Temperature-Dependent Variations in Antioxidant Activities and Phytochemical Profiles of Passiflora edulis Leaf Extracts
DOI:
https://doi.org/10.48048/tis.2025.9772Keywords:
Antioxidant activity, TPC, TFC, Saponin assay, Quercetin assayAbstract
Passiflora edulis leaves contain bioactive compounds and have beneficial and important biological activities. The focus of this research is to pinpoint the optimum water extraction temperature and methods for the greatest antioxidant activity and phytochemical content. Dry powder of P. edulis leaves was extracted using aqueous solvents with temperature variations i.e., maceration (TR), 40 °C (T40), 60 °C (T60), decoction (TD), and ultrasonic-assisted extraction (UAE). All extracts were compared for antioxidant activity (DPPH, FRAP, ABTS), compound content (total polyphenolic and flavonoid content), marker assay (saponin assay using Liebermann-Burchard reagent, and quercetin assay using TLC-densitometry), and determination of the fingerprint pattern of compounds through Fourier Transform Infrared (FTIR) spectra. The antioxidant capacity demonstrated that the extract activity was dose-dependent at all procedure temperatures. Our data showed that decoction (TD) enhanced the antioxidant capacity and also levels of water-soluble flavonoids, polyphenolic, and quercetin compounds. However, the best saponin content was obtained at T60. The FTIR spectra showed the same pattern of all extract functional groups. Therefore, the increasing extraction temperature is directly related to the increase of flavonoid and polyphenol content and their antioxidant activities.
HIGHLIGHTS
- Passiflora edulis leaf has many phytoconstituents that have different bioactivities.
- The extraction method and temperature will impact the quantity of phytoconstituents extracted.
- Decoction produced the highest antioxidant activity, while 60 °C gave the best saponin content.
- FTIR analysis showed similar spectral patterns in all extracts.
