A Review on Heavy Metals Removal using Zerovalent Iron Nanoparticles: Synthesis, Mechanism, Applications, and Challenges
DOI:
https://doi.org/10.48048/tis.2025.9702Keywords:
Adsorption, Challenges, Heavy metals, Mechanism, Oxidation, Reduction, Synthesis, Zerovalent iron nanoparticlesAbstract
The application of zerovalent iron nanoparticles in diverse organic transformations has garnered significant attention over the past decade, primarily due to the high natural availability, low toxicity, and cost-effectiveness of iron metal. The core of zerovalent iron nanoparticles, composed of zerovalent iron, acts as a reducing agent for contaminants, while its outer iron oxide shell offers active sites for chemisorption and facilitates electrostatic interactions with heavy metals. Advances in synthetic methodologies for producing Fe nanoparticles and their stabilization using various support materials have played a pivotal role in enhancing their utility in catalysis. This review consolidates understanding of the nanoscale zerovalent iron structure, its synthesis techniques, removal mechanisms, practical applications for heavy metals such as cadmium (Cd), copper (Cu), and lead (Pb), as well as the challenges associated with its use. This review explores the catalytic properties of zerovalent iron (nZVI) nanoparticles and their critical role in environmental remediation and catalysis. It highlights the factors influencing their performance, including chemical composition, size, surface properties, and the presence of stabilizing agents. The document emphasizes the importance of advanced characterization techniques such as TEM, SEM, and SAXS for understanding particle morphology and structural properties. Challenges such as agglomeration, rapid oxidation, and environmental concerns are addressed, alongside strategies to enhance their efficiency. This comprehensive discussion underscores the significance of nZVI in academic research and industrial applications, providing insights into both their potential and limitations. Additionally, it provides insights into potential future directions for advancing iron nanoparticle-mediated reduction reactions.
HIGHLIGHTS
