The Development of a Novel Nanoherbal Passionfruit (Passiflora edulis) Leaves with Safety and Analgesic Effect
DOI:
https://doi.org/10.48048/tis.2024.8412Keywords:
Passionfruit leaves, Nanoherbal, Acute toxicity, Subchronic toxicity, AnalgesicAbstract
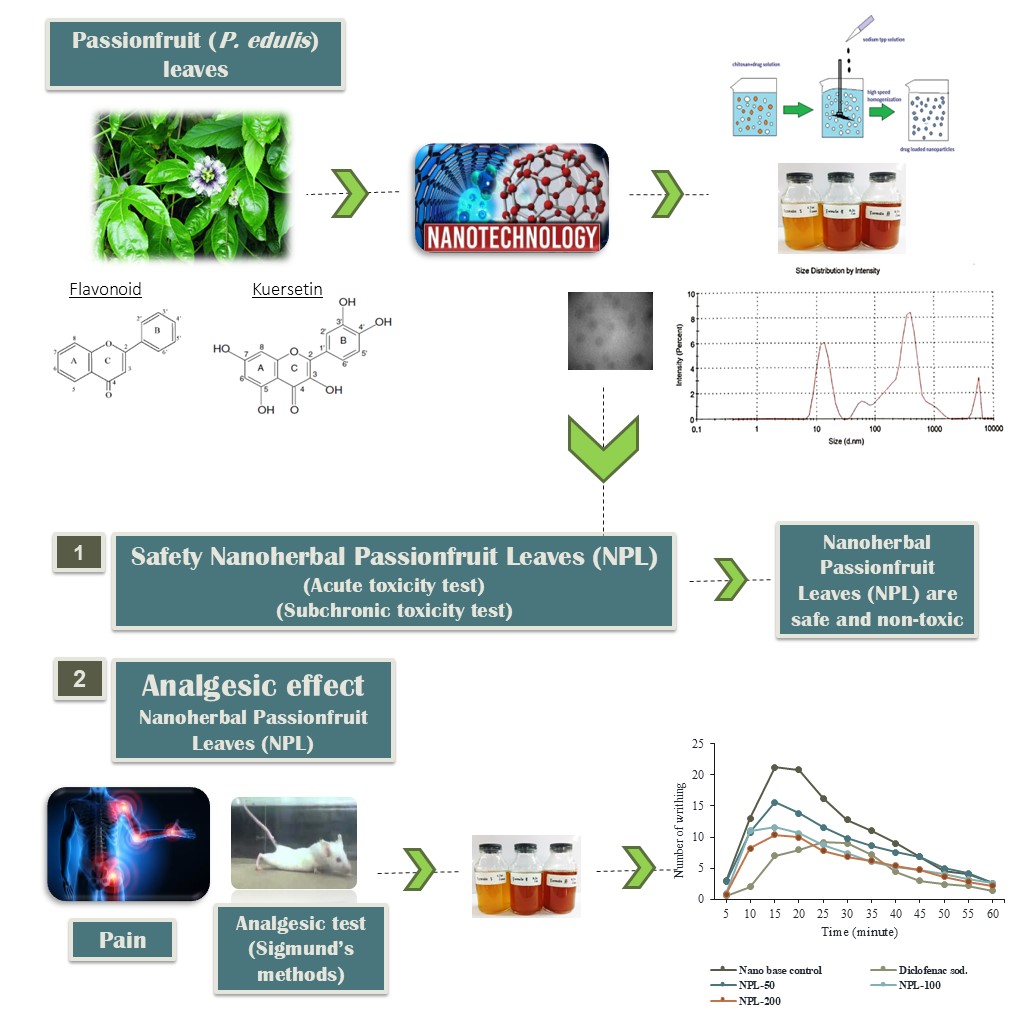
Passionfruit (Passiflora edulis) leaves are proven to contain flavonoids especially quercetin and showed a significant antinociceptive effect at acetic acid-induced wriggling. Modification of passionfruit leaf extract into nanoparticles can provide therapeutic effects precisely, quickly, and optimally. This study aimed to provide the method of preparation, and characterization of nanoherbal passionfruit leaves (NPL) and evaluate its safety profiles and analgesic effect. NPL was synthesized using ionic gelation methods, with 3 formula variations. Passionfruit leaf extract was dissolved using mixed solvents. The passionfruit leaves extract solution was suspended in Chitosan and dripped with sodium tripolyphosphate solution until nanoparticle formation. The characterization of the NPL study was evaluated by measuring the particle size, polydispersity index (PI), zeta potential, entrapment efficiency (EE), drug release, and transmission electron microscope (TEM) images. The safety profiles of NPL were then assessed in mice by acute and subchronic toxicity using OECD methods. The effectiveness of NPL as an analgesic was tested using an acetic acid test (visceral pain) in mice. We found the particle size of the NPL’s best formula (F2-NPL) obtained was 187.6 nm with polydispersity index 0.626 and zeta potential +31.2 mV. The entrapment efficiency of flavonoid content in NPL was 50.9 % and the flavonoid release for 3 h with 3 replications were 22.91, 27.88 and 25.27 % respectively. The morphology of the particles by TEM showed that the nanoparticles were circular shape. Safety evaluation in mice resulted in the LD50 of NPL being greater than 5000 mg/kg in the acute toxicity study and had no adverse effects on blood, liver, and renal profiles in the subchronic toxicity study. The NPL-200 group had better effectiveness as an analgesic (84.87 %) compared to NPL-50 (37.80 %) and NPL-100 (69.15 %). This finding indicates that the NPL is a safe and effective formula for enhancing analgesic effects. Thus, enables its applications for the treatment of various diseases related to analgesics and inflammation such as osteoarthritis.