GRAPHICAL ABSTRACT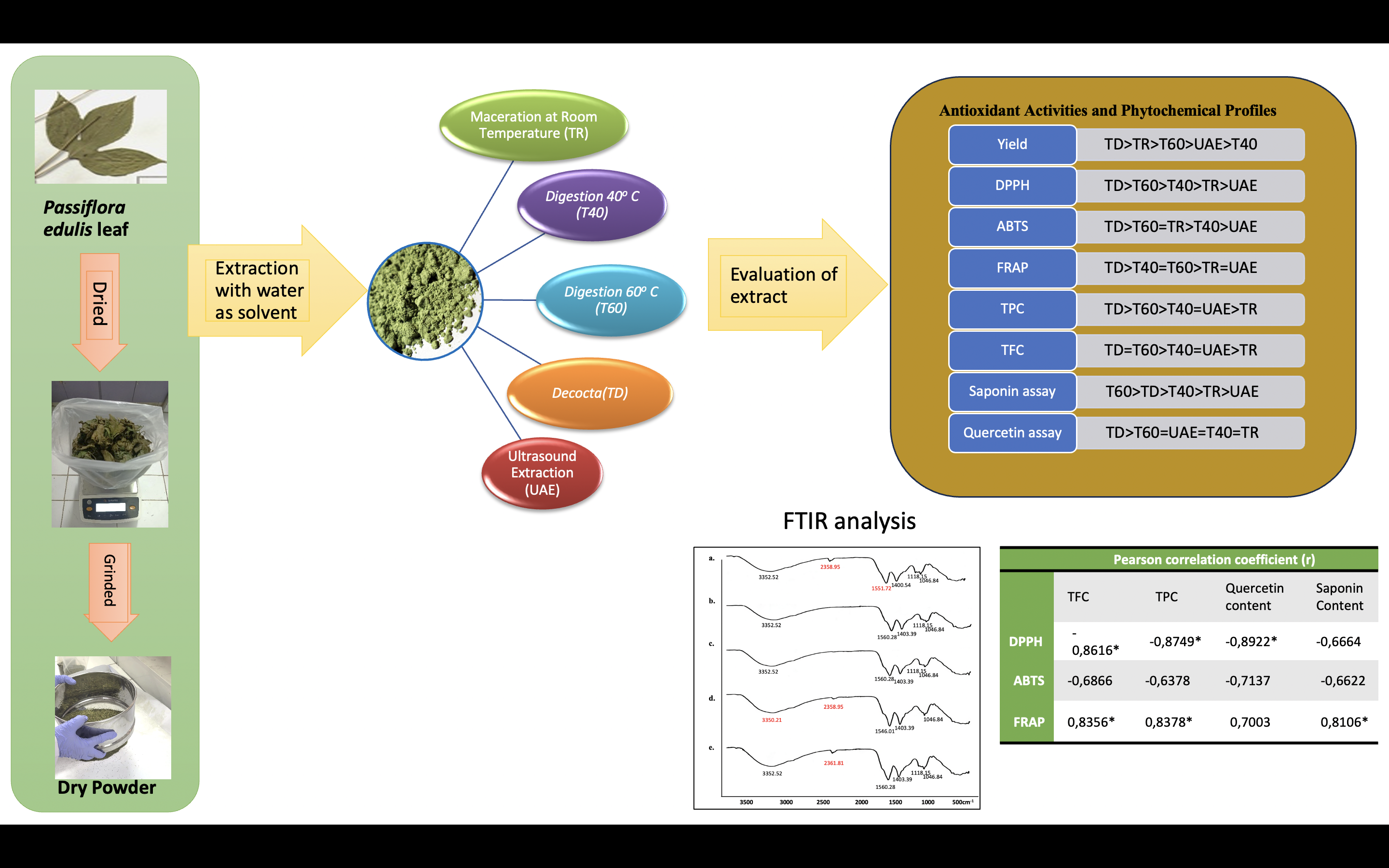
Downloads
References
F de-Paris, RD Petry, FH Reginatto, G Gosmann, J Quevedo, JB Salgueiro, F Kapczinski, GG Ortega and EP Schenkel. Pharmacochemical study of aqueous extracts of Passiflora alata Dryander and Passiflora edulis Sims. Acta Farmaceutica Bonaerense 2002; 21(1), 5-8.
N Urrego, P Sepúlveda, M Aragón, FA Ramos, GM Costa, LF Ospina and L Castellanos. Flavonoids and saponins from Passiflora edulis f. edulis leaves (purple passion fruit) and its potential anti-inflammatory activity. Journal of Pharmacy and Pharmacology 2021; 73(11), 1530-1538.
S Mohite, R Shah and N Patel. Antimicrobial activity of leaves extracts of Passiflora foetida. Asian Journal of Research in Pharmaceutical Science 2018; 8(1), 17.
KRV Bandara, C Padumadasa and DC Peiris. Potent antibacterial, antioxidant and toxic activities of extracts from Passiflora suberosa L. leaves. PeerJ 2018; 6, e4804.
F Fahleni, Y Desmiaty, RS Dewi, A Kholilah, MSN Izzah and NMD Sandhiutami. The development of a novel nanoherbal passionfruit (Passiflora edulis) leaves with safety and analgesic effect. Trends in Sciences 2024; 21(12), 8412.
V Manuel, JA Pérez and J Fernández. Chapter 36 - Passion fruit. In: AK Jaiswal (Ed.). Nutritional composition and antioxidant properties of fruits and vegetables. Academic Press, New York, 2020.
X He, F Luan, Y Yang, Z Wang, Z Zhao, J Fang, M Wang, M Zuo and Y Li. Passiflora edulis: An insight into current researches on phytochemistry and pharmacology. Frontiers in Pharmacology 2020; 11, 617.
F Fahleni, Y Desmiaty, RS Dewi, A Kholilah, MSN Izzah and NMD Sandhiutami. The development of a novel nanoherbal passionfruit (Passiflora edulis) leaves with safety and analgesic effect. Trends in Sciences 2024; 21(12), 8412.
K Yoshikawa, S Katsuta, J Mizumori and S Arihara. Four cycloartane triterpenoids and 6 related saponins from Passiflora edulis. Journal of Natural Products 2000; 63(9), 1229-1234.