- The review comprehensively discusses the application of nanomaterials for the removal of highly toxic hexavalent chromium from aqueous environments.
- The parameters include pH, initial concentration, contact time, nanomaterial dosage, and temperature, which affect Cr (VI) removal.
- Cr (VI) removal using nanomaterials involves interconnected mechanisms such as adsorption, electrostatic attraction under acidic conditions, reductive transformation to Cr (III), and subsequent complexation or surface precipitation of Cr (III) on the nanomaterial surfaces.
- Emphasis is placed on the regeneration potential of nanomaterials to enhance sustainability in practical applications.
GRAPHICAL ABSTRACT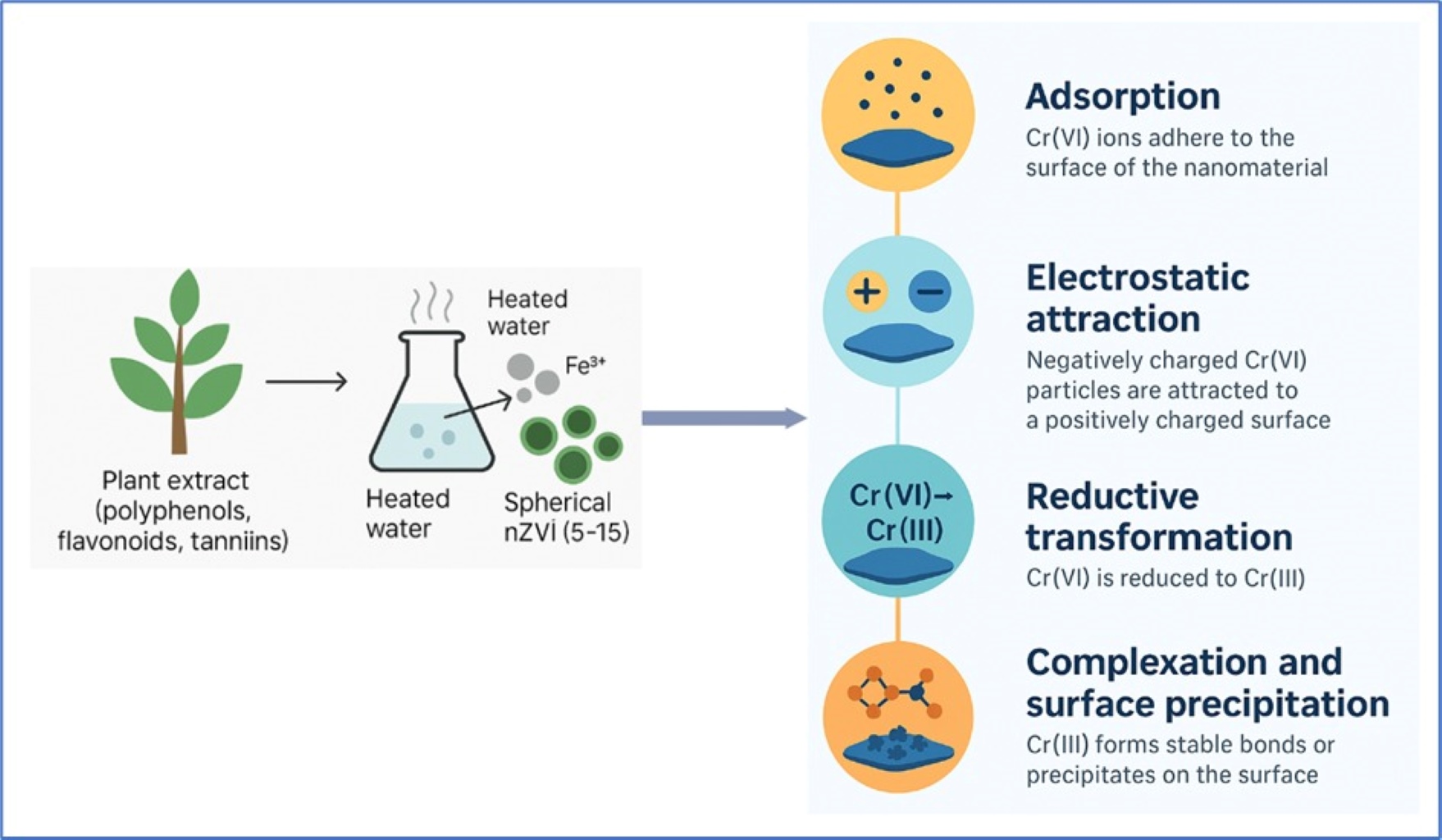
Downloads
References
J Sardans, F Montes and J Peñuelas. Electrothermal atomic absorption spectrometry to determine As, Cd, Cr, Cu, Hg, and Pb in soils and sediments: A review and perspectives. Soil and Sediment Contamination 2011; 20(4), 447-491.
X Wang, A Wang, J Ma and M Fu. Facile green synthesis of functional nanoscale zero-valent iron and studies of its activity toward ultrasound-enhanced decolorization of cationic dyes. Chemosphere 2017; 166, 80-88.
A Verma, A Roy and N Bharadvaja. Remediation of heavy metals using nanophytoremediation. In: MP Shah (Ed.). Advanced oxidation processes for effluent treatment plants. Elsevier, Amsterdam, Netherlands, 2021, p. 273-296.
M Hashim, S Mukhopadhyay, JN Sahu and B Sengupta. Remediation technologies for heavy metal contaminated groundwater. Journal of Environmental Management 2011; 92(10), 2355-2388.
H Karimi-Maleh, A Ayati, S Ghanbari, Y Orooji, B Tanhaei, F Karimi, M Alizadeh, J Rouhi, L Fu and M Sillanpää. Recent advances in removal techniques of Cr(VI) toxic ion from aqueous solution: A comprehensive review. Journal of Molecular Liquids 2021; 329(15), 115062.
G Dognani, P Hadi, H Ma, F Cabrera, AE Job, DL Agostini and BS Hsiao. Effective chromium removal from water by polyaniline-coated electrospun adsorbent membrane. Chemical Engineering Journal 2019; 372(3), 341-351.
A Kumar and A Jha. Drug development strategies. In: A Kumar and A Jha (Eds.). Anticandidal agents. Academic Press, London, 2017, p. 63-71.
