The Influence of Reactant Concentration on the Characteristics of the As-Synthesized Cryptomelane Nanoparticles Prepared using Precipitation Method and Its Catalytic Performance for the Degradation of A Dye
DOI:
https://doi.org/10.48048/tis.2025.9286Keywords:
Cryptomelane nanoparticles, Co-precipitation method, Fenton-like process, Methylene blueAbstract
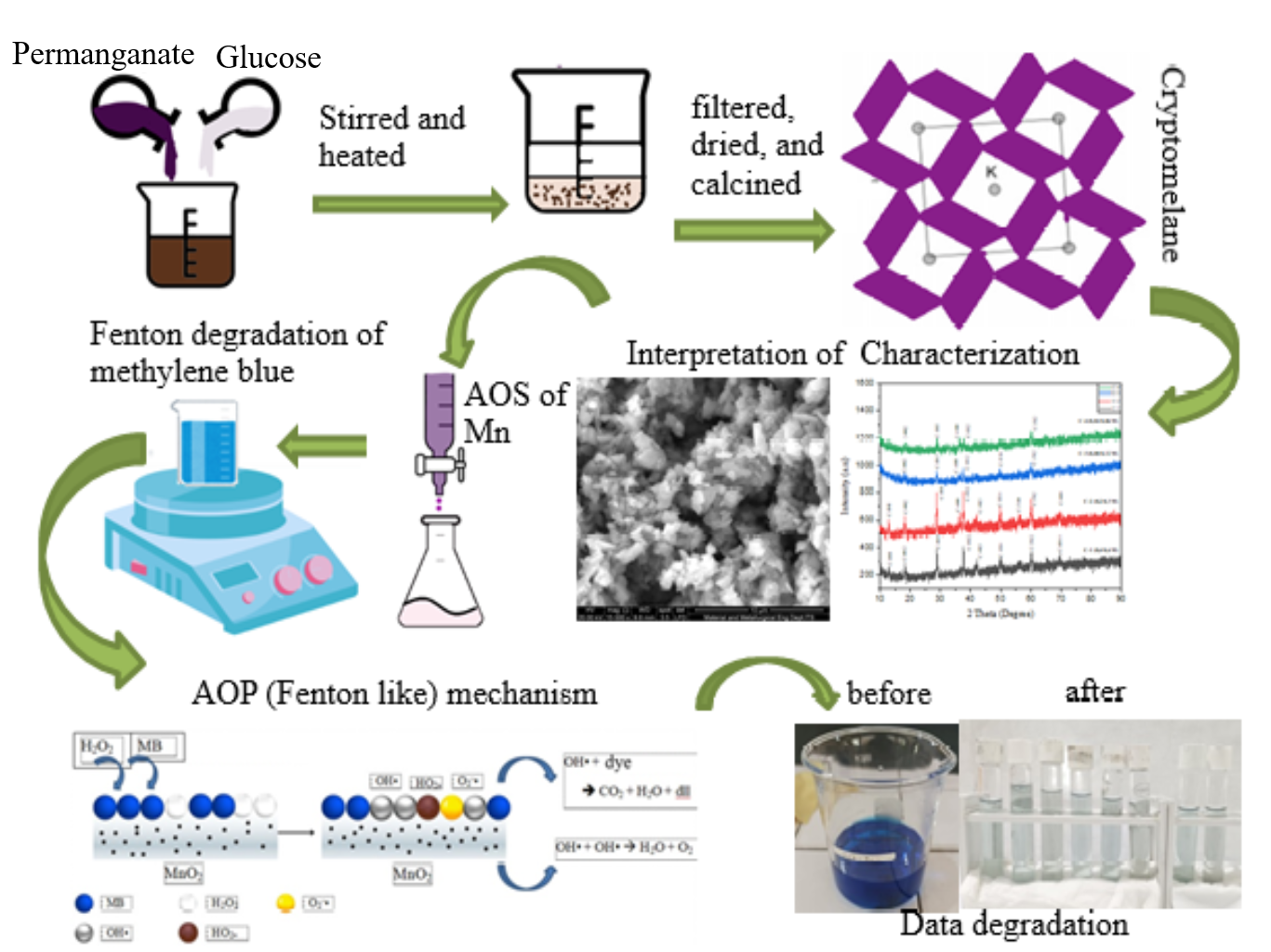
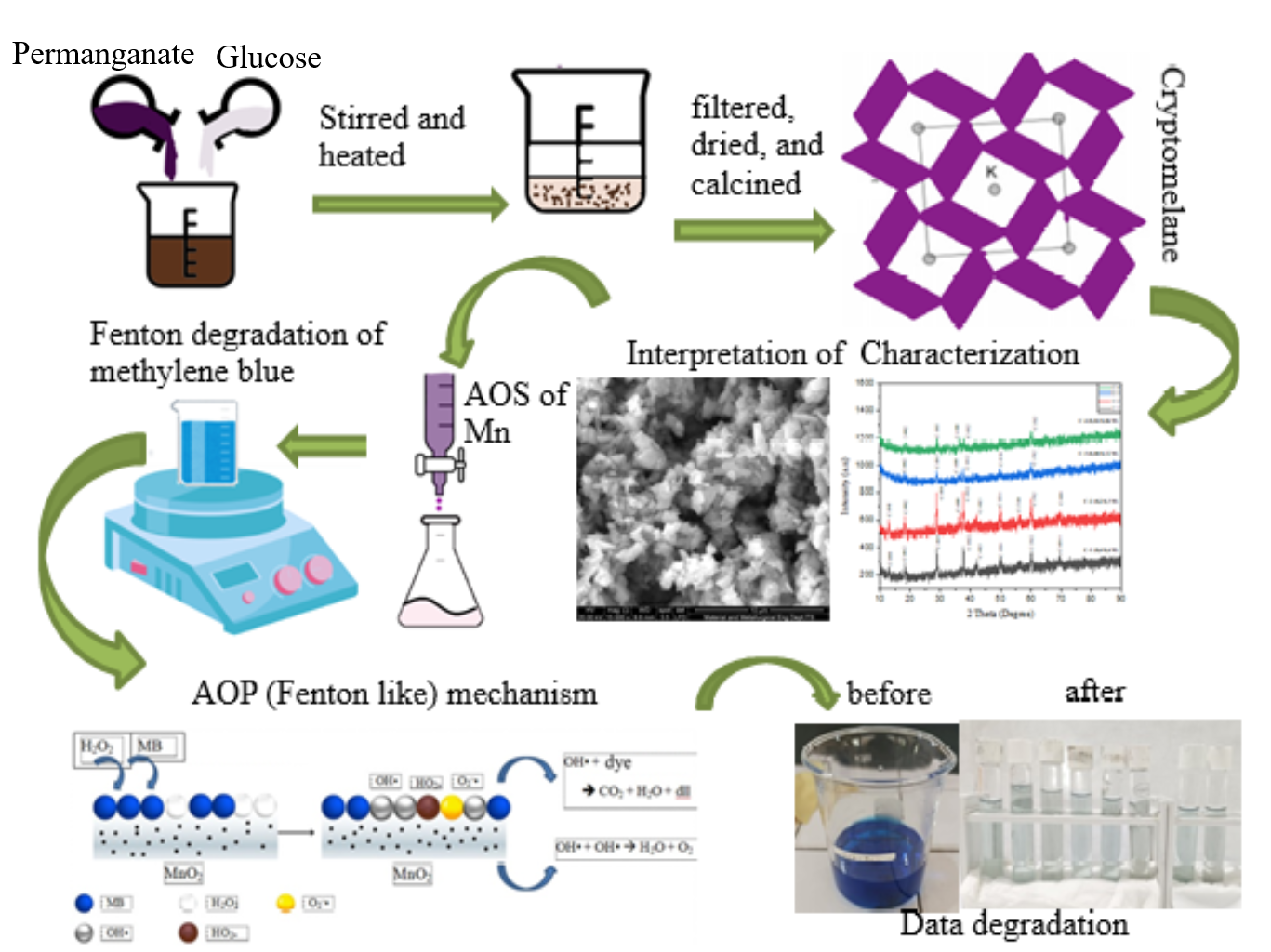
The high purity of the nanostructured cryptomelane-type manganese oxide catalysts was successfully synthesized using the different ratio concentrations of KMnO4 over glucose through a co-precipitation method. The both reactants were simultaneously mixed with the ratios of KMnO4 to glucose of 0.4M/0.6M, 0.2M/0.3M, 0.08M/0.12M and 0.04M/0.06M, respectively. The average oxidation states (AOS) Mn of the products were determined using the back-titration method, whereas the crystalite size was calculated using Scherrer equation. The XRD analysis showed that while the phase structure remained unaffected by the concentration and calcination temperature, it had a notable impact on AOS of Mn, the morphology and the particle size as shown by the SEM images. The lowest concentration ratio resulted in the small particle size and the low AOS of Mn of the as-synthesized cryptomelane, which correlated with more Mn3+ concentration or oxygen defect in the as-synthesized cryptomelane. The vibrational mode Mn-O was demonstrated by the absorption bands appearing in the FTIR characterization results. The specific Mn-O vibrations of the cryptomelane were demonstrated using Raman spectroscopy. The catalytic activity of the as-synthesized catalysts was tested in its ability to degrade MB using a Fenton-like