Deciphering Depression-Targeted Mechanisms of Pandanus odorifer Leaf Extracts Based on Component Analysis and Integrated Molecular Approaches
DOI:
https://doi.org/10.48048/tis.2025.9320Keywords:
Network pharmacology, In silico, Depression, Molecular dynamics, Pandanus odoriferAbstract
Introduction: Mental health is a worldwide health problem. Antidepressants have therapeutic effects but after prolonged use, they can cause unwanted side effects. Seeing the great potential of coastal plants such as sea pandanus (Pandanus odorifer) attracts attention in creating a more effective and safe treatment. The aim of this study is to identify the compounds contained in sea pandanus and elucidate the molecular mechanism in the target proteins of depressive diseases. Materials and methods: LC-MS has been used to identify compounds in sea pandanus. The network pharmacology approach used Cytoscape 3.9.1, OMIM, Disgenet, SEA, SwissTarget, STRING 2.0.0, Metascape, and Kyoto Encyclopedia of Genes and Genomes (KEGG) to predict the molecular antidepressant mechanism of the metabolite compounds. Validation was achieved using MOE with MAOA receptor (PDB ID: 2Y5Z). Molecular dynamics simulation using Maestro Schrödinger 2020-1 software. Results and discussion: A total of 19 active compounds were identified through LC-MS. Network pharmacology analysis showed that the MAOA target was the most influential protein target in the network pharmacology