Z Pan, X Zhu, A Satpathy, W Li, JD Fortner and DE Giammar. Cr(VI) adsorption on engineered iron oxide nanoparticles: Exploring complexation processes and water chemistry. Environmental Science & Technology 2019; 53(20), 11913-11921.
V Nishad, C Mandal and M Sahu. Study of bioaerosol disinfection kinetics and application of nonlinear regression modeling for optimization of TiO2-based photocatalytic disinfection process. Nanotechnology for Environmental Engineering 2023; 8(4), 911-922.
P Zan, C Yang, H Sun, L Zhao, Z Lv and Y He. One-pot fabricating Fe3O4/graphene nanocomposite with excellent biocompatibility and non-toxicity as a negative MR contrast agent. Colloids Surf B Biointerfaces 2016; 145, 208-216.
M Stefaniuk, P Oleszczuk and YS Ok. Review on nano zerovalent iron (nZVI): From synthesis to environmental applications. Chemical Engineering Journal 2015; 287(3), 618-632.
HK Boparai, M Joseph and DM O’Carroll. Cadmium (Cd2+) removal by nano zerovalent iron: Surface analysis, effects of solution chemistry and surface complexation modeling. Environmental Science and Pollution Research 2013; 20(9), 6210-6221.
M Mahmood-Ul-Hassan, M Yasin, M Yousra, R Ahmad and S Sarwar. Kinetics, isotherms, and thermodynamic studies of lead, chromium, and cadmium bio-adsorption from aqueous solution onto Picea smithiana sawdust. Environmental Science and Pollution Research 2018; 25(13), 12570-12578.
V Madhavi, TNVKV Prasad, AVB Reddy, BR Reddy and G Madhavi. Application of phytogenic zerovalent iron nanoparticles in the adsorption of hexavalent chromium. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2014; 116(C), 17-25.
Y Zou, X Wang, A Khan, P Wang, Y Liu, A Alsaedi, T Hayat and X Wang. Environmental remediation and application of nanoscale zero-valent iron and its composites for the removal of heavy metal ions: A review. Environmental Science & Technology 2016; 50(14), 7290-7304.
Y Li, HY Bi, XM Mao, YQ Liang and H Li. Adsorption behavior and mechanism of core-shell magnetic rhamnolipid-layered double hydroxide nanohybrid for phenolic compounds from heavy metal-phenolic. Applied Clay Science 2018; 162, 230-238.
MS Sankhla and R Kumar. Arsenic-induced neurotoxic & carcinogenic effects on humans. Open Access Journal of Toxicology 2018; 3(4), 555617.
D Chen, Y Cheng, N Zhou, P Chen, Y Wang, K Li, S Huo, P Cheng, P Peng, R Zhang, L Wang, H Liu, Y Liu and R Ruan. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review. Journal of Cleaner Production 2020; 268(3), 121725.
VK Gupta, A Nayak and S Agarwal. Bioadsorbents for remediation of heavy metals: Current status and their future prospects. Environmental Engineering Research 2015; 20(1), 1-18.
C Mandal, V Nishad and M Sahu. Impact of spatial distribution of light intensity on disinfection kinetics of Mycobacterium smegmatis and E. coli using TiO2-based photocatalyst. Nanotechnology for Environmental Engineering 2023; 8(2), 567-579.
J Fisher and World Health Organization. Elemental mercury and inorganic mercury compounds: Human health aspects, Available at: https://apps.who.int/iris/bitstream/handle/10665/42607/9241530502.pdf, accessed July 2003.
F Akbal and S Camcidotless. Copper, chromium and nickel removal from metal plating wastewater by electrocoagulation. Desalination 2011; 269(1-3), 214-222.
B Doshi, A Ayati, B Tanhaei, E Repo and M Sillanpää. Partially carboxymethylated and partially cross-linked surface of chitosan versus the adsorptive removal of dyes and divalent metal ions. Carbohydrate Polymers 2018; 197, 586-597.
A Zeraatkar Moghaddam, E Ghiamati, A Ayati and MR Ganjali. Application of the response surface methodology for optimizing the adsorptive removal of chromate using a magnetic crosslinked chitosan nanocomposite. Journal of Applied Polymer Science 2019; 136(8), 47077.