method. The results indicated that there is a strong correlation between the catalytic performance and Mn3+ concentration or oxygen defects present in the catalyst. The catalyst with the lowest concentration ratio possessing the high Mn3+ concentration or oxygen defects demonstrated better catalytic activity compared to the other as-synthesized catalysts. The optimization of the catalyst was performed by varying solution pH, H2O2 volume, catalyst mass, and initial concentration of methylene blue (MB). According to the research findings, this catalyst achieved a MB degradation rate of 97.01 ± 0.33 % after 120 min of contact time. This result was obtained under conditions of pH = 11, with 15 mL of 30 % H2O2, a catalyst mass of 50 mg and an initial MB concentration of 30 ppm.
HIGHLIGHTS
- The influence of reactan concentration had a notable impact on average oxidation state (AOS) of Mn
- There is a strong correlation between the catalytic performance and Mn3+ concentration in the catalyst.
- The co-precipitation method produces a pure cryptomelane
GRAPHICAL ABSTRACT
Downloads
References
R Yang, Y Fan, R Ye, Y Tang, X Cao, Z Yin and Z Zeng. MnO2-based materials for environmental applications. Advanced Materials 2021; 33(9), e2004862.
B Debnath, AS Roy, S Kapri and S Bhattacharyya. Efficient dye degradation catalyzed by manganese oxide nanoparticles and the role of cation valence. ChemistrySelect 2016; 1(14), 4265-4273.
C Gong, X Lv, S Liu, X Chen, R Weerasooriya and Z Ding. Novel α-MnO2/AC catalysts for heterogeneous catalytic ozonation process to remove BAA in dye wastewater. Journal of Industrial and Engineering Chemistry 2024; 141, 340-350.
