Impact of Simulated pH Conditions on Phenotypic Expression in Shrimp Pathogenic and Non-Pathogenic Vibrio campbellii Strains
DOI:
https://doi.org/10.48048/tis.2025.9403Keywords:
Aquaculture, Aquaculture management, Biofilm formation, Climate change, Luminous vibriosis, Ocean acidification, pH fluctuation, Shrimp pathogen, Vibrio campbelliiAbstract
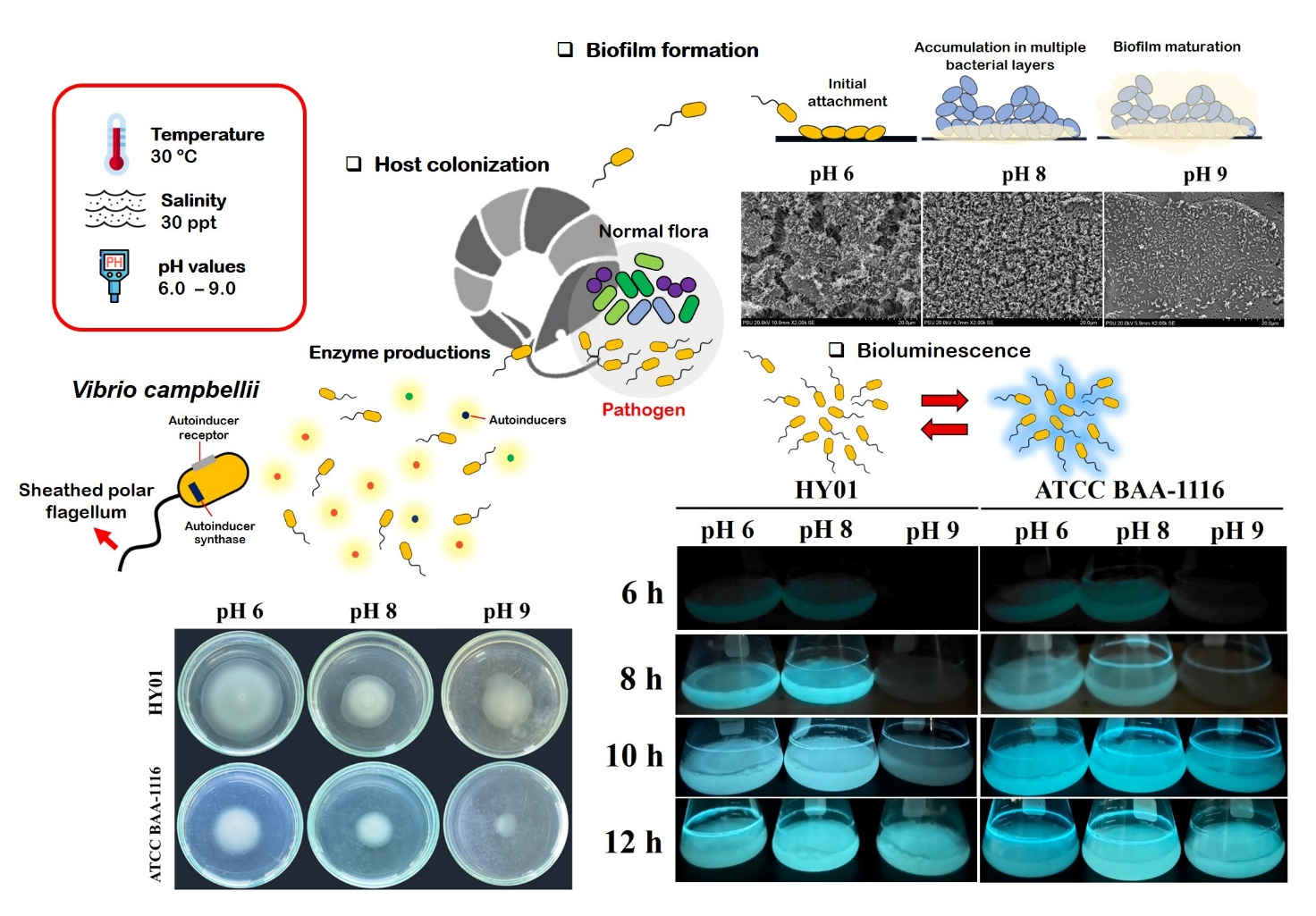
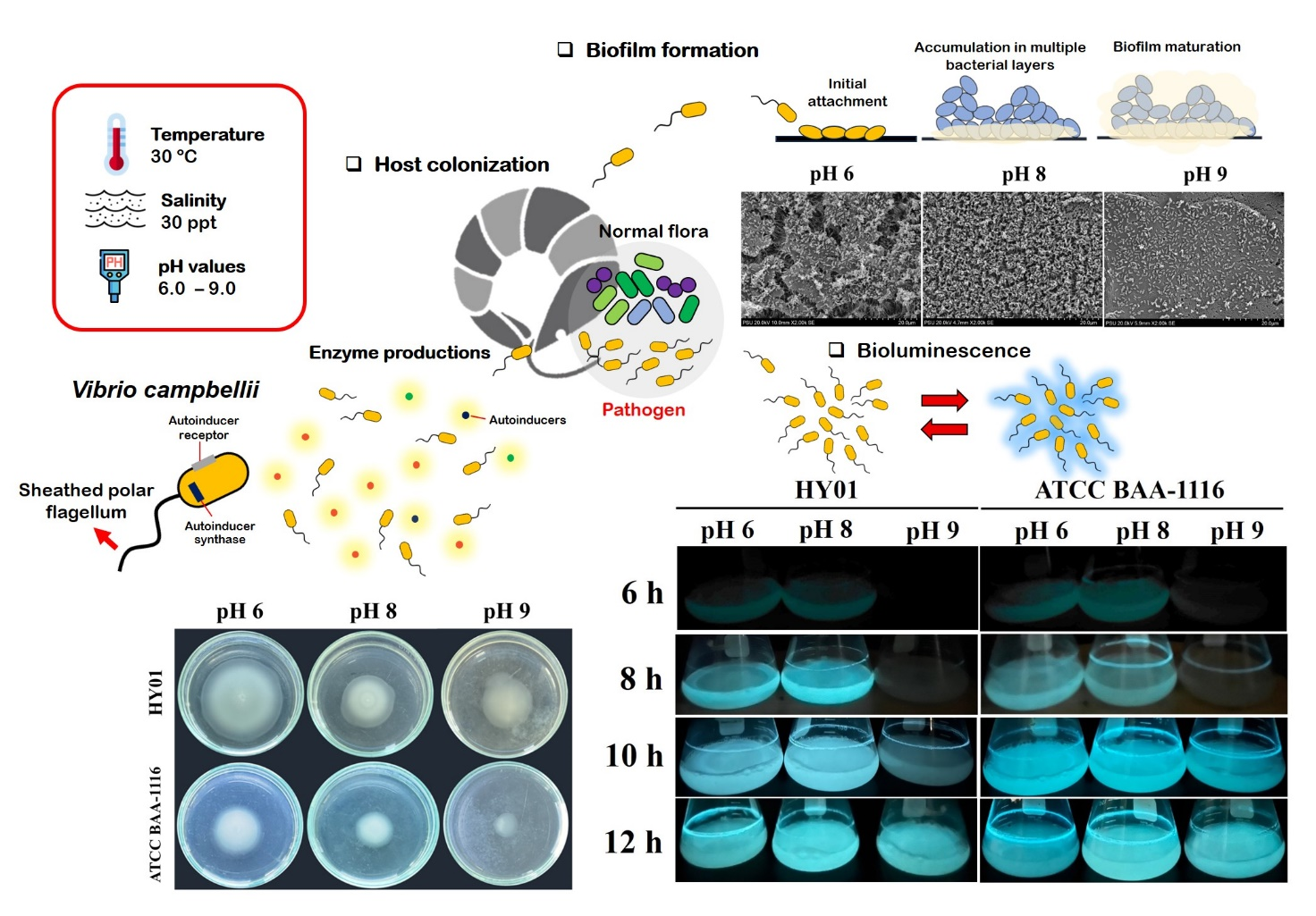
Environmental pH fluctuation in oceanic and marine ecosystems can significantly impact the distribution and behavior of pathogenic Vibrio species, including their interactions with marine invertebrates such as crustaceans. This study focused on Vibrio campbellii, a common shrimp pathogen, and its phenotypic responses to varying pH conditions. Both pathogenic strain HY01 and non-pathogenic strain ATCC BAA-1116 were cultured in 30 pL/L Luria-Bertani Sea Salt under 3 pH conditions, including pH 6 (slightly acidic), pH 8 (representing the oceanic pH), and pH 9 (alkaline). Growth patterns and phenotypic traits were evaluated. Results revealed no significant growth difference between the 2 strains under the different pH conditions, although the non-pathogenic strain showed a slight growth reduction at pH 9 during the exponential phase. Both strains were able to buffer environmental pH shifts, adjusting to near-oceanic pH levels (around pH 8). At pH 9, a stressor level for V. campbellii, delays were observed in bioluminescence, biofilm formation, exopolysaccharide