study of antidepressants from metabolite compounds of sea pandanus methanol extract. The results of in silico validation and molecular dynamics simulation support the network pharmacology findings. Benzylcarbonyl-Lys-Dab-Arg-NH2 compound showed higher affinity to MAOA receptor target compared to standard drug (fluoxetine) with S score −10.7455 kcal/mol and MMGBSA value −68.0669 kcal/mol. Conclusions: The compound Benzylcarbonyl-Lys-Dab-Arg-NH2 showed higher binding affinity towards MAOA compared to the native ligand and drug fluoxetine, also maintaining a s2interaction within the MAOA active site, indicating that Pandanus odorifer has significant promise as a novel antidepressant agent. These findings provide a strong basis for further investigation into its efficacy and safety in a clinical setting, potentially leading to the development of more effective and targeted treatments for depression.
HIGHLIGHTS
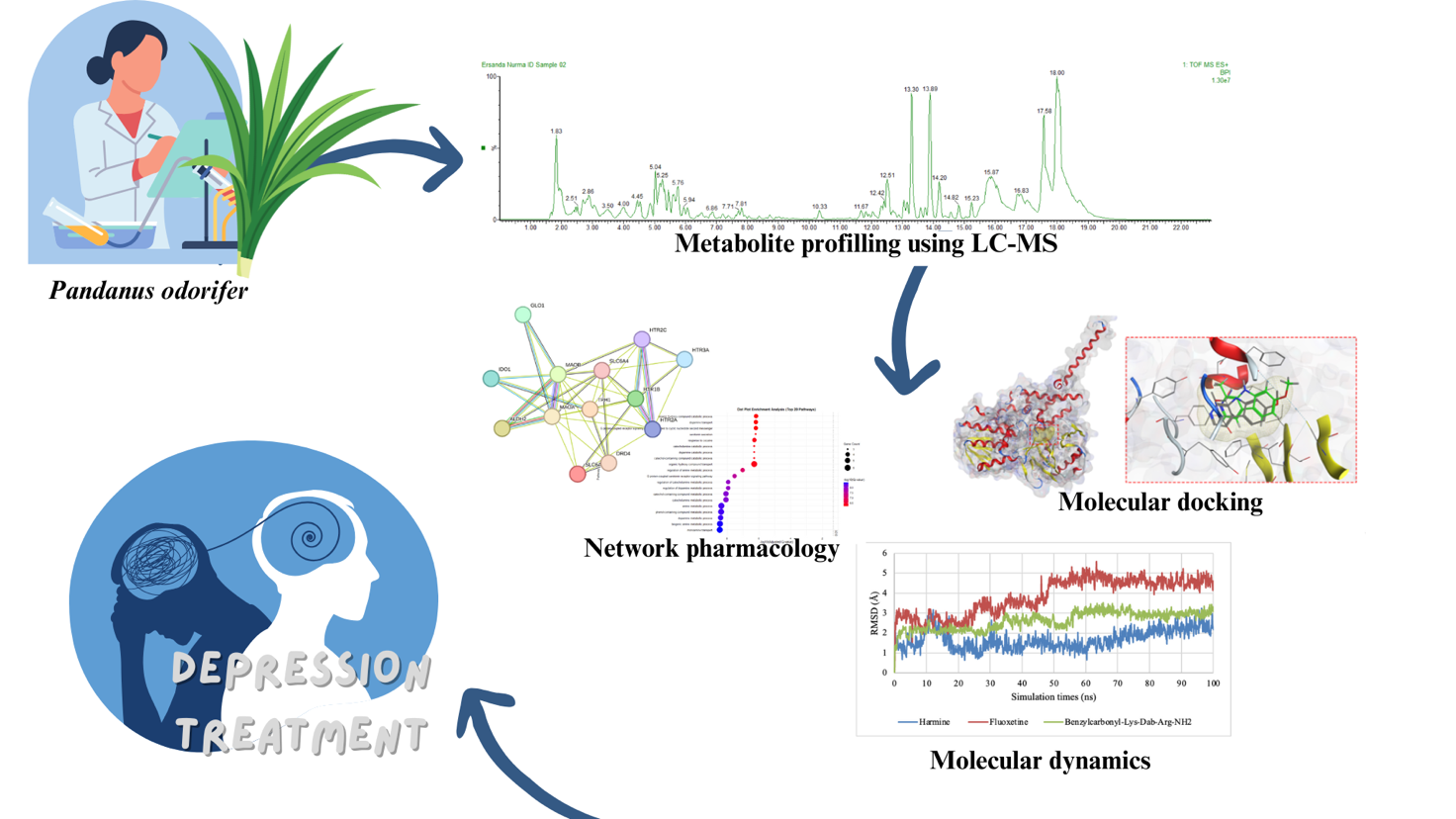
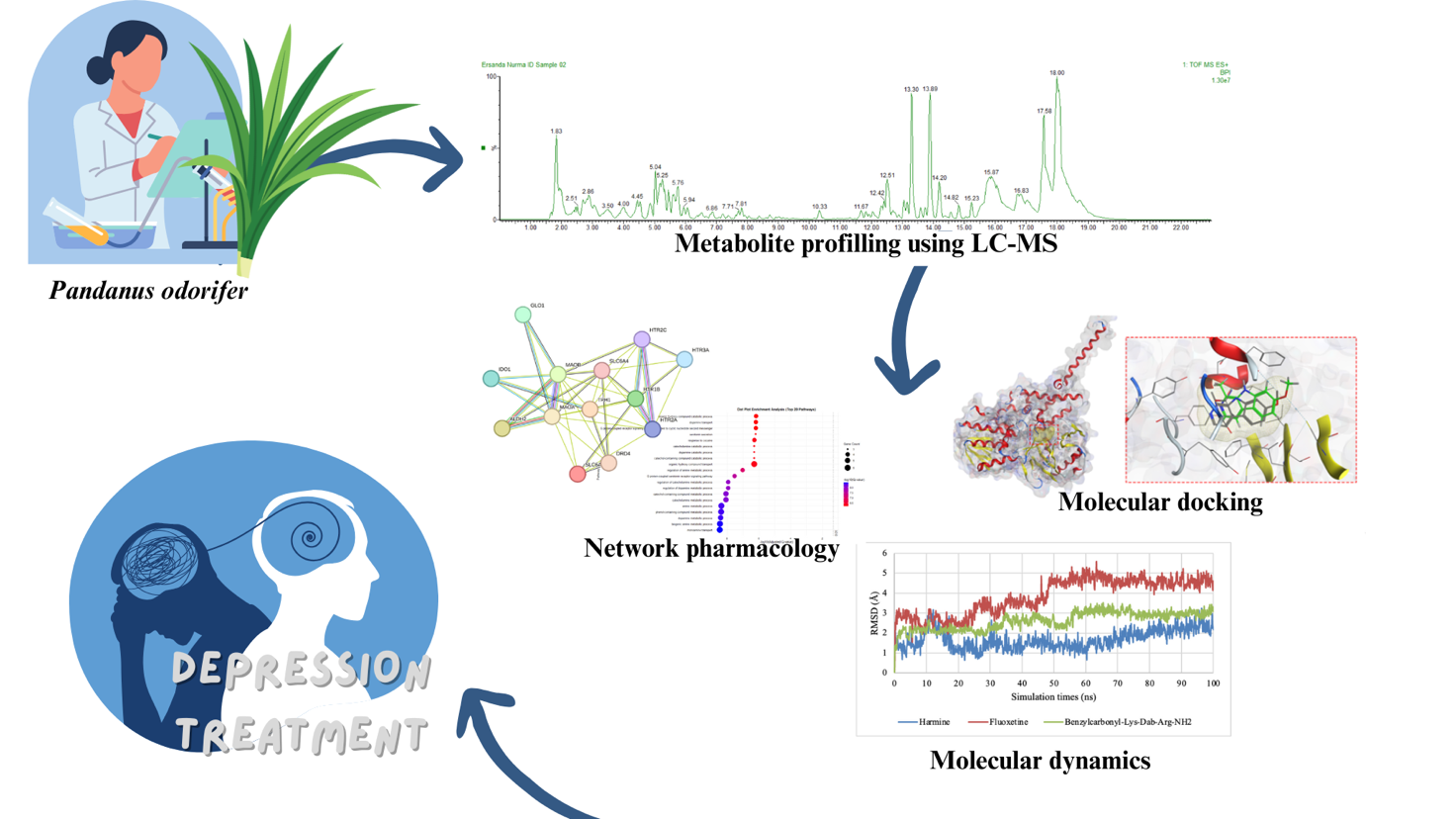
- This article focused on the exploration of Pandanus odorifer leaves as a potential source of bioactive compounds for depression therapy.
- Pandanus odorifer were profiled using LC-MS (Liquid Chromatography-Mass Spectrometry) technique to identify metabolite compounds.
- Computational-based approaches were conducted using network pharmacology, molecular docking, and molecular dynamics.