R Anggraini, SS Siregar, A Linggawati, H Sophia and A Awaluddin. Manganosite MnO-oil palm fly ash composite for pH-dependent degradation of methylene blue. Materials Today Proceedings 2023; 87(9), 370-375.
S Ching, JL Roark, N Duan and SL Suib. Sol-gel route to the tunneled manganese oxide Cryptomelane. Chemistry of Materials 1997; 9(3), 750-754.
A Awaluddin, M Agustina, RR Aulia and Muhdarina. Precursor effects on the morphology and crystallinity of manganese oxides and their catalytic application for MB degradation. AIP Conference Proceedings 2017; 1823, 020108.
A Awaluddin, E Amiruddin, SS Saidah and R Anggraini. The catalytic activity of manganosite MnO/activated carbon for photo-degradation of synthetic dye. E3S Web of Conferences 2023; 481(1), 03007.
A Awaluddin, R Zulfa, S Absus, Nurhayati, A Linggawati and SS Siregar. The enhanced catalytic activities of octahedral layer birnessite-type manganese oxide synthesized via precipitation method for the degradation of MB. IOP Conference Series: Materials Science and Engineering 2019; 509, 012011.
N Subramanian, B Viswanathan and TK Varadarajan. A facile morphology-controlled synthesis of potassium-containing manganese oxide nanostructures for electrochemical supercapacitor application. RSC Advances 2014; 4(1), 33911-33922.
S Kurniati, Asleni, A Linggawati, SS Siregar and A Awaluddin. Synthesis and catalytic activities of manganese oxides prepared by precipitation method: effects of mixing modes of reactants and calcination process. Journal of Physics: Conference Series 2019; 1351, 012035.
S Absus, R Zulfa, A Awaluddin, S Anita, SS Siregar and Prasetya. A facile synthesis of octahedral layered birnessite-type manganese oxide (OL-1) nanostructures with tremendous catalytic activity for MB degradation. AIP Conference Proceedings 2018; 2049, 020009.
