Analysis of Bioactive Compounds Piper crocatum as Inhibitors of Acetylcholinesterase In Silico and In Vitro
DOI:
https://doi.org/10.48048/tis.2025.9437Keywords:
Acetylcholinesterase inhibitors, Alzheimer’s disease, Bioactive compounds, Columbin molecular docking, Piper crocatum, PharmacokineticsAbstract
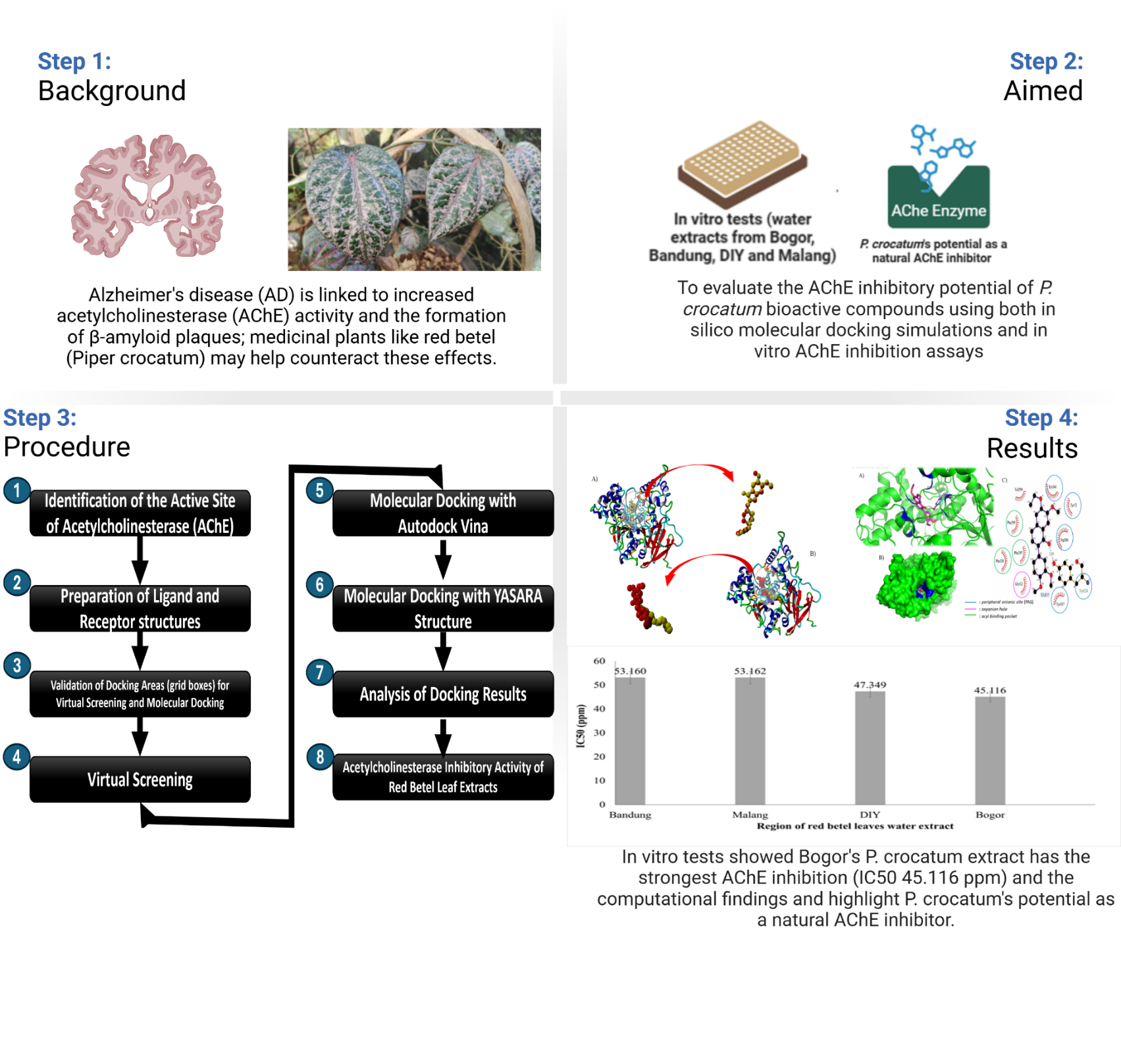
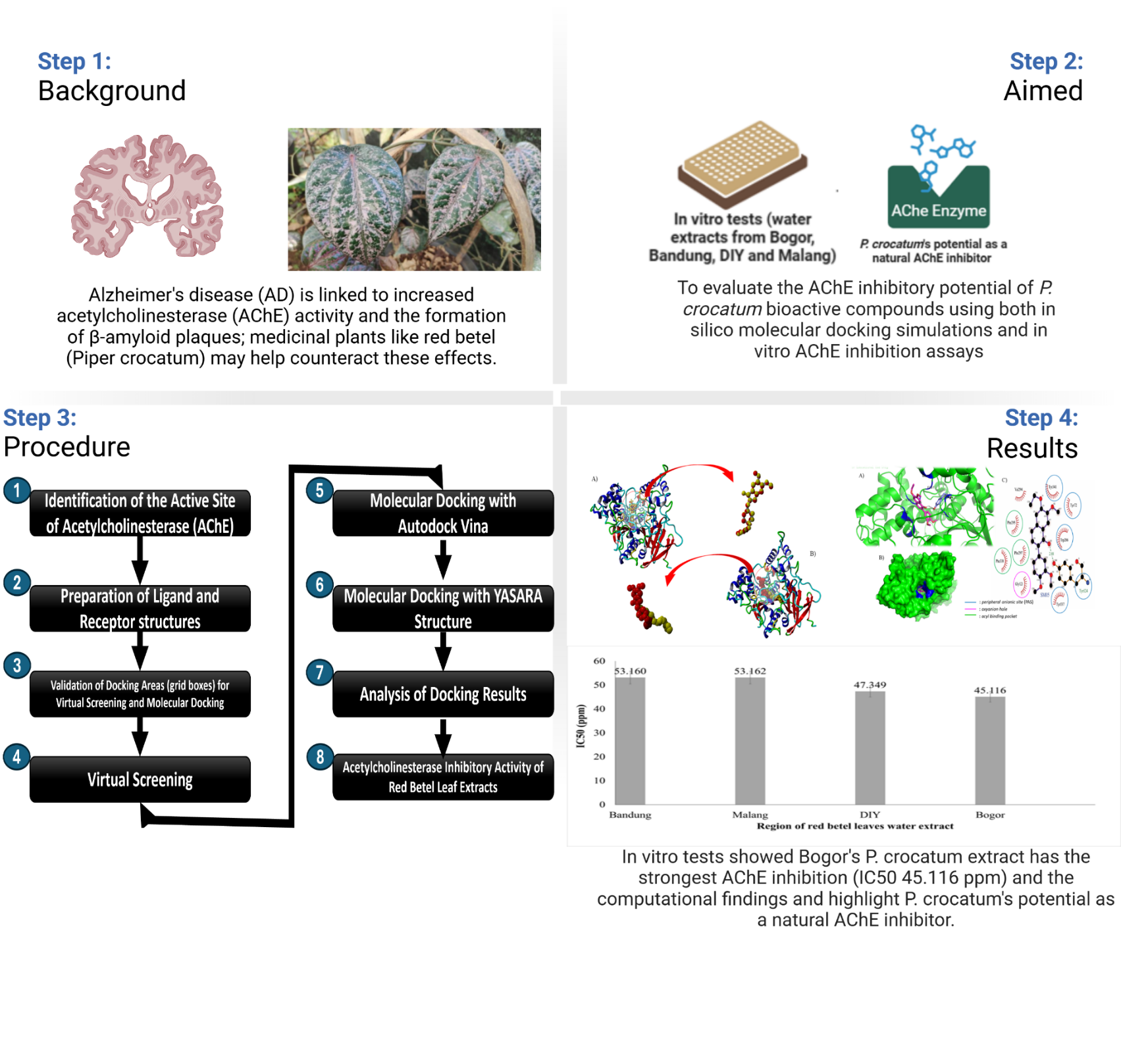
Alzheimer’s disease, the leading cause of dementia in older adults, involves memory loss and cognitive decline, with β-amyloid plaques and neurofibrillary tangles (NFTs) as key features. Acetylcholinesterase (AChE), an enzyme that breaks down acetylcholine, plays a role in the formation of these plaques and tangles. AChE is a promising target for the development of small molecule inhibitors in Alzheimer’s disease (AD) treatment. Indonesia’s native red betel (Piper crocatum) contains bioactive compounds that inhibit AChE activity, as shown in previous research. This study aims to evaluate the AChE inhibitory potential of bioactive compounds from P. crocatum extracts (water, n-hexane and ethyl acetate) using an in silico approach (molecular docking) with 3 different docking software programs. Donepezil served as the reference compound, and the results were compared with in vitro AChE inhibition assays. The potential AChE inhibitors, based on molecular docking using PyRx, Autodock Vina and YASARA Structure, from each extract are SM05 (n-hexane extract), SM15 (water extract) and SM18 (ethyl acetate extract), with the most negative ΔGbind values, measuring −8.8, −9.2 and −11.2 kcal/mol (more negative than Donepezil’s ΔGbind values). SM15 and SM18 Compounds show promise, based on its ΔGbind values, interactions with AChE, favorable pharmacokinetic properties, bioavailability, bioactivity and toxicity positioning both compounds as strong candidates for AD therapy. Both compounds were docked to the AChE substrate binding pockets (6O4W), forming hydrogen bonds with His447 and Phe297 at the “gorge” active site, and hydrophobic interactions with key amino acids in the peripheral anionic site (PAS) and substrate-binding sites (Tyr124, Phe297 and Phe338). This is consistent with in vitro assay results, which show that the ethyl acetate extract has strong inhibition, with an IC50 of 16.7908 ppm, while the water infusion extract yields a 26.621 % inhibition of AChE enzyme activity. In addition, the DIY extract exhibited the strongest AChE inhibitory activity with an IC50 of 40.799 ppm.
HIGHLIGHTS
- Piper crocatum (red betel leaf) shows potential as a natural acetylcholinesterase (AChE) inhibitor, with compounds SM05 (2-(3,4-Dimethoxyphenyl)-6-ethoxy-7-methoxy-1-naphthol), SM15 (Columbin) and SM18 (Flemiphilippinin A) identified as promising candidates through in silico molecular modeling, as indicated by the most negative binding free energy values generated through 3 molecular docking methods (Virtual Screening with PyRx, Autodock Vina and YASARA Structure), especially compound SM18 (Flemiphilippinin A) from ethyl acetate extract (−11.2 kcal/mol). This aligns with the in vitro assay results, which show that the ethyl acetate extract has the strong inhibition, with an IC50 of 16.7908 ppm, as provided by Nurinsani et al. [10].