- Network pharmacology was used to understand the interaction of bioactive compounds with molecular targets related to depression.
- Molecular docking was done to evaluate the potential binding of the compound to depression-related protein targets.
- Molecular dynamics was used to analyze the stability of compound interactions with protein targets.
GRAPHICAL ABSTRACT
Downloads
References
SC Parija. World suicide prevention day: Creating hope through action. Pondicherry Journal of Nursing 2022; 15(3), 50.
Kementerian Kesehatan Republik Indonesia. Topik kesehatan depresi (in Indonesian). Kementerian Kesehatan Republik Indonesia, Jakarta, Indonesia, 2019.
L Puspitasari. Ekstrak etanol daun pandan wangi (Pandanus amaryllifolius R.) 10 % menurunkan immobility time dan kadar kortisol tikus jantan galur wistar yang depresi (in Indonesian). Intisari Sains Medis 2017; 8(1), 24-30.
FP Bymaster, W Zhang, PA Carter, J Shaw, E Chernet, L Phebus, DT Wong and KW Perry. Fluoxetine, but not other selective serotonin uptake inhibitors, increases norepinephrine and dopamine extracellular levels in prefrontal cortex. Psychopharmacology 2002; 160, 353-361.
Z Ungvari, S Tarantini, A Yabluchanskiy and A Csiszar. Potential adverse cardiovascular effects of treatment with fluoxetine and other selective serotonin reuptake inhibitors (SSRIs) in patients with geriatric depression: Implications for atherogenesis and cerebromicrovascular dysregulation. Frontiers in Genetics 2019; 10, 455576.
