids on melanogenesis: Role of the methoxy groups. Future Pharmacology 2024; 4(1), 256-278.
Y Jeong, S Hong, HJ Jung, S Ullah, Y Hwang, H Choi, J Ko, J Lee, P Chun, HY Chung and HR Moon. Identification of a novel class of anti-melanogenic compounds, (Z)-5-(Substituted benzylidene)-3-phenyl-2-thioxothiazolidin-4-one Derivatives and their reactive oxygen species scavenging activities. Antioxidants 2022; 11(5), 948.
L Pisano, M Turco and CT Supuran. Biomedical applications of tyrosinases and tyrosinase inhibitors. In: CT Supuran (Ed.). The Enzymes. 1st ed. Academic Press, New York, 2024, p. 261-280.
T Shojazadeh, L Zolghadr, S Gharaghani, S JafarKhani, F Molaabasi, H Piri and N Gheibi. New insights into the inhibitory effect of phenol carboxylic acid antioxidants on mushroom tyrosinase by molecular dynamic studies and experimental assessment. Journal of Biomolecular Structure and Dynamics 2023; 41(22), 13404-13414.
S Goenka, F Johnson and SR Simon. Novel Chemically Modified Curcumin (CMC) derivatives inhibit tyrosinase activity and melanin synthesis in B16F10 mouse melanoma cells. Biomolecules 2021; 11(5). 674.
T Mann, W Gerwat, J Batzer, K Eggers, C Scherner, H Wenck, F Stäb, VJ Hearing, KH Röhm and L Kolbe. Inhibition of human tyrosinase requires molecular motifs distinctively different from mushroom tyrosinase. Journal of Investigative Dermatology 2018; 138(7), 1601-1608.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