production, shrimp surface colonization, motility, and caseinase production, affecting both strains. In contrast, mildly acidic conditions (pH 6) induced the highest expression of several phenotype traits. Statistical analyses indicated significant interactions between strain type and pH levels in influencing phenotypic expression. In conclusion, the pathogenic V. campbellii strain HY01 exhibited greater adaptability and virulence across various pH conditions compared to the non-pathogenic ATCC BAA-1116, emphasizing pH as a critical environmental factor in shaping the growth and pathogenic potential of V. campbellii. Our studies provide valuable insights into managing pH conditions in aquaculture environments to optimize proper shrimp cultivation and prevent cross-contamination of V. campbellii from seawater habitats to farms. These findings provide a physiological profile of Vibrio under pH stress, which can support the development of predictive outbreak models to assess the risk of luminous vibriosis, especially in to seasonal changes and ocean acidification.
HIGHLIGHTS
- The shrimp pathogenic Vibrio campbellii strain HY01 demonstrated stronger survival and higher virulence compared to the non-pathogenic campbellii strain ATCC BAA-1116 under both acidic and basic pH conditions.
- Both pathogenic and non-pathogenic campbellii strains adjusted the initial pH of their cultures to more optimal levels for growth and phenotypic expression.
- Slightly acid to slightly alkaline conditions supported favorable environments, enhancing growth, bioluminescence, biofilm formation, exopolysaccharide production, shrimp colonization, swimming motility, and protease production in both campbellii strains.
- Extreme alkalinity was identified as a pH stressor for campbellii strains, suppressing phenotypic expressions without inhibiting growth.
- These findings provide detailed phenotypic profiles of pathogenic and non-pathogenic campbellii strains in response to pH fluctuations during their growth stages.
GRAPHICAL ABSTRACT
Downloads
References
KD Brumfield, M Usmani, KM Chen, M Gangwar, AS Jutla, A Huq and RR Colwell. Environmental parameters associated with incidence and transmission of pathogenic Vibrio spp. Environmental Microbiology 2021; 23(12), 7314-7340.
LQ Jiang, J Dunne, BR Carter, JF Tjiputra, J Terhaar, JD Sharp, A Olsen, S Alin, DCE Bakker, RA Feely, JP Gattuso, P Hogan, T Ilyina, N Lange, SK Lauvset, ER Lewis, T Lovato, J Palmieri, Y Santana-Falcón, J Schwinger, …, T Ziehn. Global surface ocean acidification indicators from 1750 to 2100. Journal of Advances in Modeling Earth Systems 2023; 15(3), e2022MS003563.
RK Mohanty, A Kumar, A Mishra, DK Panda and DU Patil. Water budgeting and management: Enhancing aquacultural water productivity. Directorate of Water Management, Indian Council of Agricultural Research. Chandrasekharpur, Bhubaneswar, India, 2014.
FM Yusoff, W Umi, NM Ramli and R Harun. Water quality management in aquaculture. Cambridge prisms: Water 2024; 2, e8.