SP Rahma and D Suzana. Uji aktivitas antidepresan minyak atsiri daun kemangi (Ocimum basilicum L.) dengan metode open field test (OFT) (in Indonesian). Jurnal Farmasi dan Farmakoinformatika 2024; 2(1), 9-22.
I Rochmadi and S Rohmah. Pemanfaatan buah pandan laut sebagai pangan olahan pada masyarakat pesisir (in Indonesian). Jurnal Riset Ekonomi Pembangunan 2019; 4(2), 161-173.
R Londonkar and A Kamble. Evaluation of free radical scavenging activity of Pandanus odoratissimus. International Journal of Pharmacology 2009; 5(6), 377-380.
S Venkatesh, R Kusuma, V Sateesh, MB Reddy and R Mullangi. Antidiabetic activity of Pandanus odoratissimus root extract. Indian Journal of Pharmaceutical Education and Research 2012; 46(4), 340-345.
TT Jong and SW Chau. Antioxidative activities of constituents isolated from Pandanus odoratissimus. Phytochemistry 1998; 49(7), 2145-2148.
S Raju, NV Subbaiah, KS Reddy, A Das and KB Murugan. Potential of Pandanus odoratissimus as a CNS depressant in Swiss albino mice. Brazilian Journal of Pharmaceutical Sciences 2011; 47, 629-634.
RL Ramesh Londonkar, AK Abhaykumar Kamble and VC Reddy. Anti-inflammatory activity of Pandanus odoratissimus extract. International Journal of Pharmacology 2010; 6(3), 311-314.
A El-Shaibany, ALH Molham, S Al-Massarani, Ali Al-Ajami and A Al-Adhl. Hepatoprotective effect of Pandanus odoratissimus l inflorescence extracts in acetaminophen-treated guinea pigs. Tropical Journal of Pharmaceutical Research 2016; 15(2), 259-267.
G Mishra, RL Khosa, P Singh and KK Jha. Hepatoprotective potential of ethanolic extract of Pandanus odoratissimus root against paracetamol-induced hepatotoxicity in rats. Journal of Pharmacy and Bioallied Sciences 2015; 7(1), 45-48.
R Bahramsoltani, MH Farzaei, MS Farahani and R Rahimi. Phytochemical constituents as future antidepressants: A comprehensive review. Reviews in the Neurosciences 2015; 26(6), 699-719.
JJ Fortunato, GZ Réus, TR Kirsch, RB Stringari, GR Fries, F Kapczinski, JE Hallak, AW Zuardi, JA Crippa and J Quevedo. Effects of β-carboline harmine on behavioral and physiological parameters observed in the chronic mild stress model: Further evidence of antidepressant properties. Brain Research Bulletin 2010; 81(4-5), 491-496.
QQ Mao, YF Xian, SP Ip and CT Che. Involvement of serotonergic system in the antidepressant-like effect of piperine. Progress in Neuro-Psychopharmacology and Biological Psychiatry 2011; 35(4), 1144.
SS Mardolkar and MP Joshi. Pharmacological evaluation of Pandanus odorifer (Forssk.) Kuntze adminstered orally and by inhalation route in rats. International Journal of Current Science 2022; 12(4), 311-318.
SV Sathasivampillai, V Shanmugalingam, PR Sebastian. Pharmacological activities of extracts and isolated phytochemical constituents of Pandanus odorifer (Forssk.) Kuntze. Gümüşhane Üniversitesi Sağlık Bilimleri Dergisi. 2021, 10(3), 574-80.
T Son. Secondary metabolites of genus Pandanus: an aspect of phytochemistry. Mini-Reviews in Organic Chemistry 2019, 16(7), 689-710.
C Jia, X Pan, B Wang, P Wang, Y Wang and R Chen. Mechanism prediction of Astragalus membranaceus against cisplatin-induced kidney damage by network pharmacology and molecular docking. Evidence-Based Complementary and Alternative Medicine 2021; 2021(1), 9516726.