- The red betel leaves water extract from DIY exhibited the most potent acetylcholinesterase (AChE) inhibitory activity with the lowest IC50 value (40.799 ppm), highlighting the significant inhibitory activity of the extract.
- Flemiphilippinin A (SM18) showed the strongest binding affinity to AChE (ΔGbind −2 kcal/mol), supported by favorable pharmacokinetic properties and bioavailability, as indicated by the fulfillment of Lipinski’s Rule of 5, ADME and bioactivity scores. It acts as a Nuclear receptor ligand (0.53), while Columbin (SM15) exhibits bioactivity as a Nuclear receptor ligand [0.66] and as an Enzyme inhibitor (0.57), suggesting that both compounds have potential as therapeutic candidates for Alzheimer’s disease (AD).
- Virtual screening and molecular docking revealed robust interactions of SM05, SM15 and SM18 with critical AChE active site residues, indicating their capability to inhibit AChE activity and potentially prevent AD progression.
- The 3 compounds from red betel leaf fractions are safe and have potential as oral drugs for Alzheimer’s therapy, with oral toxicity similar to donepezil (toxicity class IV, LD50 505 mg/kg) based on ProTox3 Prediction (with toxicity class IV for SM15, toxicity class V for SM05 and SM18).
- Pharmacokinetic evaluation confirmed the 3 compounds met Lipinski’s rule of 5 and showed high gastrointestinal absorption, with SM15 excelling in drug-likeness and bioactivity parameters critical for AD therapy.
