Artemisia Vulgaris: Biological Properties of a Potential Source for Cosmetics
DOI:
https://doi.org/10.48048/tis.2025.9094Keywords:
Artemisia vulgaris, Antioxidant, Collagenase, Cosmetics, Cytotoxicity, Mugwort, TyrosinaseAbstract
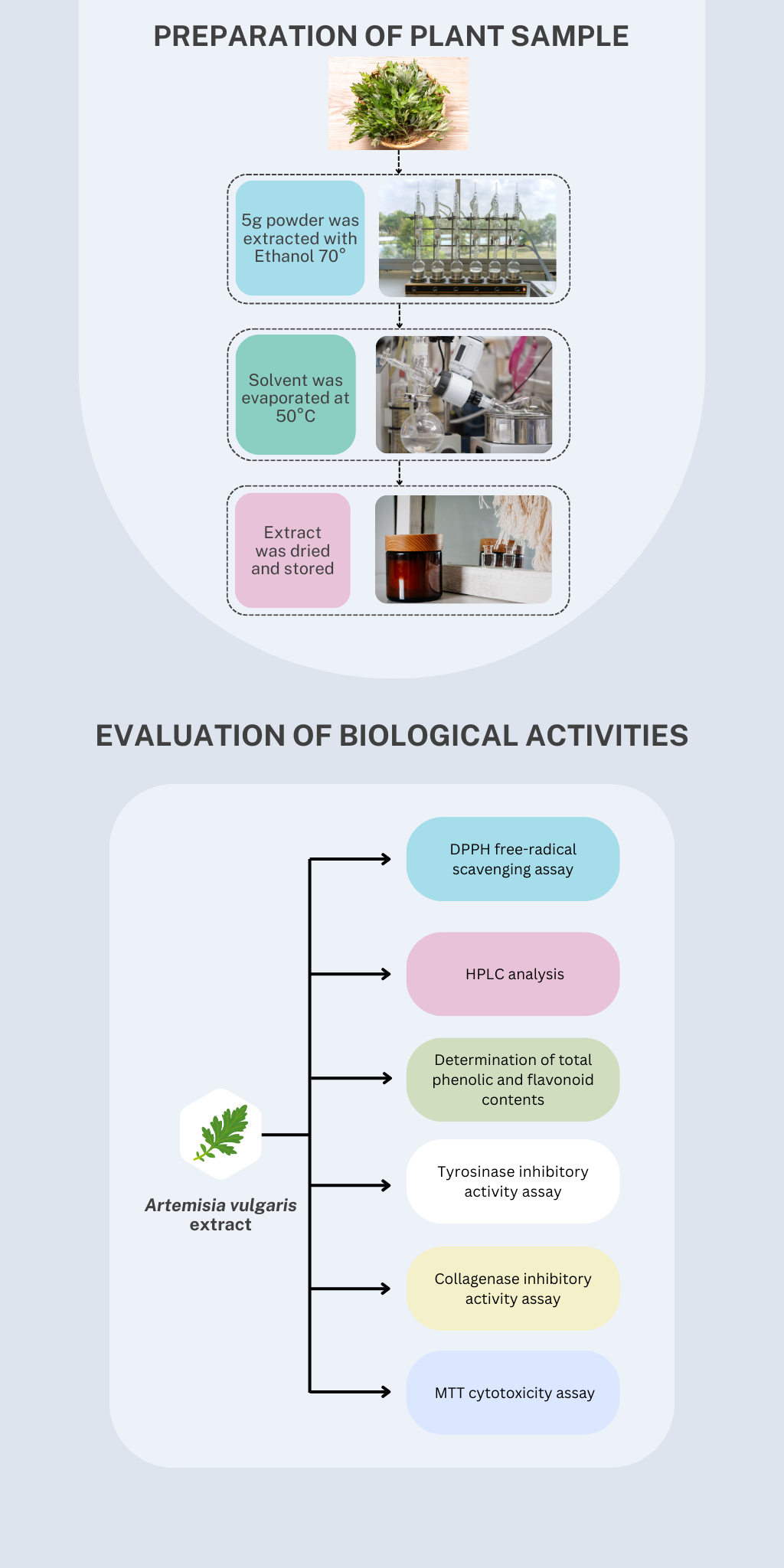
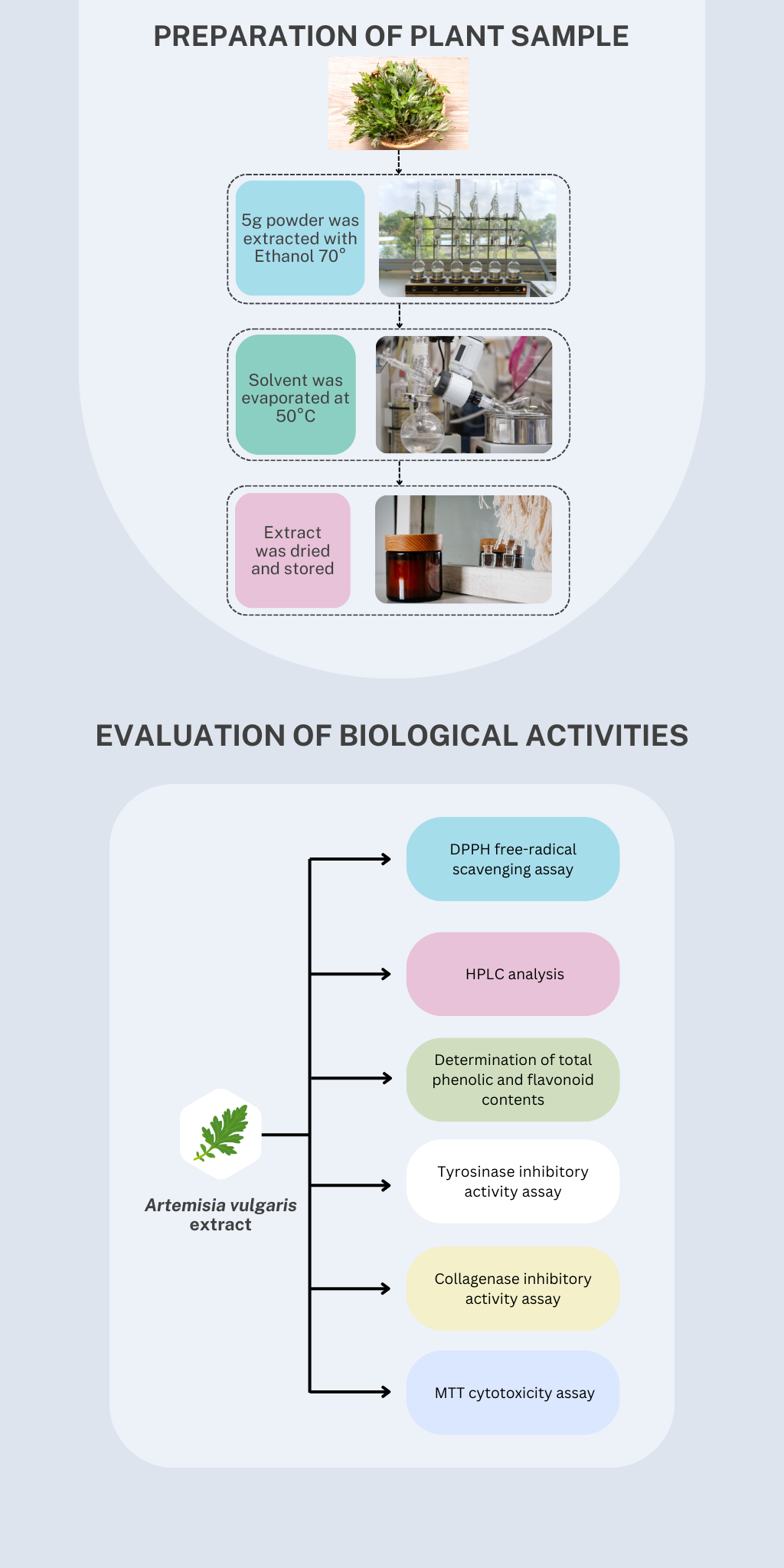
Artemisia vulgaris, commonly known as mugwort, has been traditionally used for the treatment of gastrointestinal and gynecological disorders. This plant has been scientifically reported to posess various biological activities such as antimicrobial, antioxidant and anti-inflammatory. This study aimed to investigate various biological activities including antioxidant, anti-tyrosinase, anti-collagenase and cytotoxicity. The antioxidant of this plant was evaluated using DPPH assay. Tyrosinase inhibitory assay was performed with the presence of DOPA as oxidase activity while the assessment of collagenase inhibitory activity was carried out based on spectrophotometric methods. MTT assay was conducted for the investigation of cytotoxicity against L-929 fibroblast cell lines. The IC50 of DPPH free-radical scavenging activity was recorded to be 38.42 ± 6.3 µg/mL compared to that of vitamin C (2.71 ± 0.8 µg/mL). The IC50 of tyrosinase inhibitory activity was 18.1 ± 2.4 compared to that of kojic acid (51.34 ± 2.8 µg/mL). Meanwhile, the ethanolic Artemisia vulgaris leaf extracts were capable of inhibiting collagenase activity with IC50 value of 10.38 ± 4.32 µg/mL compared to that of quercetin (8.68 ± 0.84 µg/mL). The extracts exhibited a cytotoxic effect on L-929 fibroblast cells in a dose-dependent manner; however, a significant cytotoxic effect was only documented at a dose of larger than 250 µg/mL. These promising findings partially elucidated the potential of Artemisia vulgaris as a source of cosmetics but need to be further extensively investigated.