P Zhou, R Zhou, Y Min, LP An, F Wang and QY Du. Network pharmacology and molecular docking analysis on pharmacological mechanisms of Astragalus membranaceus in the treatment of gastric ulcer. Evidence-Based Complementary and Alternative Medicine 2022; 2022(1), 9007396.
F Li, J Duan, M Zhao, S Huang, F Mu, J Su, K Liu, Y Pan, X Lu, J Li, P Wei, M Xi and A Wen. A network pharmacology approach to reveal the protective mechanism of Salvia miltiorrhiza-Dalbergia odorifera coupled-herbs on coronary heart disease. Scientific Reports 2019; 9(1), 19343.
EY Chen, CM Tan, Y Kou, Q Duan, Z Wang, GV Meirelles, NR Clark and A Ma’ayan. Enrichr: Interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinformatics 2013; 14(1), 128.
Y Liao, J Wang, EJ Jaehnig, Z Shi and B Zhang. WebGestalt 2019: Gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Research 2019; 47(W1), W199-W205.
O Mutlu, G Ulak, IK Celikyurt, FY Akar and F Erden. New approaches for the therapy of treatment refractory depression. In: G Rossi (Ed.). Psychology-selected papers. IntechOpen, London, 2012.
JR Leheste, C Curcio, L Baldinger, S Sarwar, SM Zakhary, BH Hallas, JM Horowitz and G Torres. Glutamate-based drugs for the treatment of clinical depression. Bentham Science Publishers 2008; 8(3), 170-176.
R Kaddurah-Daouk, P Yuan, SH Boyle, W Matson, Z Wang, ZB Zeng, H Zhu, GG Dougherty, J Yao, G Chen, X Guitart, PJ Carlson, A Neumeister, CA Zarate, R Krishnan, HK Manji and WC Drevets. Cerebrospinal fluid metabolome in mood disorders-remission state has a unique metabolic profile. Nature Portfolio 2012; 2(1), 667.
DM Diniz, K Bosch, F Calabrese, P Brivio, MA Riva, J Grandjean and JR Homberg. BDNF overexpression in the prelimbic cortex does not reduce anxiety- and depression-like behavior in serotonin knockout rats. Cold Spring Harbor Laboratory 2020. https://doi.org/10.1101/2020.07.01.180604
A Kaltenboeck and C Harmer. The neuroscience of depressive disorders: A brief review of the past and some considerations about the future. Brain and Neuroscience Advances 2018; 2, 1-6.
L Chiuccariello, S Houle, L Miler, RG Cooke, PM Rusjan, G Rajkowska, RD Levitan, SJ Kish, NJ Kolla, X Ou, AA Wilson and JH Meyer. Elevated monoamine oxidase a binding during major depressive episodes is associated with greater severity and reversed neurovegetative symptoms. Neuropsychopharmacology 2013; 39(4), 973-980.
G Hasler. Pathophysiology of depression: Do we have any solid evidence of interest to clinicians? World Psychiatry 2010; 9(3), 155-161.
S Chaki, Y Ago, A Palucha-Paniewiera, F Matrisciano and A Pilc. mGlu2/3 and mGlu5 receptors: Potential targets for novel antidepressants. Neuropharmacology 2013; 66, 40-52.
J Cookson. Side-effects of antidepressants. The British Journal of Psychiatry 1993; 163(S20), 20-24.
SH Lin, LT Lee and YK Yang. Serotonin and mental disorders: A concise review on molecular neuroimaging evidence. Clinical Psychopharmacology and Neuroscience 2014; 12(3), 196-202.
R Marotta, MC Risoleo, G Messina, L Parisi, M Carotenuto, L Vetri and M Roccella. The neurochemistry of autism. Brain Sciences 2020; 10(3), 163.
CL Muller, AMJ Anacker and J Veenstra-VanderWeele. The serotonin system in autism spectrum disorder: From biomarker to animal models. Neuroscience 2016; 321, 24-41.