G Bobo-García, G Davidov-Pardo, C Arroqui, P Vírseda, MR Marín-Arroyo and M Navarro. Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. Journal of the Science of Food and Agriculture 2015; 95(1), 204-209.
N M’hiri, I Ioannou, C Paris, M Ghoul and MN Boudhrioua. Comparison of the efficiency of different extraction methods on antioxidants of maltease orange peel. International Journal of Food and Nutritional Science 2016; 3(2), 1-13.
F Chemat, MA Vian, AS Fabiano-Tixier, M Nutrizio, AR Jambrak, PES Munekata, JM Lorenzo, FJ Barba, A Binello and G Cravotto. A review of sustainable and intensified techniques for extraction of food and natural products. Green Chemistry 2020; 22, 2325-2353.
YQ Ma, JC Chen, DH Liu and XQ Ye. Simultaneous extraction of phenolic compounds of citrus peel extracts: Effect of ultrasound. Ultrasonics Sonochemistry 2009; 16(1), 57-62.
ISC Sulaiman, M Basri, HRF Masoumi, WJ Chee, SE Ashari and M Ismail. Effects of temperature, time, and solvent ratio on the extraction of phenolic compounds and the anti-radical activity of Clinacanthus nutans Lindau leaves by response surface methodology. Chemistry Central Journal 2017; 11, 54.
G Bobo‐García, G Davidov‐Pardo, C Arroqui, P Vírseda, MR Marín‐Arroyo and M Navarro. Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. Journal of the Science of Food and Agriculture 2015; 95(1), 204-209.
MF Abu Bakar, NA Ismail, A Isha and ALM Ling. Phytochemical composition and biological activities of selected wild berries (Rubus moluccanus L., R. fraxinifolius Poir., and R. alpestris Blume). Evidence-Based Complementary and Alternative Medicine 2016; 2016(1), 2482930.
M Chatatikun and A Chiabchalard. Thai plants with high antioxidant levels, free radical scavenging activity, anti- tyrosinase and anti-collagenase activity. BMC Complementary and Alternative Medicine 2017; 17, 487.
MS Mora-Ocación, AC Morillo-Coronado and EH Manjarres-Hernández. Extraction and quantification of saponins in quinoa (Chenopodium quinoa Willd.) genotypes from Colombia. International Journal of Food Science 2022; 2022(1), 7287487.
A Kumar, D Kumar, S Kumar and R Shri. Comparative evaluation of quercetin content in 3 varieties of Allium cepa using TLC densitometry. International Journal of Advances in Pharmacy, Biology and Chemistry 2015; 4(3), 612-619.
SM Zucolotto, C Fagundes, FH Reginatto, FA Ramos, L Castellanos, C Duque and EP Schenkel. Analysis of C-glycosyl flavonoids from South American Passiflora species by HPLC-DAD and HPLC-MS. Phytochemical Analysis 2012; 23(3), 232-239.
NMD Sandhiutami, Y Desmiaty and MH Fakhran. Antinociceptive and anti-inflammatory effect of Passiflora edulis leaves extract in acetic acid-induced pain and carrageenan-induced paw oedema in vivo. Pharmacy Education 2024; 24(2), 78-85.
CH Geow, MC Tan, SP Yeap and NL Chin. A review on extraction techniques and its future applications in industry. European Journal of Lipid Science and Technology 2021; 123(4), 2000302.
F Lumuindong and CF Mamuaja. Interaction of temperature and extraction time on the rendement and some properties of lime pectin (Citrus Aurantifolia S). Journal of Agriculture 2022; 1(3), 103-109.
PK Mukherjee, N Maity, NK Nema and BK Sarkar. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011; 19(1), 64-73.
H Abramovič, B Grobin, N Poklar Ulrih and B Cigić. Relevance and standardization of in vitro antioxidant assays: ABTS, DPPH, and Folin-Ciocalteu. Journal of Chemistry 2018; 2018(1), 4608405.