HIGHLIGHTS
- The development of traditional medicine continues to be improved, to obtain the availability of safe, high quality, efficacious traditional medicines that are scientifically tested and can be widely utilized both for self-treatment by the community and in health services
- It is known that the most widely reported flavonoid derivative contained in edulis leaves is quercetin. Quercetin has poor bioavailability, solubility, and stability, reducing its therapeutic effect.
- By creating nanoparticles, adjustments can be made to natural products including quercetin in flavonoids in order to maximize their activity, boost their bioavailability, solubility, and capacity to be swiftly absorbed in the body to deliver the best possible therapeutic effects
- In developing nanoherbal passionfruit leaves using the ionic galation method, several solvents were used. It is known that no chemical substance can be said to be completely safe. It is important to study the toxicity profile further to obtain safe traditional medicines.
- Nanoparticle formulation of passion fruit leaf extract can provide better pain inhibition effectiveness in mice
GRAPHICAL ABSTRACT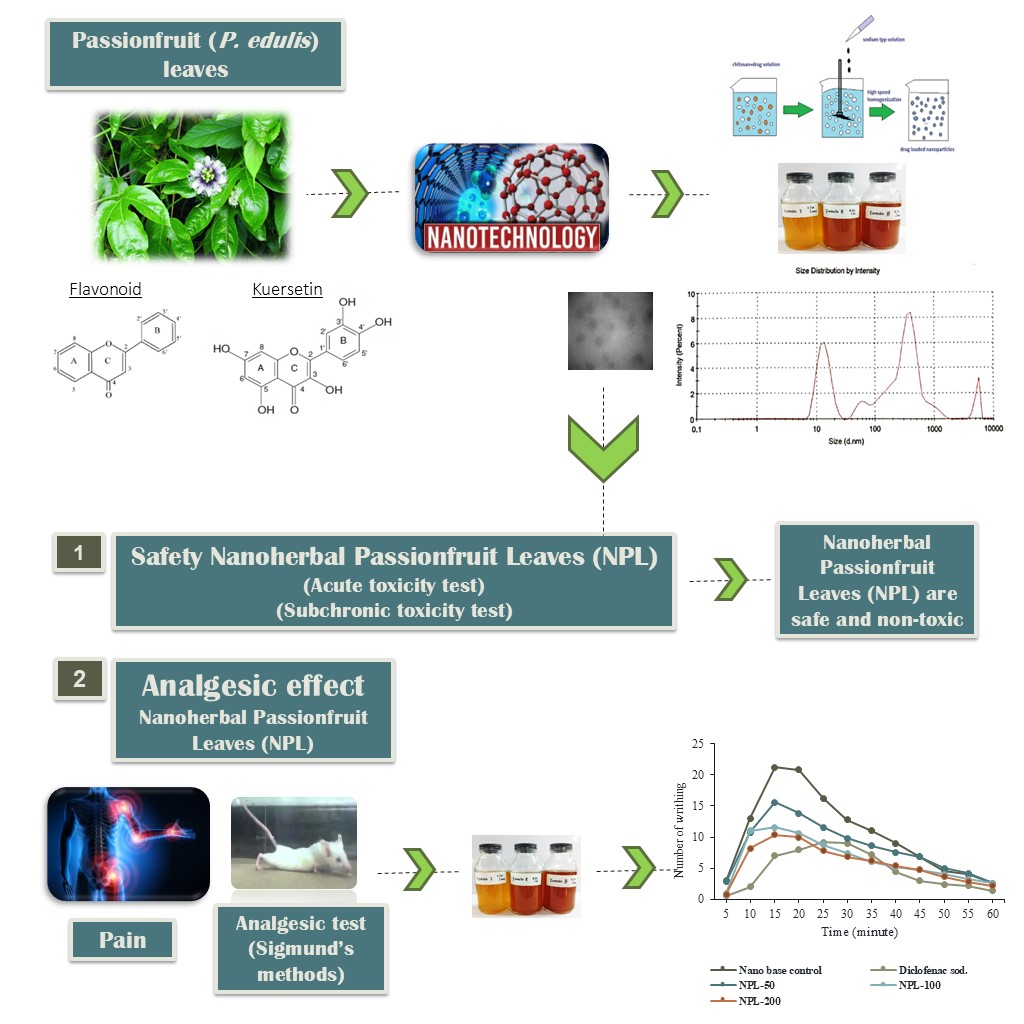
Downloads
References
JK Kim, KH Kim, YC Shin, BH Jang and SG Ko. Utilization of traditional medicine in primary health care in low-and middle-income countries: a systematic review. Health Policy Plan. 2020; 35, 1070-83.