GRAPHICAL ABSTRACT
Downloads
References
SH Lu, JW Wu, HL Liu, JH Zhao, KT Liu, CK Chuang, HY Lin, WB Tsai and Y Ho. The discovery of potential acetylcholinesterase inhibitors: A combination of pharmacophore modeling, virtual screening, and molecular docking studies. Journal of Biomedical Science 2011; 18, 8.
PA Nogara, AR de Saraiva, DC Bueno, LJ Lissner, CLD Corte, MM Braga, DB Rosemberg and JBT Rocha. Virtual screening of acetylcholinesterase inhibitors using the Lipinski’s rule of 5 and ZINC databank. BioMed Research International 2015; 2015(1), 870389.
A Hálová, J Janoutová, L Ewerlingová, V Janout, O Bonczek, T Zeman, T Gerguri, VJ Balcar and O Šerý. CHAT gene polymorphism rs3810950 is associated with the risk of Alzheimer’s disease in the Czech population. Journal of Biomedical Science 2018; 25, 41.
Alzheimer’s Disease International. World Alzheimer report 2023 reducing dementia risk: Never too early, never too late. Alzheimer’s Disease International, London, 2023.
MS García-Ayllón, DH Small, J Avila and J Sáez-Valero. Revisiting the role of acetylcholinesterase in Alzheimer’s disease: Cross-talk with P-tau and β-amyloid. Frontiers in Molecular Neuroscience 2011; 4, 22.
S Darvesh. Butyrylcholinesterase as a diagnostic and therapeutic target for Alzheimer’s disease. Current Alzheimer Research 2016; 13(10), 1173-1177.
R Khoury, N Grysman, J Gold, K Patel and GT Grossberg. The role of 5 HT6-receptor antagonists in Alzheimer’s disease: An update. Expert Opinion on Investigational Drugs 2018; 27(6), 523-533.
S Bordoloi, K Pathak, M Devi, R Saikia, J Das, VH Kashyap, D Das, MZ Ahmad and BA Abdel-Wahab. Some promising medicinal plants used in Alzheimer’s disease: An ethnopharmacological perspective. Discover Applied Sciences 2024; 6(5), 215.
RG Putri, M Safithri, H Husnawati and R Kurniasih. Penambatan molekuler senyawa aktif sirih merah (Piper crocatum) pada butirilkolinesterase sebagai kandidat antialzheimer (in Indonesian). ALCHEMY Jurnal Penelitian Kimia 2023; 19(1), 68-85.
EYY Nurinsani, D Andrianto and M Safithri. Acetylcholinesterase inhibition activity and phytochemical screening of red betel leaf (Piper crocatum Ruiz & Pav) as anti-dementia agents. BIO Web of Conferences 2024; 123, 02009.
World Health Organization. Global accelerated action for the health of adolescents (AA-HA!). World Health Organization, Geneva, Switzerland, 2023.
M Safithri and A Kurniawati. Formula of Piper crocatum, Cinnamomum burmanii, and Zingiber officinale extracts as a functional beverage for diabetics. International Food Research Journal 2016; 23(3), 1123-1130.
S Wang, X Kong, Z Chen, G Wang, J Zhang and J Wang. Role of natural compounds and target enzymes in the treatment of Alzheimer’s disease. Molecules 2022; 27(13), 4175.
R Kurniasih. 2018, Struktur prediksi dan identifikasi substrat spesifik lakase isolat lokal neorspora crassa inaCC F226 (in Indonesian). Ph. D. Dissertation. Bogor Agricultural University, Bogor, Indonesia.
R Agarwal and JC Smith. Speed vs accuracy: Effect on ligand pose accuracy of varying box size and exhaustiveness in AutoDock Vina. Molecular Informatics 2023; 42(2), 2200188.
O Trott and AJ Olson. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry 2010; 31(2), 455-461.
M Weni, M Safithri and DSH Seno. Molecular docking of active compounds Piper crocatum on the a-glucosidase enzyme as antidiabetic. Indonesian Journal of Pharmaceutical Science and Technology 2020; 7(2), 64-72.