KEC Velez, RE Leighton, AW Decho, JL Pinckney and RS Norman. Modeling pH and temperature effects as climatic hazards in Vibrio vulnificus and Vibrio parahaemolyticus planktonic growth and biofilm formation. GeoHealth 2023; 7(4), e20022GH000769.
A Singh and TG Barnard. Surviving the acid barrier: Responses of pathogenic Vibrio cholerae to simulated gastric fluid. Applied Microbiol and Biotechnology 2016; 100(2), 815-824.
JH Yoon, YM Bae and SY Lee. Effects of varying concentrations of sodium chloride and acidic conditions on the behavior of Vibrio parahaemolyticus and Vibrio vulnificus cold-starved in artificial sea water microcosms. Food Science and Biotechnology 2017; 26(3), 829-839.
S Çam and R Brinkmeyer. The effects of temperature, pH, and iron on biofilm formation by clinical versus environmental strains of Vibrio vulnificus. Folia Microbiologica 2020; 65(3), 557-566.
KD Brumfield, AJ Chen, M Gangwar, M Usmani, NA Hasan, AS Jutla, A Huq and RR Colwell. Environmental factors influencing occurrence of Vibrio parahaemolyticus and Vibrio vulnificus. Applied and Environmental Microbiology 2023; 89(6), e0030723.
BL Pipes and MK Nishiguchi. Nocturnal acidification: A coordinating cue in the Euprymna scolopes-Vibrio fischeri symbiosis. International Journal of Molecular Sciences 2022; 23(7), 3743.
L Wang, Y Chen, H Huang, Z Huang, H Chen and Z Shao. Isolation and identification of Vibrio campbellii as a bacterial pathogen for luminous vibriosis of Litopenaeus vannamei. Aquaculture Research 2015; 46(2), 395-404.
S Kumar, CB Kumar, V Rajendran, N Abishaw, PSS Anand, S Kannapan, VK Nagaleekar, KK Vijayan and SV Alavandi. Delineating virulence of Vibrio campbellii: A predominant luminescent bacterial pathogen in Indian shrimp hatcheries. Scientific Reports 2021; 11, 15831.
P Rattanama, K Srinitiwarawong, JR Thompson, R Pomwised, K Supamattaya and V Vuddhakul. Shrimp pathogenicity, hemolysis, and the presence of hemolysin and TTSS genes in Vibrio harveyi isolated from Thailand. Diseases of Aquatic Organisms 2009; 86(2), 113-122.
B Lin, Z Wang, AP Malanoski, EA O’Grady, CF Wimpee, V Vuddhakul, NJ Alves, FL Thompson, B Gomez-Gil and GJ Vora. Comparative genomic analyses identify the Vibrio harveyi genome sequenced strains BAA-1116 and HY01 as Vibrio campbellii. Environmental Microbiology Reports 2010; 2(1), 81-89.
Ž Pavlinec, IG Zupičić, D Oraić, I Lojkić, B Fouz and S Zrnčić. Biochemical and molecular characterization of three serologically different Vibrio harveyi strains isolated from farmed Dicentrarchus labrax from the Adriatic Sea. Scientific Reports 2022; 12(1), 7309.
N Orel, E Fadeev, GJ Herndl, V Turk and T Tinta. Recovering high-quality bacterial genomes from cross-contaminated cultures: A case study of marine Vibrio campbellii. BMC Genomics 2024; 25(1), 146.
J Schwarz, S Brameyer, E Hoyer and K Jung. The interplay of AphB and CadC to activate acid resistance of Vibrio campbellii. Journal of Bacteriology 2023; 205(4), e0045722.
P Kissalai, S Preeprem, V Vuddhakul and P Mittraparp-Arthorn. Phylogenetic analysis of atypical hemolysin gene in Vibrio campbellii and effects of cultivation salinity and pH on hemolytic activity and virulence. Walailak Journal of Science and Technology 2018; 17(4), 324-333.
YY Chen, JC Chen, KC Tseng, YC Lin and CL Huang. Activation of immunity, immune response, antioxidant ability, and resistance against Vibrio alginolyticus in white shrimp Litopenaeus vannamei decrease under long-term culture at low pH. Fish & Shellfish Immunology 2015; 46(2), 192-199.