VE Pakade, NT Tavengwa and LM Madikizela. Recent advances in hexavalent chromium removal from aqueous solutions by adsorptive methods. RSC Advances 2019; 9(45), 26142-26164.
Z Farooqi, M Akram, R Begum, W Wu and A Irfan. Inorganic nanoparticles for reduction of hexavalent chromium: Physicochemical aspects. Journal of Hazardous Materials 2021; 402, 123535.
BH Hintermeyer and EL Tavani. Adsorption, biosorption and bioaccumulation used to remove chromium (III) from tanning wastewaters: A critical review. Journal of the Society of Leather Technologists and Chemists 2013; 97(6), 231-237.
B Tanhaei, MP Chenar, N Saghatoleslami, M Hesampour, M Kallioinen, M Sillanpää and M Mänttäri. Removal of nickel ions from aqueous solution by micellar-enhanced ultrafiltration, using mixed anionic-non-ionic surfactants. Separation and Purification Technology 2014; 138, 169-176.
U Misra, V Nishad and SP Singh. Introduction to membrane distillation and its application in emerging contaminants removal. Energy, Environment, and Sustainability. In: SP Singh, AK Agarwal, T Gupta and SM Maliyekkal (Eds.). New trends in emerging environmental contaminants. Springer, Singapore, 2022, p. 427-462.
X Pei, L Gan, Z Tong, H Gao, S Meng, W Zhang, P Wang and Y Chen. Robust cellulose-based composite adsorption membrane for heavy metal removal. Journal of Hazardous Materials 2021; 406, 124746.
JE Efome, D Rana, T Matsuura and CQ Lan. Experiment and modeling for flux and permeate concentration of heavy metal ion in adsorptive membrane filtration using a metal-organic framework incorporated. Chemical Engineering Journal 2018; 352, 737-744.
S Deng, X Liu, J Liao, H Lin and Fang Liu. PEI modified multiwalled carbon nanotube as a novel additive in PAN nanofiber membrane for enhanced removal of heavy metal ions. Chemical Engineering Journal 2019; 375, 122086.
Z Zhao, H An, J Lin, M Feng, V Murugadoss, T Ding, H Liu, Q Shao, X Mai, N Wang, H Gu, S Angaiah and Z Guo. Progress on the photocatalytic reduction removal of chromium contamination. The Chemical Record 2019; 19(5), 873-882,
Y Liu, D Yang, T Xu, Y Shi, L Song and ZZ Yu. Continuous photocatalytic removal of chromium (VI) with structurally stable and porous Ag/Ag3PO4/reduced graphene oxide microspheres. Chemical Engineering Journal 2020; 379, 122200.
G Niu, C Si, J Jiao, Q Han, M Guo and M Li. An electron-rich metal-organic framework for highly efficient photocatalytic reduction of Cr (VI). Journal of Alloys and Compounds 2020; 830(224), 154696.
TK Tran, KF Chiu, CY Lin and HJ Leu. Electrochemical treatment of wastewater: Selectivity of the heavy metals removal process. International Journal of Hydrogen Energy 2017; 42(45), 27741-27748.
C Carolin, P Kumar, A Saravanan, GJ Joshiba and M Naushad. Efficient techniques for the removal of toxic heavy metals from aquatic environment: A review. Journal of Environmental Chemical Engineering 2017; 5(3), 2782-2799.
LY Kit and JS Chang. Bioremediation of heavy metals using microalgae: Recent advances and mechanisms. Bioresource Technology 2020; 303(2), 122886.
V Kumar and SK Dwivedi. Hexavalent chromium reduction ability and bioremediation potential of Aspergillus flavus CR500 isolated from electroplating wastewater. Chemosphere 2019; 237, 124567.
S Singh, D Kapoor, S Khasnabis, J Singh and PC Ramamurthy. Mechanism and kinetics of adsorption and removal of heavy metals from wastewater using nanomaterials. Environmental Chemistry Letters 2021; 19(3), 2351-2381.
O Lobacheva, N Dzhevaga and A Danilov. The method of removal yttrium (III) and ytterbium (III) from dilute aqueous solutions. Journal of Ecological Engineering 2016; 17(2), 38-42.