KMM Koriem. Importance of herbapassiflorae in medicinal applications: Review on experimental and clinical pharmacology. Biointerface Res. Appl. Chem. 2021; 11, 12886-900.
X He, F Luan, Y Yang, Z Wang, Z Zhao, J Fang, M Wang, M Zuo and Y Li. Passiflora edulis: an insight into current researches on phytochemistry and pharmacology. Front. Pharmacol. 2020; 11, 617.
S Rai, JC Nagar and M Mukim. Pharmacological and medicinal importance of passiflora edulis: A review. Int. J. Res. Rev. 2022; 9, 341-9.
NMD Sandhiutami, Y Desmiaty, MH Fakhran and Fahleni. IAI special edition: antinociceptive and anti-inflammatory effect of passiflora edulis leaves extract in acetic acid-induced pain and carrageenan-induced paw oedema in vivo. Pharm. Educ. 2024; 24, 78-85.
H Yuan, Q Ma, L Ye and G Piao. The traditional medicine and modern medicine from natural products. Molecules 2016; 21, 559.
JB Calixto. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz. J. Med. Biol. Res. 2000; 33, 179-89.
X Cai, Z Fang, J Dou, A Yu and G Zhai. Bioavailability of quercetin: problems and promises. Curr. Med. Chem. 2013; 20, 2572-82.
J Zhao, J Yang and Y Xie. Improvement strategies for the oral bioavailability of poorly water-soluble flavonoids: An overview. Int. J. Pharm. 2019; 570, 118642.
AR Bilia, V Piazzini, C Guccione, L Risaliti, M Asprea, G Capecchi and MC Bergonzi. Improving on nature: the role of nanomedicine in the development of clinical natural drugs. Planta Med. 2017; 83, 366-81.
MF Manzoor, A Hussain, A Sameen, A Sahar, S Khan, R Siddique, RM Aadil and B Xu. Novel extraction, rapid assessment and bioavailability improvement of quercetin: A review. Ultrason. Sonochem. 2021; 78, 105686.
R Shegokar. Chapter 2 - Role of nanocarriers and their surface modification in targeting delivery of bioactive compounds. In: H Rachmawati, A Larasati, AC Adi and R Shegokar (Eds). Nanopharmaceuticals. Elsevier Science, Amsterdam, Netherland, 2020.
AA Yetisgin, S Cetinel, M Zuvin, A Kosar and O Kutlu. Therapeutic nanoparticles and their targeted delivery applications. Molecules 2020; 25, 2193.
R Kumar and M Sharma. Herbal nanomedicine interactions to enhance pharmacokinetics, pharmacodynamics, and therapeutic index for better bioavailability and biocompatibility of herbal formulations. J. Mater. Nanosci. 2018; 5, 35-60.
J Zhang, K Hu, L Di, P Wang, Z Liu, J Zhang, P Yue, W Song, J Zhang, T Chen, Z Wang, Y Zhang, X Wang, C Zhan, Y Cheng, X li, Q Li, J Fan, Y Shen, J Han and H Qiao. Traditional herbal medicine and nanomedicine: Converging disciplines to improve therapeutic efficacy and human health. Adv. Drug. Deliv. Rev. 2021; 178, 113964.
SR Alizadeh, N Savadkouhi and MA Ebrahimzadeh. Drug design strategies that aim to improve the low solubility and poor bioavailability conundrum in quercetin derivatives. Expert Opin. Drug Discov. 2023; 18, 1117-32.
