Unraveling Potential Bioactive Antimycobacterial Compounds from Endophytic Aspergillus tubingensis and Syncephalastrum racemosum: An Integrated Bioassay, Metabolite Profiling, and Molecular Mechanism Approach
DOI:
https://doi.org/10.48048/tis.2026.12071Keywords:
Antimycobacterial, Aspergillus tubingensis, Syncephalastrum racemosum, Ethyl acetate extract, Harrisonia perforata, Molecular docking, Mass Spectrometry, Antimycobacterial, Aspergillus tubingensis, Syncephalastrum racemosum, Ethyl acetate extract, Harrisonia perforata, Molecular docking, Mass SpectrometryAbstract
Tuberculosis (TB), a significant global health threat, ranks third in the world, with 67% of cases occurring in productive age groups. Natural compounds, such as fungal secondary metabolites, exhibit various pharmaceutically relevant activities, including antimycobacterial activity. Our ongoing research investigated the ethyl acetate extract of endophytic fungi Aspergillus tubingensis and Syncephalastrum racemosum from Rui (Harrisonia perforata) against Mycobacterium tuberculosis. The extract was obtained by fermenting the fungi on PDB and extracting the liquid culture using ethyl acetate. It showed antimycobacterial activity against Mycobacterium tuberculosis H37Rv by culturing the crude extract and bacteria in Middlebrook 7H9 Broth and Middlebrook 7H10 Agar. UHPLC-MS/MS profiling revealed about 85 compounds from Aspergillus tubingensis and 105 from Syncephalastrum racemosum. Molecular docking analysis revealed potential dual-action mechanisms by targeting two crucial enzymes in M. tuberculosis: KatG and KasA. Potential metabolites include bis(methylbenzylidene)sorbitol, sphinganine, and a chromenone derivative. In conclusion, the crude extract of Aspergillus tubingensis and Syncephalastrum racemosum demonstrates potential as an antimycobacterial agent.
HIGHLIGHTS
Our study focuses on an ethyl acetate extract of endophytic fungi Aspergillus tubingensis and Syncephalastrum racemosum isolated from Harrisonia perforata collected from Palu, Central Sulawesi, Indonesia. Endophytic fungi produce secondary metabolites with various pharmacological activities, including antibacterial activity. Tuberculosis (TB) remains a significant health problem, especially in Asia and Indonesia. The