FS Hoseinian, B Rezai and E Kowsari. The main factors effecting the efficiency of Zn (II) flotation: Optimum conditions and separation mechanism. Journal of Environmental Management 2018; 207, 169-179.
E Deliyanni, GZ Kyzas and KA Matis. Various flotation techniques for metal ions removal. Journal of Molecular Liquids 2016; 225, 260-264.
AS Abyaneh and MH Fazaelipoor. Evaluation of rhamnolipid (RL) as a biosurfactant for the removal of chromium from aqueous solutions by precipitate flotation. Journal of Environmental Management 2016; 165, 184-187.
S Aoudj, A Khelifa, N Drouiche, R Belkada and D Miroud. Simultaneous removal of chromium (VI) and fluoride by electrocoagulation-electroflotation: Application of a hybrid Fe-Al anode. Chemical Engineering Journal 2015; 267, 153-162.
J Korak, R Huggins and M Arias-Paic. Regeneration of pilot-scale ion exchange columns for hexavalent chromium removal. Water Research 2017; 118, 141-151.
R Soltani, A Marjani, M Hosseini and S Shirazian. Synthesis and characterization of novel N-methylimidazolium-functionalized KCC-1: A highly efficient anion exchanger of hexavalent chromium. Chemosphere 2020; 239, 124735.
SVAR Sastry, BS Rao and DV Padma. Studies on selective batch adsorption of Cu(II) and Cr(VI) from aqueous solution. TEST Engineering and Management 2020; 83, 6794-6797.
S Bhattacharjee, F Habib, N Darwish and A Shanableh. Iron sulfide nanoparticles prepared using date seed extract: Green synthesis, characterization and potential application for removal of ciprofloxacin and chromium. Powder Technology 2021; 380(3), 219-228.
W Xu, T Yang, S Liu, L Du, Q Chen, X Li, J Dong, Z Zhang, S Lu, Y Gong, L Zhou, Y Liu and X Tan. Insights into the synthesis, types and application of iron nanoparticles: The overlooked significance of environmental effects. Environment International 2022; 158(2), 106980.
D Karabelli, Ç Üzüm, T Shahwan, AE Eroğlu, TB Scott, KR Hallam and I Lieberwirth. Batch removal of aqueous Cu2+ ions using nanoparticles of zero-valent iron: A study of the capacity and mechanism of uptake. Industrial & Engineering Chemistry Research 2008; 47(14), 4758-4764.
RA Crane and TB Scott. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. Journal of Hazardous Materials 2012; 211-212, 112-125.
SVAR Sastry. Green synthesis and characterization of silver nano particles. Journal of Water and Environmental Nanotechnology 2020; 5, 81-91.
MAV Ramos, W Yan, X Li, BE Koel and W Zhang. Simultaneous oxidation and reduction of arsenic by zero-valent iron nanoparticles: Understanding the significance of the core-shell structure. Journal of Physical Chemistry 2009; 113(33), 14591-14594.
P Wang, J Hu, T Liu, G Han, W Ma and J Li. New insights into ball-milled zero-valent iron composites for pollution remediation: An overview. Journal of Cleaner Production 2023; 385, 135513.
MM Tarekegn, AM Hiruy and AH Dekebo. Nano zero valent iron (nZVI) particles for the removal of heavy metals (Cd2+, Cu2+ and Pb2+) from aqueous solutions. RSC Advances 2021; 11(30), 18539-18551.
MM Serda. 2013, Synteza i aktywność biologiczna nowych analogów tiosemikarbazonowych chelatorów żelaza. Ph. D. Dissertation. Uniwersytet śląski, Katowice, Poland.
C Kim, JY Ahn, TY Kim, WS Shin and I Hwang. Activation of persulfate by nanosized zero-valent iron (NZVI): mechanisms and transformation products of NZVI. Environmental Science & Technology 2018; 52(6), 3625-3633.
N Nassar and M Husein. Preparation of iron oxide nanoparticles from FeCl3 solid powder using microemulsions. Physica Status Solidi (A) Applications and Materials 2006; 203(6), 1324-1328.