AS Abouzied, S Alqarni, KM Younes, SM Alanazi, DM Alrsheed, RK Alhathal, B Huwaimel and AM Elkashlan. Structural and free energy landscape analysis for the discovery of antiviral compounds targeting the cap-binding domain of influenza polymerase PB2. Scientific Reports 2024; 14(1), 25441.
BD Rosiarto, AR Puspaningtyas and D Holidah. Studi aktivitas antioksidan senyawa 1-(p-klorobenzoiloksimetil)-5-fluorourasil dengan metode molecular docking dan metode DPPH (antioxidant activity of 1-(p-chlorobenzoyloxymethyl)-5-fluorouracyl using molecular docking and DPPH method) (in Indonesian). E-Jurnal Pustaka Kesehatan 2014; 2(1), 95-99.
H Dvir, I Silman, M Harel, TL Rosenberry and JL Sussman. Acetylcholinesterase: From 3D structure to function. Chemico-Biological Interactions 2010; 187(1-3), 10-22.
J Cheung, MJ Rudolph, F Burshteyn, MS Cassidy, EN Gary, J Love, MC Franklin and JJ Height. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. Journal of Medicinal Chemistry 2012; 55(22), 10282-10286.
C Seniya, GJ Khan and K Uchadia. Identification of potential herbal inhibitor of acetylcholinesterase associated Alzheimer’s disorders using molecular docking and molecular dynamics simulation. Biochemistry Research International 2014; 2014(1), 705451.
XY Meng, HX Zhang, M Mezei and M Cui. Molecular docking: A powerful approach for structure-based drug discovery. Current Computer Aided-Drug Design 2011; 7(2), 146-157.
M Atanasova, N Yordanov, I Dimitrov, S Berkov and I Doytchinova. Molecular docking study on galantamine derivatives as cholinesterase inhibitors. Molecular Informatics 2015; 34(6-7), 394-403.
I Doytchinova, M Atanasova, I Valkova, G Stavrakov, I Philipova, Z Zhivkova, D Zheleva-Dimitrova, S Konstantinov and I Dimitrov. Novel hits for acetylcholinesterase inhibition derived by docking-based screening on ZINC database. Journal of Enzyme Inhibition and Medicinal Chemistry 2018; 33(1), 768-776.
F Ramadani. Aktivitas antioksidan, total tanin ekstrak dan fraksi daun sirih merah (Piper crocatum) dan identifikasi dengan LC-MS (in Indonesian). Bogor Agricultural University, Bogor, Indonesia, 2018.
CK Joshi and HK Joshi. Molecular docking analysis of acetylcholinesterase with phytocompounds from medicinal plants. Multilogic in Science 2015; 4, 164-168.
A Alvarez, R Alarcon, C Opazo, EO Campos, FJ Munoz, FH Calderon, F Dajas, MK Gentry, BP Doctor, FGD Mello and NC Inestrosa. Stable complexes involving acetylcholinesterase and amyloid-β peptide change the biochemical properties of the enzyme and increase the neurotoxicity of Alzheimer’s fibrils. The Journal of Neuroscience 1998; 18(9), 3213-3223.
P Paixão, LF Gouveia and JAG Morais. Prediction of the human oral bioavailability by using in vitro and in silico drug related parameters in a physiologically based absorption model. International Journal of Pharmaceutics 2012; 429(1-2), 84-98.
GD Stanciu, A Luca, RN Rusu, V Bild, SI Beschea Chiriac, C Solcan, W Bild and DC Ababei. Alzheimer’s disease pharmacotherapy in relation to cholinergic system involvement. Biomolecules 2019; 10(1), 40.
CA Lipinski. Lead- and drug-like compounds: The rule-of-5 revolution. Drug Discovery Today: Technologies 2004; 1(4), 337-341.
SK Bhal. LogP-making sense of the value. Tropical Journal of Natural Product Research 2007; 4(12), 1208-1214.
SM Alwan. Computational calculations of molecular properties and molecular docking of new and reference cephalosporins on penicillin binding proteins and various β-lactamases. Journal of Pharmacy and Pharmacology 2016; 4(5), 212-225.
F Li, ZM Wang, JJ Wu, J Wang, SS Xie, JS Lan, W Xu, LY Kong and XB Wang. Synthesis and pharmacological evaluation of donepezil-based agents as new cholinesterase/monoamine oxidase inhibitors for the potential application against Alzheimer’s disease. Journal of Enzyme Inhibition and Medicinal Chemistry 2016; 31, 41-53.