F Araujo, N Shrestha, PL Granja, J Hirvonen, HA Santos and B Sarmento. Safety and toxicity concerns of orally delivered nanoparticles as drug carriers. Expert Opin. drug Metab. Toxicol. 2015; 11, 381-93.
C Ceylan, ME Tatlipinar, S Tuccar, GZ Omurtag and FJ Akbuga. Evaluation of the in vivo Preclinical Toxicity of Targeted Nanoparticles. 1st ed. Jenny Stanford Publishing, Dubai, United Arab Emirates, 2021.
C Egbuna, VK Parmar, J Jeevanandam, SM Ezzat, KC Patrick-Iwuanyanwu, CO Adetunji, J Khan, EN Onyeike, CZ Uche, M Akram, MS Ibrahim, NME Mahdy, CG Awuchi, K Saravanan, H Tijjani, UE Odoh, M Messaoudi, JC Ifemeje, MC Olisah, NJ Ezeofor, CJ Chikwendu and CG Ibeabuchi. Toxicity of nanoparticles in biomedical application: Nanotoxicology. J. Toxicol. 2021; 2021, 9954443.
R Abbasi, G Shineh, M Mobaraki, S Doughty and L Tayebi. Structural parameters of nanoparticles affecting their toxicity for biomedical applications: A review. J. Nanopart. Res. 2023; 25, 43.
T Jansen, L Claassen, IV Kamp and DRM Timmermans. All chemical substances are harmful: public appraisal of uncertain risks of food additives and contaminants. Food Chem. Toxicol. 2020; 136, 110959.
J Boutrand. Biocompatibility and Performance of Medical Devices. In: WH De Jong, JW Carraway and RE Geertsma (Eds). In vivo and in vitro testing for the biological safety evaluation of biomaterials and medical devices. Woodhead Publishing, Cambridge, 2020.
A Gissi, K Louekari, L Hoffstadt, N Bornatowicz and AM Aparicio. Alternative acute oral toxicity assessment under REACH based on sub-acute toxicity values. ALTEX-Alternatives Anim. Exp. 2017; 34, 353-61.
BPBP Obat and M Nomor. Tahun 2022 on Guidelines for preclinical toxicity tests in vivo. Indonesian Food and Drug Authority, Indonesia, 2022, p. 1-53.
NMD Sandhiutami, RS Dewi, S Khairani and SAM Widyadari. Safety evaluation of the development of a curcumin nanoparticle formula in mice and in-vitro antioxidant potential safety evaluation of the development of a curcumin nanoparticle formula in mice and in-vitro antioxidant potential. Jurnal Ilmu Kefarmasian Indonesia. 2022; 20, 63-72.
MCLD Carmo, IM Martins, AER Magalhaes, MRM Junior and JA Macedo. Passion fruit (Passiflora edulis) leaf aqueous extract ameliorates intestinal epithelial barrier dysfunction and reverts inflammatory parameters in Caco-2 cells monolayer. Food Res. Int. 2020; 133, 109162.
MR Priamsari and CH Kristanti. Effect of Solvent Type on the yield amount and total flavonoid content of yellow passion fruit peel extract (Passiflora edulis var. flavicarpa). Indones. J. Med. Sci. 2023; 10, 37-42.
W Arozal, M Louisa, D Rahmat, P Chendrana and NMD Sandhiutami. Development, characterization and pharmacokinetic profile of chitosan-sodium tripolyphosphate nanoparticles based drug delivery systems for curcumin. Adv. Pharm. Bull. 2021; 11, 77-85.
MH Zarei, Z Lorigooini, HA Khoei and E Bijad. Acute oral toxicity assessment of galbanic acid in albino rat according to OECD 425 TG. Toxicol. Rep. 2023; 11, 111-5.
NMD Sandhiutami, S Khairani, RS Dewi, ZR Hakim and AR Pradani. Anti-inflammatory and analgesic activity of musa balbisiana peels in vivo. Borneo J. Pharm. 2022; 5, 81-92.
NMD Sandhiutami, AT Atayoglu, Y Sumiyati, Y Desmiaty and RA Hidayat. The combination of colocasia esculenta L. and zingiber officinale potentially inhibits inflammation and pain. Jurnal Ilmu Kefarmasian Indonesia 2023; 21, 81-9.
AVV Nikezic, AM Bondzic and VM Vasic. Drug delivery systems based on nanoparticles and related nanostructures. Eur. J. Pharm. Sci. 2020; 151, 105412.
R Kumar, SV Dalvi and PF Siril. Nanoparticle-based drugs and formulations: Current status and emerging applications. ACS Appl. Nano Mater. 2020; 3, 4944-61.