J Sinzig and G Lehmkuhl. What do we know about the serotonergic genetic heterogeneity in attention-deficit/hyperactivity and autistic disorders? Psychopathology 2007; 40(5), 329-337.
X Yang, G Wang, X Gong, C Huang, Q Mao, L Zeng, P Zheng, Y Qin, F Ye, B Lian, C Zhou, H Wang, W Zhou and P Xie. Effects of chronic stress on intestinal amino acid pathways. Physiology & Behavior 2019; 204, 199-209.
NNJ Rommelse, B Franke, HM Geurts, CA Hartman and JK Buitelaar. Shared heritability of attention-deficit/hyperactivity disorder and autism spectrum disorder. European Child & Adolescent Psychiatry 2010; 19(3), 281-295.
T Fan, Y Hu, J Xin, M Zhao and J Wang. Analyzing the genes and pathways related to major depressive disorder via a systems biology approach. Brain and Behavior 2020; 10(2), e01502.
CB Nemeroff and MJ Owens. The role of serotonin in the pathophysiology of depression: As important as ever. American Association for Clinical Chemistry 2009; 55(8), 1578-1579.
E Żmudzka, K Sałaciak, J Sapa and K Pytka. Serotonin receptors in depression and anxiety: Insights from animal studies. Life Sciences 2018; 210, 106-124.
P Belujon and AA Grace. Dopamine system dysregulation in major depressive disorders. International Journal of Neuropsychopharmacology 2017; 20(12),1036-1046.
Y Higuchi, T Soga and IS Parhar. Regulatory pathways of monoamine oxidase a during social stress. Frontiers in Neuroscience 2017; 11, 604.
JH Meyer, N Ginovart, A Boovariwala, S Sagrati, D Hussey, A Garcia, T Young, N Praschak-Rieder, AA Wilson and S Houle. Elevated monoamine oxidase a levels in the brain: An explanation for the monoamine imbalance of major depression. Archives of General Psychiatry 2006; 63(11), 1209.
JPM Finberg and JM Rabey. Inhibitors of MAO-A and MAO-B in psychiatry and neurology. Frontiers in Pharmacology 2016; 7, 340.
J Ekowati, BA Tejo, S Maulana, WA Kusuma, R Fatriani, R Fatriani, NS Rmadhanti, N Norhayati, KA Nofianti, MI Sulistyowaty, MS Zubair, T Yamauchi and IS Hamid. Potential utilization of phenolic acid compounds as anti-inflammatory agents through TNF-α convertase inhibition mechanisms: A network pharmacology, docking, and molecular dynamics approach. ACS Omega 2023; 8(49), 46851-46868.
KE Hevener, W Zhao, DM Ball, K Babaoglu, J Qi, SW White and RE Lee. Validation of molecular docking programs for virtual screening against dihydropteroate synthase. Journal of Chemical Information and Modeling 2009; 49(2), 444-460.
LD Colibus, M Li, C Binda, A Lustig, DE Edmondson and A Mattevi. Three-dimensional structure of human monoamine oxidase A (MAO A): Relation to the structures of rat MAO A and human MAO B. National Academy of Sciences 2005; 102(36), 12684-12689.
RM Geha, K Chen, J Wouters, F Ooms and JC Shih. Analysis of conserved active site residues in monoamine oxidase A and B and their three-dimensional molecular modeling. Journal of Biological Chemistry 2002; 277(19), 17209-17216.
IA Vakser. Challenges in protein docking. Current Opinion in Structural Biology 2020; 64, 160-165.
J Aghajani, P Farnia, P Farnia, J Ghanavi and AA Velayati. Molecular dynamic simulations and molecular docking as a potential way for designed new inhibitor drug without resistance. Tanaffos 2022; 21(1), 1-14.
V Salmaso and S Moro. Bridging molecular docking to molecular dynamics in exploring ligand-protein recognition process: An overview. Frontiers in Pharmacology 2018; 9, 923.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