R Mukherjee, R Kumar, A Sinha, Y Lama and AK Saha. A review on synthesis, characterization, and applications of nano zero valent iron (nZVI) for environmental remediation. Critical Reviews in Environmental Science and Technology 2016; 46(5), 443-466.
TK Teal, DP Lies, BJ Wold and DK Newman. Spatiometabolic stratification of Shewanella oneidensis biofilms. Applied and Environmental Microbiology 2006; 72(11), 7324-7330.
DV Padma and SVAR Sastry. Biosorption of hexavalent chromium using mallet flower leaves powder as adsorbent. TEST Engineering and Management 2020; 83, 15714-15729.
KP Kumar, W Paul and CP Sharma. Green synthesis of gold nanoparticles with Zingiber officinale extract: Characterization and blood compatibility. Process Biochemistry 2011; 46(10), 2007-2013.
T Wang, J Lin, Z Chen, M Megharaj and R Naidu. Green synthesized iron nanoparticles by green tea and eucalyptus leaves extracts used for removal of nitrate in aqueous solution. Journal of Cleaner Production 2014; 83, 413-419.
MA Ebrahimzadeh, S Mortazavi-Derazkola and MA Zazouli. Eco-friendly green synthesis and characterization of novel Fe3O4/SiO2/Cu2O-Ag nanocomposites using Crataegus pentagyna fruit extract for photocatalytic degradation of organic contaminants. Journal of Materials Science: Materials in Electronics 2019; 30(12), 10994-11004.
KS Prasad, P Gandhi and K Selvaraj. Synthesis of green nano iron particles (GnIP) and their application in adsorptive removal of As(III) and As(V) from aqueous solution. Applied Surface Science 2014; 317, 1052-1059.
M. Nasrollahzadeh, S. M. Sajadi, and M. Maham. Green synthesis of palladium nanoparticles using Hippophae rhamnoides Linn leaf extract and their catalytic activity for the Suzuki-Miyaura coupling in water. Journal of Molecular Catalysis A: Chemical 2015; 396, 297-303.
X Weng, L Huang, Z Chen, M Megharaj and R Naidu. Synthesis of iron-based nanoparticles by green tea extract and their degradation of malachite. Industrial Crops and Products 2013; 51, 342-347.
S Li, W Wang, W Yan and WX Zhang. Nanoscale zero-valent iron (nZVI) for the treatment of concentrated Cu (II) wastewater: A field demonstration. Environmental Science: Processes & Impacts 2014; 16(3), 524-533.
M Nasrollahzadeh, S Mohammad Sajadi, A Rostami-Vartooni and M Khalaj. Green synthesis of Pd/Fe3O4 nanoparticles using Euphorbia condylocarpa M. bieb root extract and their catalytic applications as magnetically recoverable and stable recyclable catalysts for the phosphine-free Sonogashira and Suzuki coupling reactions. Journal of Molecular Catalysis A: Chemical 2015; 396, 31-39.
M Zhang, F He, D Zhao and X Hao. Degradation of soil-sorbed trichloroethylene by stabilized zero valent iron nanoparticles: Effects of sorption, surfactants, and natural organic matter. Water Research 2011; 45(7), 2401-2414.
WX Zhang and DW Elliott. Applications of iron nanoparticles for groundwater remediation. Remediation Journal 2006; 16(2), 7-21.
Y Zhang, J Huang, Z Dong, Y Zhan, J Xi, J Xiao, S Huang and F Tian. Pd-Fe bimetallic nanoparticles anchored on N-doped carbon-modified graphene for efficient catalytic organic reactions. Carbon Letters 2023; 33(1), 77-87.
F He and D Zhao. Hydrodechlorination of trichloroethene using stabilized Fe-Pd nanoparticles: Reaction mechanism and effects of stabilizers, catalysts and reaction conditions. Applied Catalysis B: Environmental 2008; 84(3-4), 533-540.
BI Kharisov, OV Kharissova and HVR Dias. Nanomaterials for environmental protection. Wiley, New Jersey, U.S, 2015, p. 592.