CP Jimenez-Gomez and JA Cecilia. Chitosan: A natural biopolymer with a wide and varied range of applications. Molecules 2020; 25, 3981.
AM Abdelgawad and SM Hudson. Chitosan nanoparticles: polyphosphates cross-linking and protein delivery properties. Int. J. Biol. Macromol. 2019; 136, 133-42.
S Kumar, JP Singh, D Giri and S Mishra. Effect of polydispersity on the dynamics of active brownian particles. Phys. Rev. E 2021; 104, 024601.
J Xu, M Yildiztekin, D Han, C Keskin, A Baran, MF Baran, A Eftekhari, CA Ava, SI Kandemir, DB Cebe, B Dag, A Beilerli and R Khalilov. Biosynthesis, characterization, and investigation of antimicrobial and cytotoxic activities of silver nanoparticles using solanum tuberosum peel aqueous extract. Heliyon 2023; 9, e19061.
J Zhang, Q Xiang, L Shen, J Ling, C Zhou, J Hu and L Chen. Surface charge-dependent bioaccumulation dynamics of silver nanoparticles in freshwater algae. Chemosphere 2020; 247, 125936.
A Yusuf, ARZ Almotairy, H Henidi, OY Alshehri and MS Aldughaim. Nanoparticles as drug delivery systems: a review of the implication of nanoparticles’ physicochemical properties on responses in biological systems. Polymers 2023; 15, 1596.
M Soltanzadeh, SH Peighambardoust, B Ghanbarzadeh, M Mohammadi and JM Lorenzo. Chitosan nanoparticles as a promising nanomaterial for encapsulation of pomegranate (Punica granatum L.) peel extract as a natural source of antioxidants. Nanomaterials 2021; 11, 1439.
K Jafernik, A Ladniak, E Blicharska, K Czarnek, H Ekiert, AE Wiacek and A Szopa. Chitosan-based nanoparticles as effective drug delivery systems - a review. Molecules 2023; 28, 1963.
PN Azizah and Y Herdiana. Review article: Chitosan nanoparticles to improve nutraceutical quality. Farmaka 2023; 21, 399-409.
H Sadaquat, M Akhtar, M Nazir, R Ahmad, Z Alvi and N Akhtar. Biodegradable and biocompatible polymeric nanoparticles for enhanced solubility and safe oral delivery of docetaxel: In vivo toxicity evaluation. Int. J. Pharm. 2021; 598, 120363.
N Munot, U Kandekar, PS Giram, K Khot, A Patil and S Cavalu. A comparative study of quercetin-loaded nanocochleates and liposomes: formulation, characterization, assessment of degradation and in vitro anticancer potential. Pharmaceutics 2022; 14, 1601.
RTA Elbesthi, KY Ozdemir, Y Tacstan, S Bilen and AY Sonmez. Effects of ribwort plantain (plantago lanceolata) extract on blood parameters, immune response, antioxidant enzyme activities, and growth performance in rainbow trout (oncorhynchus mykiss). Fish Physiol. Biochem. 2020; 46, 1295-307.
G Silva-Santana, JC Bax, DCS Fernandes, DTL Bacellar, C Hooper, AASO Dias, CB Silva, AMD Souza, S Ramos, RA Santos,TR Pinto, MA Ramao and AL Mattos-Guaraldi. Clinical hematological and biochemical parameters in swiss, BALB/c, C57BL/6 and B6D2F1 mus musculus. Anim. Models Exp. Med. 2020; 3, 304-15.
NC Azubike, PU Achukwu, CN Okwuosa, DC Nwachukwu, OS Onwukwe and AO Onyemelukwe. Subacute toxicity profile of the leaves of colocasia esculenta [L. Schott] in albino rats. Res. J. Med. Plant 2016; 10, 340-8.
ME Jemli, SM Ezzat, M Kharbach, ES Mostafa, RA Radwan, YE Jemli, O El-Guourrami, S Ahid, Y Cherrh, A Zayed and K Alaoui. Bioassay-guided isolation of anti-inflammatory and antinociceptive metabolites among 3 Moroccan Juniperus leaves extract supported with in vitro enzyme inhibitory assays. J. Ethnopharmacol. 2024; 331, 118285.
N Sa’adah, N Chasanah, SDI Pertami, PD Rohmaniar, AA Adriansyah and AM Ulah. Analgesic effect of trembesi leaf extract (Samanea saman (Jacq.) Merr.) on white mice (Mus musculus). Padjadjaran J. Dent. Res. Students 2022; 6, 120-6.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