current TB medicine has become resistant to Mycobacterium tuberculosis, the leading cause of the disease. Therefore, finding new TB treatment agents is crucial. Our study aims to identify new agents for TB treatment and encourage scientists to explore endophytic fungi as potential sources. Our study is the first to profile the metabolites of Aspergillus tubingensis and Syncephalastrum racemosum for treating Mycobacterium. We conducted fungi fermentation and performed HPLC-HRMS to profile the metabolites. In vitro bioassays examined their activity, and molecular docking analyzed their molecular mechanisms.
GRAPHICAL ABSTRACT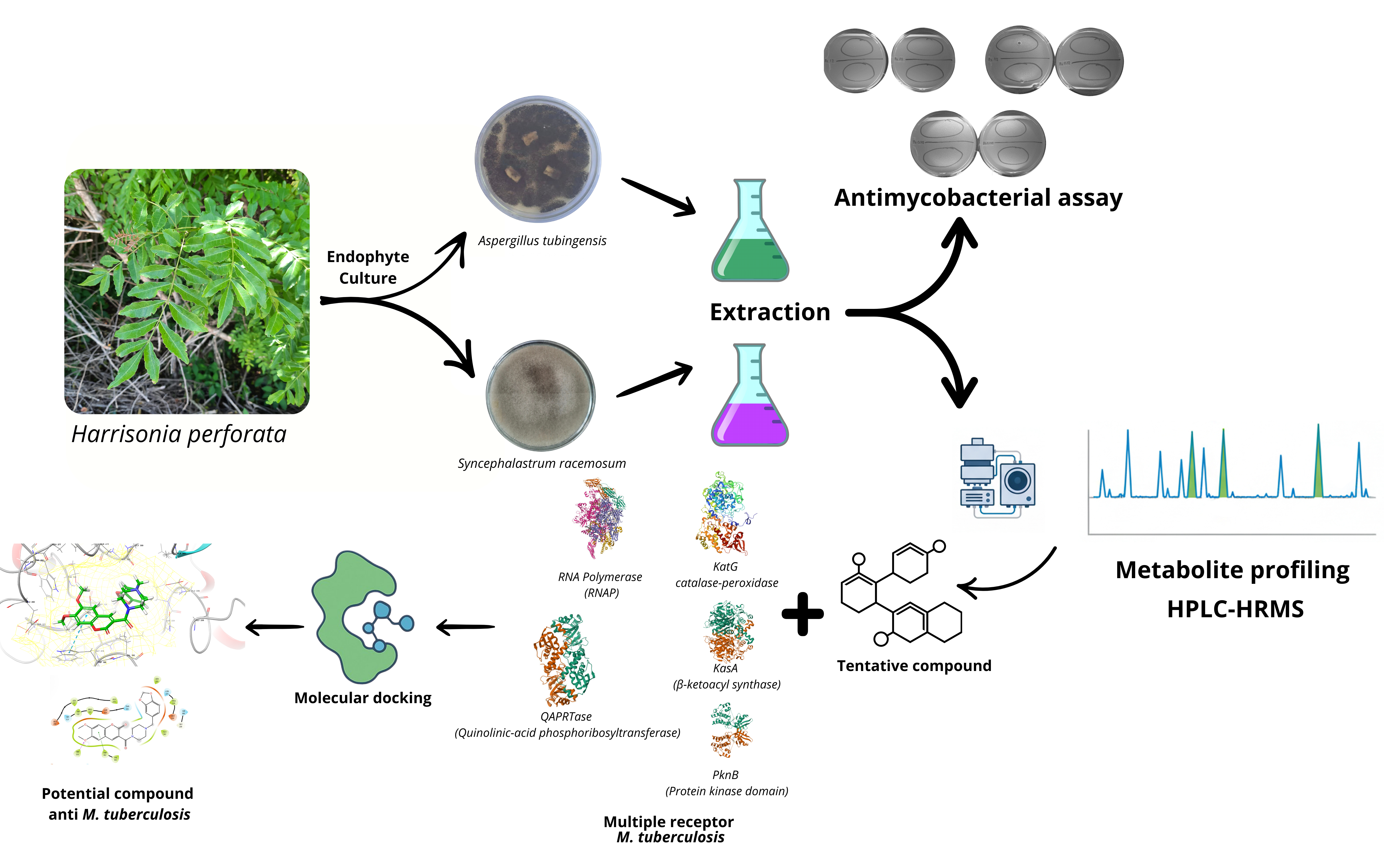
Downloads
References
Amr-review.org, Available at: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf, accessed October 2025.
JA Priyanto, ME Prastya, ENW Hening and RI Astuti. Exploring antimycobacterial potential and profiling secondary metabolite gene clusters in the whole genome of Bacillus isolated from dogfruit (Archidendron pauciflorum). Journal of Applied Pharmaceutical Science 2025; 15(3), 194-205.
Tbindonesia, Available at: https://www.tbindonesia.or.id, accessed October 2025.
Kompas, Available at: https://www.kompas.com/sains/read/2022/02/11/170500823/kemenkes-sebut-tuberkulosis-di-indonesia-masuk-3-besar-kasus-terbanyak-di?page=all, accessed October 2025.
CF Deacon. Physiology and pharmacology of DPP-4 in glucose homeostasis and the treatment of Type 2 diabetes. Front Endocrinol (Lausanne) 2019; 10, 80.
DJ Payne, MN Gwynn, DJ Holmes and DL Pompliano. Drugs for bad bugs: Confronting the challenges of antibacterial discovery. Nature Reviews Drug Discovery 2007; 6, 29-40.
World Health Organization, Available at: https://www.who.int/publications/i/item/9789241548809, accessed October 2025.
DJ Newman and GM Cragg. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. Journal of Natural Products 2020; 83, 770-803.
D Permatasari, R Pitopang, S Anam and dan Ivan. Uji daya hambat ekstrak batang tumbuhan harrisonia perforatamerr. terhadap pertumbuhan bakteri shigella dysentriae. Biocelebes 2015; 9(1), 1-7.
IO Aimang, Ramadhanil, S Anam and Ivan. Uji daya hambat daun harrisonia perforatamerr. terhadap pertumbuhan bakteri escherichia coli dan salmonella typhi. Biocelebes 2015; 9, 20-27.