J Quinn, C Geiger, C Clausen, K Brooks, C Coon, S O’Hara, T Krug, D Major, WS Yoon, A Gavaskar and T Holdsworth. Field demonstration of DNAPL dehalogenation using emulsified zero-valent iron. Environmental Science & Technology 2005; 39(5), 1309-1318.
P Bennett, F He, D Zhao, B Aiken and L Feldman. In situ testing of metallic iron nanoparticle mobility and reactivity in a shallow granular aquifer. Journal of Contaminant Hydrology 2010; 116(1-4), 35-46.
X Li, DW Elliott and WX Zhang. Zero-valent iron nanoparticles for abatement of environmental pollutants: Materials and engineering aspects. Critical Reviews in Solid State and Material Sciences 2006; 31(4), 111-122.
C Gao, W Yu, Y Zhu, M Wang, Z Tang, L Du, M Hu, L Fang and X Xiao. Preparation of porous silicate supported micro-nano zero-valent iron from copper slag and used as persulfate activator for removing organic contaminants. Science of The Total Environment 2021; 754, 142131.
H Dong, L Li, Y Wang, Q Ning, B Wang and G Zeng. Aging of zero-valent iron-based nanoparticles in aqueous environment and the consequent effects on their reactivity and toxicity. Water Environment Research 2020; 92, 646-661.
T Phenrat, T Thongboot and GV Lowry. Electromagnetic induction of zerovalent iron (ZVI) powder and nanoscale zerovalent iron (NZVI) particles enhances dechlorination of trichloroethylene in contaminated groundwater and soil: Proof of concept. Environmental Science & Technology 2016; 50(2), 872-880.
X Dong, LQ Ma and Y Li. Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. Journal of Hazardous Materials 2011; 190(1-3), 909-915.
C Wang, DR Baer, JE Amonette, MH Engelhard, J Antony and Y Qiang. Morphology and electronic structure of the oxide shell on the surface of iron nanoparticles. Journal of the American Chemical Society 2009; 131(25), 8824-8832.
W Stumm and JJ Morgan. Aquatic chemistry: Chemical equilibria and rates in natural waters. 3 Ed. Wiley-Interscience, New Jersey, U.S., 2012, p. 1827.
RBN Baig, S Verma, RS Varma and MN Nadagouda. Magnetic Fe@g-C3N4: A photoactive catalyst for the hydrogenation of alkenes and alkynes. ACS Sustainable Chemistry & Engineering 2016; 4(3), 1661-1664.
S Mourdikoudis, RM Pallares and NTK Thanh. Characterization techniques for nanoparticles: Comparison and complementarity upon studying nanoparticle properties. Nanoscale 2018; 27(10), 12871-12934.
Z Ban, YA Barnakov, F Li, VO Golub and CJ O’Connor. The synthesis of core-shell iron@gold nanoparticles and their characterization. Journal of Materials Chemistry 2005; 15(43), 4660-4662.
AL Kustov, AM Frey, KE Larsen, T Johannessen, JK Nørskov and CH Christensen. CO methanation over supported bimetallic Ni-Fe catalysts: From computational studies towards catalyst optimization. Applied Catalysis A: General 2007; 320(2), 98-104.
N Koi, T Oku and M Nishijima. Fe nanowire encapsulated in boron nitride nanotubes. Solid State Commun 2005; 136(6), 342-345.
R Hudson, V Chazelle, M Bateman, R Roy, CJ Li and A Moores. Sustainable synthesis of magnetic ruthenium-coated iron nanoparticles and application in the catalytic transfer hydrogenation of ketones. ACS Sustainable Chemistry & Engineering 2015; 3(5), 814-820.
Z Pan, Y Lin, B Sarkar, G Owens and Z Chen. Green synthesis of iron nanoparticles using red peanut skin extract: Synthesis mechanism, characterization and effect of conditions on chromium removal. Journal of Colloid and Interface Science 2019; 558, 106-114.
Y Wei, Z Fang, L Zheng and EP Tsang. Biosynthesized iron nanoparticles in aqueous extracts of Eichhornia crassipes and its mechanism in the hexavalent chromium removal. Applied Surface Science 2017; 399, 322-329.