ME Giordano, R Caricato and MG Lionetto. Concentration dependence of the antioxidant and prooxidant activity of trolox in hela cells: Involvement in the induction of apoptotic volume decrease. Antioxidants 2020; 9(11), 1058.
Y Desmiaty, NMD Sandhiutami, E Mulatsari, FA Maziyah, K Rahmadhani, HOZ Algifari and FA Jantuna. Antioxidant and anti-inflammatory activity through inhibition of NF-κB and sEH of some citrus peel and phytoconstituent characteristics. Saudi Pharmaceutical Journal 2024; 32(2), 101959.
RD Gregg, L Righetti, J Buchli and S Schaal. Constrained accelerations for controlled geometric reduction: Sagittal-plane decoupling for bipedal locomotion. In: Proceedings of the 10th IEEE-RAS International Conference on Humanoid Robots, Nashville, Tennessee. 2010.
IFF Benzie and M Devaki. The ferric reducing/antioxidant power (FRAP) assay for non-enzymatic antioxidant capacity: Concepts, procedures, limitations and applications. In: R Apak, E Capanoglu and F Shahidi (Eds.). Measurement of antioxidant activity and capacity: Recent trends and applications. Wiley Online Library, New Jersey, 2017, p. 77-106.
E Checkouri, F Reignier, C Robert-Da Silva and O Meilhac. Evaluation of polyphenol content and antioxidant capacity of aqueous extracts from 8 medicinal plants from Reunion Island: Protection against oxidative stress in red blood cells and preadipocytes. Antioxidants 2020; 9(10), 959.
D Serea, NN Condurache, I Aprodu, OE Constantin, GE Bahrim, N Stănciuc, S Stanciu and G Rapeanu. Thermal stability and inhibitory action of red grape skin phytochemicals against enzymes associated with metabolic syndrome. Journal of Antioxidants 2022; 11(1), 118.
SL Rodríguez De Luna, RE Ramírez-Garza and SO Serna Saldívar. Environmentally friendly methods for flavonoid extraction from plant material: Impact of their operating conditions on yield and antioxidant properties. Scientific World Journal 2020; 2020(1), 6792069.
LSSD Mesquita, JWCD Mesquita, RP Dutra, MCA Batista, S Malik and FM Mendonça. Extraction parameters affect flavonoids content and antioxidant activities in Passiflora edulis. Journal of Chemical and Pharmaceutical Research 2016; 8(10), 99-107.
A Antony and M Farid. Effect of temperatures on polyphenols during extraction. Applied Sciences 2022; 12(4), 2107.
JKD Silva, CBB Cazarin, TC Colomeu, ÂG Batista, LMM Meletti, JAR Paschoal, SB Júnior, MF Furlan, FGR Reyes, F Augusto, MRM Júnior and RDL Zollner. Antioxidant activity of aqueous extract of passion fruit (Passiflora edulis) leaves: In vitro and in vivo study. Food Research International 2013; 53(2), 882-890.
K Cheng, H Gao, RR Wang, Y Liu, YX Hou, XH Liu, K Liu and W Wang. Evaluation of extraction and degradation methods to obtain chickpeasaponin B1 from chickpea (Cicer arietinum L.). Molecules 2017; 22(2), 332.
M George. Qualitative & quantitative phytochemical analysis on the leaves & fruits of Passiflora foetida. International Journal of Pharmaceutical Science Invention 2017; 6, 26-30.
SD Ramaiya, JS Bujang and MH Zakaria. Assessment of total phenolic, antioxidant, and antibacterial activities of Passiflora species. The Scientific World Journal 2014; 2014(1), 167309.
AEBP Leal, APD Oliveira, RF Santos, JMD Soares, EM Lavor, MC Pontes, JT Lima, ADDC Santos, JC Tomaz, GGD Oliveira, FC Neto, NP Lopes, LA Rolim and JRGDS Almeida. Determination of phenolic compounds, in vitro antioxidant activity and characterization of secondary metabolites in different parts of Passiflora cincinnata by HPLC-DAD-MS/MS analysis. Natural Product Research 2020; 34(7), 995-1001.