P Tuntiwachwuttikul, P Phansa, Y Pootaeng-On and WC Taylor. Chromones from the branches of Harrisonia perforata. Chemical and Pharmaceutical Bulletin (Tokyo) 2006; 54(1), 44-47.
S Anam, A Syamsidi, Musyahidah, N Ambianti, A Widodo and MS Zubair. Isolation of endophytic fungi from benalu batu (Begonia Medicinalis) and their toxicity on Artemia Salina. Jurnal Ilmiah Farmasi (Scientific Journal of Pharmacy) 2022; 2022, 20-30.
RCG Corrêa, SA Rhoden, TR Mota, JL Azevedo, JA Pamphile, CGM de Souza, M de Lourdes Teixeira de Moraes Polizeli, A Bracht and RM Peralta. Endophytic fungi: Expanding the arsenal of industrial enzyme producers. Journal of Industrial Microbiology and Biotechnology 2014; 41(10), 1467-1478.
A Alvin, KI Miller and BA Neilan. Exploring the potential of endophytes from medicinal plants as sources of antimycobacterial compounds. Microbiological Research 2014; 169(7-8), 483-495.
CR de Carvalho, MQ Maia, M Sobral, GMD Pereira, K da Silva, MJS Vital, JÉ Zilli, CA Rosa and LH Rosa. Diversity and antimicrobial activity of culturable endophytic fungi associated with the neotropical ethnomedicinal plants Copaifera langsdorffii and Copaifera pubiflora. South African Journal of Botany 2021; 142, 305-315.
S Dettrakul, P Kittakoop, M Isaka, S Nopichai, C Suyarnsestakorn, M Tanticharoen and Y Thebtaranonth. Antimycobacterial pimarane diterpenes from the fungus Diaporthe sp. Bioorganic & Medicinal Chemistry Letters 2003; 13(7), 1253-1255.
A Zerroug, N Sadrati, R Demirel, B Sabrina and D Harzallah. Antibacterial activity of endophytic fungus, Penicillium griseofulvum MPR1 isolated from medicinal plant, Mentha pulegium L. African Journal of Microbiology Research 2018; 12(48), 1056-1066.
H Mohamed, W Ebrahim, M El-Neketi, MF Awad, H Zhang, Y Zhang and Y Song. In vitro phytobiological investigation of bioactive second¬ary metabolites from the Malus domestica-De¬rived endophytic fungus Aspergillus tubingensis strain AN103. Molecules 2022; 27(12), 3762.
KI Tapfuma, K Nyambo, F Adu-Amankwaah, L Baatjies, L Smith, N Allie, M Keyster, AG Loxton, M Ngxande, R Malgas-Enus and V Mavumengwana. Antimycobacterial activity and molecular docking of methanolic extracts and compounds of marine fungi from Saldanha and False Bays, South Africa. Heliyon 2022; 8, 12406.
WJ Andrioli, TLBV Simão, DP Ferreira, MH Araújo, SD Calixto, JK Bastos, L Seldin, E Lasunskaia and MF Muzitano. Antimycobacterial and anti-inflammatory activities of metabolites from endophytic and soil fungi. Phytomedicine Plus 2022; 2(3), 100312.
M Azhari, N Merliani, M Singgih, M Arai and E Julianti. Insights into natural products from marine-derived fungi with antimycobacterial properties: Opportunities and challenges. Marine Drugs 2025; 23, 279.
S Anam, A Syamsidi, MFU Tunreng, HF Djaleha, WN Arisca, G Syaputra, Iklima, MF Indriani, A Widodo,
R Pratiwi, Yuliet and AR Razak. Isolation of endophytic fungi from rui (Harrisonia perforata (Blanco) Merr.) and determining their antibacterial, antioxidant, and cytotoxic activity. HAYATI Journal of Biosciences 2024; 31(3), 443-456.
TNT Hamzah, SY Lee, A Hidayat, R Terhem, I Faridah-Hanum and R Mohamed. Diversity and characterization of endophytic fungi isolated from the tropical mangrove species, Rhizophora mucronata, and identification of potential antagonists against the soil-borne fungus, Fusarium solani. Frontiers Microbiology 2018; 9, 1707.