S Asleni, A Kurniati, A Linggawati, SS Siregar and A Awaluddin. The tremendous influence of calcination process on the phase structure and catalytic activity of precipitation-processed MnO2. Journal of Physics Conference Series 2019; 1351(1), 012038.
P Enrico. Impact of textile industry liquid waste on the environment and application of ECO printing techniques as an effort to reduce waste. Moda 2018; 1(1), 5-13.
AM Wati, FW Mahatmanti, Jumaeri and AT Prasetya. Methylene blue adsorption by activated coal fly ash using microwave-assisted hydrothermal process. Journal of Chemical Research 2022, 18(1), 58-69.
AB Baunsele and H Missa. Langmuir and freundlich equation test on methylene blue adsorption by using coconut fiber biosorbent. Walisongo Journal of Chemistry 2021; 4(2), 131-138.
Fathoni. Utilization of technical bentonite as dye adsorbent. UNESA Journal of Chemistry 2016; 5(3), 18-22.
S Hashemian, MK Ardakani and H Salehifar. Kinetics and thermodynamics of adsorption methylene blue onto tea waste/CuFe2O4 composite. American Journal of Analytical Chemistry 2013; 4(7), 1-7.
AT Saputra, Y Tiandho and F Afriani. Studi kinetika pada proses elektrokoagulasi zat warna metilen biru. Indonesian Journal of Physics Research 2022; 3(1), 1-11.
Maryudi, A Aktawan and S Amelia. Treatment of MB dye waste using activated charcoal and activated zeolite with fe catalyst and hydrogen peroxide oxidizer. Journal of Chemical Research 2021; 12(2), 112-120.
YA Kakame, AD Wuntu and H Koleangen. Degradation and adsorption of MB dye using calcined ag-fish bone composite. Chemistry Progress 2018; 11(2), 58-62.
KC He, Y Chen, Z Tang and Y Hu. Removal of heavy metal ions from aqueous solution by zeolite. Environmental Science and Pollution Research 2016; 23(3), 2778-2788.
L Sun, D Hu, Z Zhang and X Deng. Oxidative degradation of MB via pds-based advanced oxidation process using natural pyrite. International Journal of Environmental Research and Public Health 2019; 16(23), 4773,
YD Deng and R Zhao. Advanced oxidation processes (AOPs) in wastewater treatment. Current Pollution Reports 2015; 1, 167-176.
M Subkhan, A Awaluddin, Prasetya, SS Siregar and R Anggraini. Degradasi Katalitik Zat Warna Metil Jingga Menggunakan Katalis Oksida Mangan Manganosite. Photon: Jurnal Sain Dan Kesehatan 2018; 9(1), 177-183.
D Yu, Y Ren, X Yu, X Fan, L Wang, R Wang, Z Zhao, K Cheng, Y Chen, Z Sojka, A Kotarba, Y Wei and J Liu. Facile synthesis of birnessite-type K2Mn4O8 and cryptomelane-type K2-xMn8O16 catalysts and their excellent catalytic performance for soot combustion with high resistance to H2O and SO2. Applied Catalysis B: Environmental 2021; 285, 119779.
ME Becerra, AM Suarez, NP Arias and O Giraldo. Decomposition of the MB dye using layered manganese oxide materials synthesized by solid state reactions. International Journal of Chemical Engineering 2018; 2018, 4902376
SS Siregar and A Awaluddin. Synthesis and catalytic activity of Birnessite-Type Manganese Oxide synthesized by solvent-free method. IOP Conference Series Materials Science and Engineering 2018; 345(1), 012005.
M Ghaly, SS Metwally, EA El-Sherief, EA Saad and ROA Rahman. Utilization of synthetic nano-cryptomelane for enhanced removal of cesium and cobalt ions from single and binary solutions. Journal of Radioanalytical and Nuclear Chemistry 2022; 331(1-3), 1821-1838.
Y Zhou, Y Feng, H Xie, J Lu, D Ding and S Rong. Cryptomelane nanowires for highly selective self-heating photothermal synergistic catalytic oxidation of gaseous ammonia. Applied Catalysis B: Environmental 2023; 331, 122668.