MS Abesekara, KNR Kosvinna and BMWPK Amarasinghe. Adsorption and desorption studies of Ni2+ ions on to coconut shell char. IOP Conference Series: Earth and Environmental Science 2020; 427, 012005.
R Garg, M Mittal, S Tripathi and NO Eddy. Core to concept: Synthesis, structure, and reactivity of nanoscale zero-valent iron (NZVI) for wastewater remediation. Environmental Science and Pollution Research 2024; 31(60), 67496-67520.
Q Chen, Y Yao, X Li, J Lu, J Zhou and Z Huang. Comparison of heavy metal removals from aqueous solutions by chemical precipitation and characteristics of precipitates. Journal of Water Process Engineering 2018; 26, 289-300.
R Anjali and S Shanthakumar. Insights on the current status of occurrence and removal of antibiotics in wastewater by advanced oxidation processes. Journal of Environmental Management 2019; 246, 51-62.
RV Mathai, JC Mitra, SK Sar and MK Jindal. Adsorption of chromium (VI) from aqueous phase using Aegle marmelos leaves: Kinetics, isotherm and thermodynamic studies. Chemical Data Collections 2022; 39, 100871.
X Zhao, W Liu, Z Cai, B Han, T Qian and D Zhao. An overview of preparation and applications of stabilized zero-valent iron nanoparticles for soil and groundwater remediation. Water Research 2016; 100(1), 245-266.
NF Fahim, BN Barsoum, AE Eid and MS Khalil. Removal of chromium(III) from tannery wastewater using activated carbon from sugar industrial waste. Journal of Hazardous Materials 2006; 136(2), 303-309.
PS Kumar, S Ramalingam, V Sathyaselvabala, SD Kirupha, A Murugesan and S Sivanesan. Removal of cadmium(II) from aqueous solution by agricultural waste cashew nut shell. Korean Journal of Chemical Engineering 2012; 29(6), 756-768.
G Basaran, D Kavak, N Dizge, Y Asci, M Solener and B Ozbey. Comparative study of the removal of nickel(II) and chromium(VI) heavy metals from metal plating wastewater by two nanofiltration membranes. Desalination Water Treat 2016; 57(46), 21870-21880.
JJ Moreno-Barbosa, C López-Velandia, ADP Maldonado, L Giraldo and JC Moreno-Piraján. Removal of lead(II) and zinc(II) ions from aqueous solutions by adsorption onto activated carbon synthesized from watermelon shell and walnut shell. Adsorption 2013; 19(2-4), 675-685.
R Wang, L Deng, X Fan, K Li, H Lu and W Li. Removal of heavy metal ion cobalt (II) from wastewater via adsorption method using microcrystalline cellulose-magnesium hydroxide. International Journal of Biological Macromolecules 2021; 189(1), 607-617.
Y Sun, X Li, W Zhang and HP Wang. A method for the preparation of stable dispersion of zero-valent iron nanoparticles. Colloids and Surfaces A Physicochemical and Engineering Aspects 2007; 308(1-3), 60-66.
F Mujeeb, P Bajpai and N Pathak. Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of Aegle marmelos. BioMed Research International 2014; 2014(1), 497606.
L Shi, T Wang, H Zhang, K Chang, X Meng, H Liu and J Ye. An amine‐functionalized iron (III) metal-organic framework as efficient visible‐light photocatalyst for Cr (VI) reduction. Advanced Science 2015; 2(3), 1500006.
NA Rashidi and S Yusup. A review on recent technological advancement in the activated carbon production from oil palm wastes. Chemical Engineering Journal 2017; 314, 277-290.
Y Wu, CYu Guan, N Griswold, L Hou, F Xin, A Hu, Z Hu and CP Yu. Zero-valent iron-based technologies for removal of heavy metal(loid)s and organic pollutants from the aquatic environment: Recent advances and perspectives. Journal of Cleaner Production 2020; 277(7), 123478
BR Albuquerque, SA Heleno, M Oliveira, L Barros and ICFR Ferreira. Phenolic compounds: Current industrial applications, limitations and future challenges. Food & Function 2021; 12(1), 14-29.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