J Wang, X Wei, X Qin, X Lin, X Zhou, S Liao, B Yang, J Liu, Z Tu and Y Liu. Arthpyrones A-C, pyridone alkaloids from a sponge-derived fungus Arthrinium arundinis ZSDS1-F3. Organic Letters 2015; 17, 656-659.
AN Garmana, EY Sukandar and I Fidrianny. Uji aktivitas ekstrak beberapa tumbuhan terhadap Mycobacterium tuberculosis galur sensitif dan resisten. Acta Pharmaceutica Indonesia 2011; 36(3-4), 35-39.
Tuberculosis Cluster Bureau of AIDS, T., and SITs. Standard Operating Procedure for Drug Susceptibility Testing in Tuberculosis Laboratory, International Training Workshop on “Laboratory Methods for Drug Susceptibility Testing inTuberculosis”. Bangkok, 2007.
H Ardalani, S Anam, KJK Kromphardt, D Staerk and KT Kongstad. Coupling microplate-based antibacterial assay with liquid chromatography for high-resolution growth inhibition profiling of crude extracts: Validation and proof-of-concept study with Staphylococcus aureus. Molecules 2021; 26(6), 1550.
KT Kongstad, SG Wubshet, A Johannesen, L Kjellerup, A-ML Winther, AK Jäger and D Staerk. High-resolution screening combined with HPLC-HRMS-SPE-NMR for identification of fungal plasma membrane H+-ATPase inhibitors from plants. Journal of Agricultural and Food Chemistry 2014; 62(24), 5595-5602.
N Fabre, I Rustan, E de Hoffmann and J Quetin-Leclercq. Determination of flavone, flavonol, and flavanone aglycones by negative ion liquid chromatography electrospray ion trap mass spectrometry. Journal of the American Society for Mass Spectrometry 2001; 12, 707-715.
A Hagag, MF Abdelwahab, AMA El-Kader and MA Fouad. The endophytic aspergillus strains: A bountiful source of natural products. Journal of Applied Microbiology 2022; 132, 4150-4169.
AE Rahmawati, F Romadhonsyah, BM Gemantari, A Nurrochmad, S Wahyuono and P Astuti. Effect of light exposure on secondary metabolite production and bioactivities of Syncephalastrum racemosum endophyte. Tropical Journal of Natural Product Research 2021; 5, 312-318.
MC Ferreira, CL Cantrell, DE Wedge, VN Gonçalves, MR Jacob, S Khan, CA Rosa and LH Rosa. Antimycobacterial and antimalarial activities of endophytic fungi associated with the ancient and narrowly endemic neotropical plant Vellozia gigantea from Brazil. Memórias do Instituto Oswaldo Cruz 2017; 112(10), 692-697.
J Li, R Duan, ES Traore, RC Nguyen, I Davis, WP Griffth, DC Goodwin, AA Jarzecki and A Liu. Indole N-Linked hydroperoxyl adduct of protein-derived cofactor modulating catalase-peroxidase functions. Angewandte Chemie International Edition 2024; 63, 202407018.
P Kumar, GC Capodagli, D Awasthi, R Shrestha, K Maharaja, P Sukheja, S-G Li, D Inoyama, M Zimmerman, HPH Liang, J Sarathy, M Mina, G Rasic, R Russo, AL Perryman, T Richmann, A Gupta, E Singleton, S Verma, S Husain, ... , D Alland. Synergistic lethality of a binary inhibitor of mycobacterium tuberculosis KasA. mBio 2018; 9(6), 02101-02117.
O Burastero, M Cabrera, ED Lopez, LA Defelipe, JP Arcon, R Durán, MA Marti and AG Turjanski. Specificity and reactivity of Mycobacterium tuberculosis serine/threonine kinases PknG and PknB. Journal of Chemical Information and Modeling 2022; 62(7), 1723-1733.