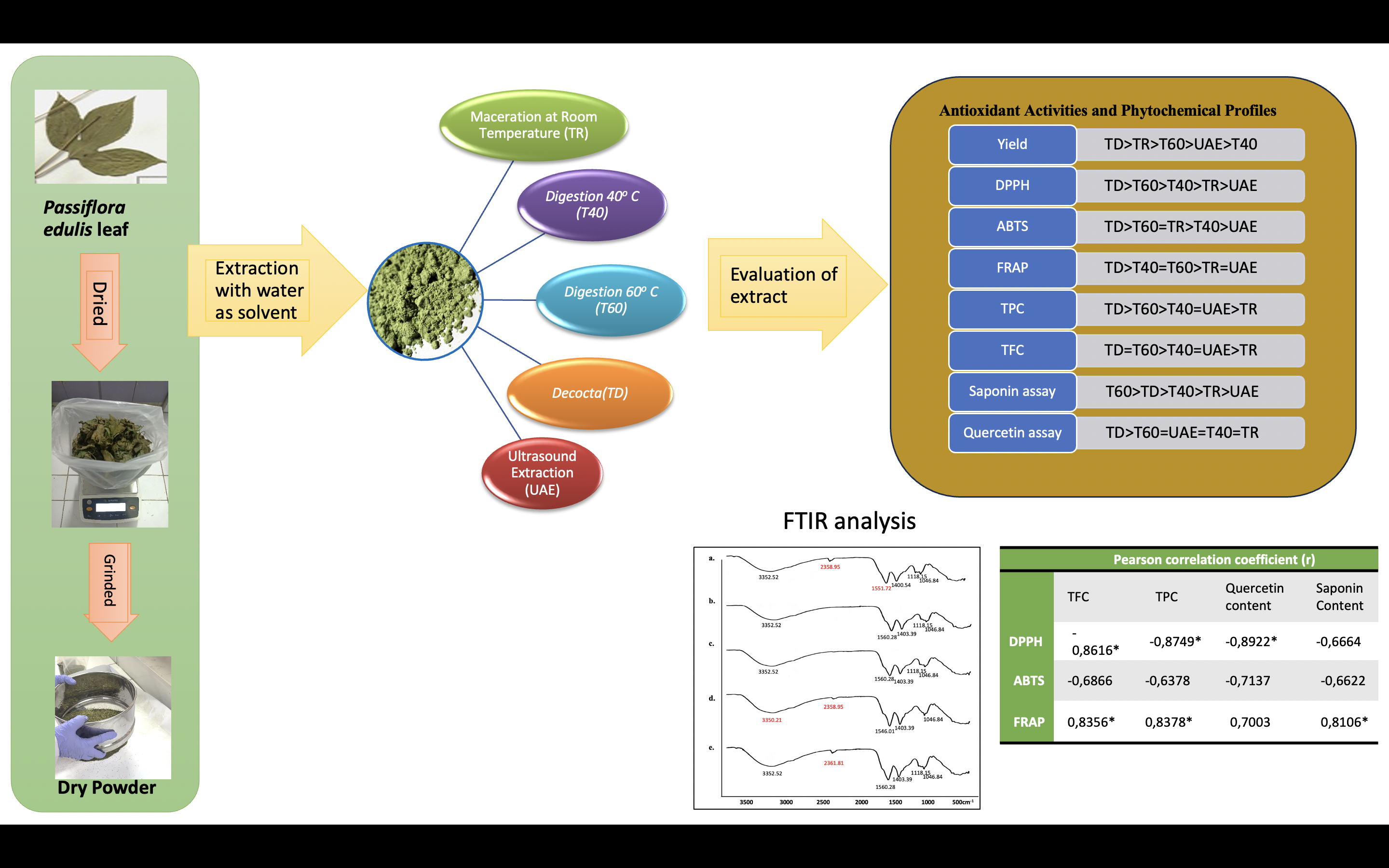
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