D Andrianto, S Hermita and S Haryanti. The classification of betel leaves (Piper betle) from 15 ethnics in eastern Indonesia based on phytochemicals fingerprint analysis. Biodiversitas Journal of Biological Diversity 2019; 21(1), 252-257.
EW Purnama, M Safithri and D Andrianto. Clusterization of red betel leaves (Piper Crocatum) from various regions in Indonesia based on secondary metabolite fingerprint analysis and cytotoxicity values. Indonesian Journal of Applied Research 2023; 4(2), 170-182.
F Kurniawan. Review of geographic factors support mapping in Yogyakarta related to food security conditions on the physiological quality of talented athletes. Advances in Health Sciences Research 2022; 43, 289-294.
R Ismail and A Ismail. Spatial analysis on influence of elevation to coronavirus disease-19 infection fatality ratio in Bandung Regency and Bandung City. IOP Conference Series: Earth and Environmental Science 2024; 1404(1), 012050.
R Jannah, H Helmi and S Sufardi. Soil physical and chemical properties in Patcholi planting area in Teungoh Geunteut Village, Kecamatan lhoong, Aceh Besar. Jurnal Ilmiah Mahasiswa Pertanian 2022; 7(3), 431-439.
W Abd Karim, A Farajallah and B Suryobroto. Exploration and prevalence of gastrointestinal worm in buffalo from West Java, Central Java, East Java and Lombok, Indonesia. Aceh Journal of Animal Science 2016; 1(1), 1-15.
S Shukla, N Kumar, P Bhardwaj, P Pandita, MK Patel, MS Thakur, R Kumar, M Rawat and S Saxena. Effect of cold arid high-altitude environment on bioactive phytochemical compounds of organically grown Brassicaceae vegetables for nutri-health security in mountainous regions. Scientific Reports 2024; 14(1), 15976.
AS Huda, AEZ Hasan and M Safithri. Acetylcholinesterase enzyme inhibitor and antioxidant activities from a mixture extracts of black tea, red betel, Cinnamon and Curcuma. Current Biochemistry 2023; 9(2), 63-72.
B Kharel, L Rusalepp, B Bhattarai, A Kaasik, P Kupper, R Lutter, P Mänd, G Rohula-Okunev, K Rosenvald and A Tullus. Effects of air humidity and soil moisture on secondary metabolites in the leaves and roots of Betula pendula of different competitive status. Oecologia 2023; 202(2), 193-210.
L Gali and F Bedjou. Antioxidant and anticholinesterase effects of the ethanol extract, ethanol extract fractions and total alkaloids from the cultivated Ruta chalepensis. South African Journal of Botany 2019; 120, 163-169.
A Plaskova and J Mlcek. New insights of the application of water or ethanol-water plant extract rich in active compounds in food. Frontiers in Nutrition 2023; 10, 1118761.
H Khan, S Amin, MA Kamal and S Patel. Flavonoids as acetylcholinesterase inhibitors: Current therapeutic standing and future prospects. Biomedicine & Pharmacotherapy 2018; 101, 860-870.
RR Elfirta, PR Ferdian, I Saskiawan, TH Handayani, KFG Mandalika, R Riffiani, K Kasirah and UMS Purwanto. Antioxidant properties of kombucha beverage infused with Ganoderma lucidum and green tea from Camellia sinensis (L.) Kuntze with several fermentation times. Karbala International Journal of Modern Science 2024; 10(1), 12.
N Marliani, IM Artika, M Rafi, M Syukur, RY Galingging and W Nurcholis. Phenolic and flavonoid content with agro morphological characters of 12 accessions of Justica gendarussa grew in Indonesia. Biodiversitas Journal of Biological Diversity 2022; 23(10), 5101-5107.
JE Lee, JTM Jayakody, JI Kim, JW Jeong, KM Choi, TS Kim, C Seo, I Azimi, J Hyun and B Ryu. The influence of solvent choice on the extraction of bioactive compounds from Asteraceae: A comparative review. Foods 2024; 13(19), 3151.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