T Muralisankar, P Kalaivani, SH Thangal and P Santhanam. Growth, biochemical, antioxidants, metabolic enzymes and hemocytes population of the shrimp Litopenaeus vannamei exposed to acidified seawater. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2021; 239, 108843.
BL Bassler, EP Greenberg and AM Stevens. Cross-species induction of luminescence in the quorum-sensing bacterium Vibrio harveyi. Journal of Bacteriology 1997; 179(12), 4043-4045.
A Parker. Pattern recognition of ocean pH. Nonlinear Engineering 2016; 5(3), 205-217.
G Balakrishnan, S Peyail, K Ramachandran, A Theivasigamani, KA Savji, M Chokkaiah and P Nataraj. Growth of cultured white leg shrimp Litopenaeus Vannamei (Boone 1931) in different stocking density. Advances in Applied Science Research 2011; 2(3), 107-113.
Supriatna, A Darmawan and A Maizar. Pathway analysis of pH in whiteleg shrimp, Litopenaeus vannamei concrete pond intensifies in Banyuwangi East Java. IOP Conference Series: Earth and Environmental Science 2023; 1191, 12015.
C Nithya and SK Pandian. The in vitro antibiofilm activity of selected marine bacterial culture supernatants against Vibrio spp. Archives of Microbiology 2010; 192(10), 843-854.
C Kirchhoff and H Cypionka. Propidium ion enters viable cells with high membrane potential during live-dead staining. Journal of Microbiological Methods 2017; 142, 79-82.
J Liu, K Fu, Y Wang, C Wu, F Li, L Shi, Y Ge and L Zhou. Detection of diverse n-acyl-homoserine lactones in Vibrio alginolyticus and regulation of biofilm formation by N-(3-Oxodecanoyl) homoserine lactone in vitro. Frontiers in Microbiology 2017; 8, 1097.
N Han, MFR Mizan, IK Jahid and SD Ha. Biofilm formation by Vibrio parahaemolyticus on food and food contact surfaces increases with rise in temperature. Food Control 2016; 70, 161-166.
S Santhakumari, R Jayakumar, R Logalakshmi, NM Prabhu, AKA Nazar, SK Pandian and AV Ravi. In vitro and in vivo effect of 2,6-Di-tert-butyl-4-methylphenol as an antibiofilm agent against quorum sensing mediated biofilm formation of Vibrio spp. Internatioanl Journal of Food Microbiology 2018; 281, 60-71.
B Han, X Zheng, K Baruah and P Bossier. Sodium ascorbate as a quorum-sensing inhibitor leads to decreased virulence in Vibrio campbellii. Frontiers in Microbiology 2020; 11, 1054.
SB Prayitno and JW Latchford. Experimental infections of crustaceans with luminous bacteria related to Photobacterium and Vibrio. effect of salinity and pH on infectiosity. Aquaculture 1995; 132(1-2), 105-112.
K Gundogdu, AO Iturriza, M Orruño, I Montánchez, H Eguiraun, I Martinez, I Arana and VR Kaberdin. Addressing the joint impact of temperature and pH on Vibrio harveyi adaptation in the time of climate change. Microorganisms 2023; 11(4), 1075.
K Nozaki, T Kuroda, T Mizushima and T Tsuchiya. A new Na+/H+ antiporter, NhaD, of Vibrio parahaemolyticus. Biochimica et Biophysica Acta 1998; 1362(2), 213-220.
K Herz, S Vimont, E Padan and P Berche. Roles of NhaA, NhaB, and NhaD Na+/H+ antiporters in survival of Vibrio cholerae in a saline environment. Journal of Bacteriology 2003; 185(4), 1236-1244.
E Padan, E Bibi, M Ito and TA Krulwich. Alkaline pH homeostasis in bacteria: New insights. Biochimica et Biophysica Acta 2005; 1717(2), 67-88.