NTH Nguyen, GT Tran, NTT Nguyen, TT Nguyen, DTC Nguyen and TV Tran. A critical review on the biosynthesis, properties, applications and future outlook of green MnO2 nanoparticles. Environmental Research 2023; 231(2), 116262.
C Stella, N Soundararajan and K Ramachandran. Structural, optical, dielectric and magnetic properties of Mn1xCoxO2 nanowires. Superlattices and Microstructures 2014; 71, 203-210.
T Jakubek, C Hudy, J Gryboś, H Manyar and A Kotarba. Thermal transformation of birnessite (OL) towards highly active cryptomelane (OMS 2) catalyst for soot oxidation. Catalysis Letters 2019; 149(8), 2218-2225.
J Hou, Y Li, L Liu, R Lu and X Zhao. Effect of giant OVD on the catalytic oxidation of OMS-2 nanorods. Journal of Materials Chemistry A 2013; 1(23), 6736-6741.
Y Meng, W Song, H Huang, Z Ren, SY Chen and SL Suib. Structure-property relationship of 555 bifunctional MnO2 nanostructures: highly efficient, ultra-stable electrochemical water 556 oxidation and oxygen reduction reaction catalysts identified in alkaline media. Journal of the American Chemical Society 2014; 136(32), 11452-11464.
DM Robinson, YB Go, M Mui, G Gardner, Z Zhang, DDT Mastrogiovanni, E Garfunkel, J Li, M Greenblatt, GC Dismukes. Photochemical water oxidation by crystalline polymorphs of 630 manganese oxides: structural requirements for catalysis. Journal of the American Chemical Society 2013; 135(9), 3494-3501.
U Maitra, B Naidu, A Govindaraj and CNR Rao. Importance of trivalency and the eg1 632 configuration in the photocatalytic oxidation of water by Mn and Co oxides. Proceedings of the National Academy of Sciences of the United States of America 2013; 110(29), 11704-11707
W Yuan, X Yang, C Liu, L Xue, W Niu, Q Yan, Y Zhu, J Han, W Guo and B Zhang. High-surface-area Co-Cu-B monolithic self-supported catalyst for efficient sodium borohydride hydrolysis. Processes 2024, 12(7), 1384.
JR Frade. Kinetics of nucleation and growth. Journal of Materials Science 1993; 28, 6715-6718
Y Zheng, Y Shen, Y Ma, J Wang, X Wu, M Yang, M Xu and Jiao Wang. Nucleation, growth, and aggregation kinetics of KCI produced by stirred crystallization. Applied Physics A 2023; 129(9), 651.
H Zhang, S Du, Y Wang and F Xue. Prevention of crystal agglomeration: Mechanisms, factors, and impact of additives. Crystals 2024; 14(8), 676.
S Jia, X Wan, T Yao, S Guo, Z Gao, J Wang and J Gong. Separation performance and agglomeration behavior analysis of solution crystallization in food engineering. Food Chemistry 2023; 419, 136051.
LA Vijaya, K Mounika, T Jyothi and C Sailu. Effect of temperature and processing time on agglomerates prepared by spherical crystallization. Journal of Emerging Technologies and Innovative Research 2021; 8(5), d375-d383.
C Yu, G, Li, L Wei, Q Fan, Q Shu and JC Yu. Fabrication, characterization of B-MnO2 microrod catalysts and their performance in rapid degradation of dyes of high concentration. Catalysis Today 2021; 224, 154-162.
TT Yu, KL Li, XL Guo, F Li, JM Huang and YX Zhang. Facile decolorization of methylene blue by morphology-dependence δ-MnO2 nanosheets -modified diatomite. Journal of Physiscs and Chemsistry of Solids 2015; 87, 196-202.
Z Erda, Nurhayati, E Amirudin, R Anggraini, SS Siregar and A Awaluddin. Influencing parameters for degradation of MB using the catalyst bentonite supported manganosite MnO synthesized via facile, one-pot sol-gel route. Journal of Physics: Conference Series. 2021; 2049(1), 012061.
S Naniwa, A Yamamoto and H Yoshida. Visible light-induced Minisci reaction through photoexcitation of surface Ti-peroxo species. Catalysis Science & Technology 2021; 11(10), 3376-3384.
HJ Ciu, HZ Huang, B Yuan and ML Fu. Decolorization of RhB dye by manganese oxides: Effect of crystal type and solution pH. Geochemical Transaction 2015; 16(1), 10.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