AL Garner, J Rammohan, JP Huynh, LM Onder, J Chen, B Bae, D Jensen, LA Weiss, AR Manzano, SA Darst, EA Campbell, BE Nickels, EA Galburt and CL Stallings. Effects of increasing the affinity of CarD for RNA polymerase on mycobacterium tuberculosis growth, rRNA transcription, and virulence. Journal of Bacteriology 2017; 199(4), 0069816
J Ekowati, BA Tejo, S Maulana, WA Kusuma, R Fatriani, NS Ramadhanti, N Norhayati, KA Nofianti,
MI Sulistyowaty, MS Zubair, T Yamauchi and IS Hamid. potential utilization of phenolic acid compounds as anti-inflammatory agents through TNF-α convertase inhibition mechanisms: A network pharmacology, docking, and molecular dynamics approach. ACS Omega 2023; 8(49), 46851-46868.
J Fantini and FJ Barrantes. Sphin-golipid/cholesterol regulation of neurotransmitter receptor conformation and function. Biochimica et Biophysica Acta (BBA) - Biomembranes 2009; 1788(11), 2345-2361.
TC Kunz and V Kozjak-Pavlovic. Diverse facets of sphingolipid involvement in bacterial infections. Frontiers in Cell and Developmental Biology 2019; 7, 203.
S Sholekha, S Budiarti, AEZ Hasan, NPRA Krishanti and AT Wahyudi. Antimicrobial potential of an actinomycete Gordonia terrae JSN1.9-Derived orange pigment extrac. HAYATI Journal of Biosciences 2024; 31(1), 161-170.
DJ Bibel, R Aly and HR Shinefield. Antimicrobial activity of sphingosines. Journal of Investigative Dermatology 1992; 98(3), 269-273.
Y Wu, Y Liu, E Gulbins and H Grassmé. The anti-infectious role of sphingosine in microbial diseases. Cells 2021; 10(5), 1105.
SK Garg, E Volpe, G Palmieri, M Mattei, D Galati, A Martino, MS Piccioni, E Valente, E Bonanno, PD Vito, PM Baldini, LG Spagnoli, V Colizzi and M Fraziano. Sphingosine 1-phosphate induces antimicrobial activity both in vitro and in vivo. The Journal of Infectious Diseases 2004; 189(11), 2129-2138.
Y Wu, F Schnitker, Y Liu, S Keitsch, F Caicci, F Schumacher, A Riehle, B Pollmeier, J Kehrmann, B Kleuser, M Kamler, I Szabo, H Grassmé and E Gulbins. Sphingosine kills Mycobacteria and suppresses mycobacterial lung infections. Journal of Molecular Medicine 2025; 103(5), 547-558.
Bis(methylbenzylidene)sorbitol, Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Bis_methylbenzylidene_sorbitol, accessed October 2025.
AAG Indraningrat, PPCP Purnami, E Damayanti, MD Wijaya, DAPS Masyeni and NLPEK Sari. Antibacterial potential of pseudomonas aeruginosa ISP1RL4 isolated from seaweed eucheuma cottonii against multidrug-resistant bacteria. Biomedical & Pharmacology Journal 2024; 17(4), 2341-2354.
RS Keri, S Budagumpi, RK Pai and RG Balakrishna. Chromones as a privileged scaffold in drug discovery: A review. European Journal of Medicinal Chemistry 2014; 78, 340-374.
S Kamboj and R Singh. Chromanone-A prerogative therapeutic scaffold: An overview. Arabian Journal for Science and Engineering 2022; 47(1), 75-111.
V Nalla, A Shaikh, S Bapat, R Vyas, M Karthikeyan, P Yogeeswari, D Sriram and M Muthukrishnan. Identification of potent chromone embedded [1,2,3]-triazoles as novel anti-tubercular agents. Royal Society Open Science 2018; 5(4), 171750.
MF Alkaltham, AI Almansour, N Arumugam, SK Vagolu, T Tønjum, SI Alaqeel, S Rajaratnam and
V Sivaramakrishnan. Activity against Mycobacterium tuberculosis of a new class of spirooxindolopyrrolidine embedded chromanone hybrid heterocycles. RSC Advances 2024; 14(17), 11604-11613.
O Graudejus, RDP Wong, N Varghese, S Wagner and B Morrison. Bridging the gap between in vivo and in vitro research: Reproducing in vitro the mechanical and electrical environment of cells in vivo. Frontiers in Cellular Neuroscience 2018; 12.
E Madorran, A Stožer, S Bevc and U Maver. In vitro toxicity model: Upgrades to bridge the gap between preclinical and clinical research. Bosnian Journal of Basic Medical Sciences 2020; 20(2), 157-168.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