A Rahmani, C Mathien, A Bidault, NL Goïc, C Paillard and V Pichereau. External pH modulation during the growth of Vibrio tapetis, the aetiological agent of brown ring disease. Journal of Applied Microbiology 2020; 129(1), 3-16.
B Kostiuk, ME Becker, CN Churaman, JJ Black, SM Payne, S Pukatzki and BJ Koestler. Vibrio cholerae alkalizes its environment via citrate metabolism to inhibit enteric growth in vitro. Microbiology Spectrum 2023; 11(2), e0491722.
J Schwarz, K Schumacher, S Brameyer and K Jung. Bacterial battle against acidity. FEMS Microbiology Reviews 2022; 46(6), fuac037.
RE Showalter, MO Martin and MR Silverman. Cloning and nucleotide sequence of luxR, a regulatory gene controlling bioluminescence in Vibrio harveyi. Journal of Bacteriology 1990; 172(6), 2946-2954.
SY Kim, SE Lee, YR Kim, CM Kim, PY Ryu, HE Choy, SS Chung and JH Rhee. Regulation of Vibrio vulnificus virulence by the LuxS quorum-sensing system. Molecular Microbiology 2003; 48(6), 1647-1664.
S Yeo, H Park, Y Ji, S Park, J Yang, J Lee, JM Mathara, H Shin and W Holzapfel. Influence of gastrointestinal stress on autoinducer-2 activity of two Lactobacillus species. FEMS Microbiology Ecology 2015; 91(7), fiv065.
Y Gu, B Li, J Tian, R Wu and Y He. The response of LuxS/AI-2 quorum sensing in Lactobacillus fermentum 2-1 to changes in environmental growth conditions. Annals of Microbiology 2018; 68, 287-294.
VV Kuts and AD Ismailov. Physiological and emission characteristics of the luminescent bacterium Photobacterium Phosphoreum from the White Sea. Microbiology 2009; 78, 554-558.
LA Vitukhnovskaya and AD Ismailov. Effect of Na+ and K+ ions on the luminescence of intact Vibrio harveyi cells at different pH values. Microbiology 2001; 70(4), 525-530.
R Calogero, C Rizzo, E Arcadi, MG Stipa, P Consoli, T Romeo and P Battaglia. Isolation and identification of luminescent bacteria in deep sea marine organisms from Sicilian Waters (Mediterranean Sea). Journal of Marine Science and Engineering 2022; 10(8), 1113.
S Çam and R Brinkmeyer. The effects of temperature, pH, and iron on biofilm formation by clinical versus environmental strains of Vibrio vulnificus. Folia Microbiologica 2020; 65(3), 557-566.
G Luo, L Huang, Y Su, Y Qin, X Xu, L Zhao and Q Yan. flrA, flrB and flrC regulate adhesion by controlling the expression of critical virulence genes in Vibrio alginolyticus. Emerging Microbes & Infections 2016; 5(8), e85.
X Xu, H Li, X Qi, Y Chen, Y Qin, J Zheng and X Jiang. cheA, cheB, cheR, cheV, and cheY are involved in regulating the adhesion of Vibrio harveyi. Frontiers in Cellular and Infection Microbiology 2021; 10, 591751.
D Gu, K Wang, T Lu, L Li and X Jiao. Vibrio parahaemolyticus CadC regulates acid tolerance response to enhance bacterial motility and cytotoxicity. Journal of Fish Diseases 2021; 44(8), 1155-1168.
VN Tran, PNV Nguyen and TTH Nguyen. Effects of pH, temperature and oxygen-limited condition on the virulence of Vibrio parahaemolyticus. Science and Technology Development Journal 2020; 23(3), 574-580.
M Venugopal and AV Saramma. Characterization of alkaline protease from Vibrio fluvialis strain VM10 isolated from a mangrove sediment sample and its application as a laundry detergent additive. Process Biochemistry 2006; 41(6), 1239-1243.
M Kozhikotte, J Puthumana, D Jose, B Sreelakshmi, P Gopinath and SAVB Singh. Alkaline protease from a non-toxigenic mangrove isolate of Vibrio sp. V26 with potential application in animal cell culture. Cytotechnology 2013; 65(2), 199-212.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