HIGHLIGHTS
- The power of ethanolic Artemisia vulgaris leaf extracts, capable of being a potential tyrosinase inhibitor, may thus be used as a treatment for some skin disorders associated with melanin hyperpigmentation and can be incorporated into cosmetic formulas for skin-whitening effects.
- The present finding suggest that the ethanolic A. vulgaris leaf extracts may be considered as natural collagenase inhibitors that are required for a new source of anti-aging agents, in order to avoid the skin became aged or wrinkled.
- The ethanolic A. vulgaris leaf extracts were not significantly toxic to L-929 fibroblast cells at a concentration of less than 250 µg/mL with the small number of abnormal morphology cells.
GRAPHICAL ABSTRACT
Downloads
References
K Sumit, S Vivek, S Sujata and B Ashish. Herbal cosmetics: Used for skin and hair. Inventi Rapid: Cosmeceuticals 2012; 2012(4), 1-7.
M Kunal. Antioxidant analysis of essential oils and methanolic extracts of Artemisia vulgaris. International Journal of Agriculture Sciences 2018; 10(7), 5710-5713.
LS Joshi and HA Pawar. Herbal cosmetics and cosmeceuticals: An overview. Natural Products Chemistry & Research 2015; 3(2), 1000170.
DJ Brown and AM Dattner. Phytotherapeutic approaches to common dermatologic conditions. Archives of Dermatology 1998; 134(11), 1401-1404.
J Koo and S Arain. Traditional Chinese medicine for the treatment of dermatologic disorders. Archives of Dermatology 1998; 134(11), 1388-1393.
G Lyss, TJ Schmidt, I Merfort and HL Pahl. Helenalin, an anti-inflammatory sesquiterpene lactone from Arnica, selectively inhibits transcription factor NF-kappaB. Biological Chemistry 1997; 378(9), 951-961.
SEE Efem and CI Iwara. The antimicrobial spectrum of honey and its clinical significance. Infection 1992; 20(4), 227-229.
SK Katiyar, N Ahmad and H Mukhtar. Green tea and skin. Archives of Dermatology 2000; 136(8), 989-994.
IR Record and IE Dreosti. Protection by black tea and green tea against UVB and UVA+B induced skin cancer in hairless mice. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 1998; 422(1), 191-199.
AK Mohiuddin. Cosmetics in use: A pharmacological review. Journal of Dermatology & Cosmetology 2019; 3(2), 50-67.
GS Bamunuarachchi, WD Ratnasooriya, S Premakumara and PV Udagama. Antimalarial properties of Artemisia vulgaris L. ethanolic leaf extract in a Plasmodium berghei murine malaria model. Journal of Vector Borne Diseases 2013; 50(4), 278-284.
T Rasheed, M Bilal, HMN Iqbal and C Li. Green biosynthesis of silver nanoparticles using leaves extract of Artemisia vulgaris and their potential biomedical applications. Colloids and Surfaces B: Biointerfaces 2017; 158, 408-415.
H Ekiert, J Pajor, P Klin, A Rzepiela, H Ślesak and A Szopa. Significance of Artemisia vulgaris L. (common mugwort) in the history of medicine and its possible contemporary applications substantiated by phytochemical and pharmacological studies. Molecules 2020; 25(19), 4415.
P Si-Hyang, C Duck-Moon, C Byeong-Dae, C Yeung-Joon and C Jin-Ho. Antioxidative effects of skinned mugwort (Artemisia vulgaris L.) extracts on UV-irradiated hairless mouse skin (in Korean). Journal of the Korean Society of Food Science and Nutrition 2008; 37(1), 20-26.
TK Lee and CS Vairappan. Antioxidant, antibacterial and cytotoxic activities of essential oils and ethanol extracts of selected South East Asian herbs. Journal of Medicinal Plant Research 2011; 5(21), 5284-5290.
HC Huang, HF Wang, KH Yih, LZ Chang and TM Chang. Dual bioactivities of essential oil extracted from the leaves of Artemisia argyi as an antimelanogenic versus antioxidant agent and chemical composition analysis by GC/MS. International Journal of Molecular Sciences 2012; 13(11), 14679-14697.
WJ Zhang, CX You, K Yang, R Chen, Y Wang, Y Wu, ZF Geng, HP Chen, HY Jiang, Y Su, N Lei, P Ma, SS Du and ZW Deng. Bioactivity of essential oil of Artemisia argyi Lévl. et Van. and its main compounds against Lasioderma serricorne. Journal of Oleo Science 2014; 63(8), 829-837.
O Folin and V Ciocalteu. On tyrosine and tryptophane determinations in proteins. Journal of Biological Chemistry 1927; 73(2), 627-650.
EN Sembiring, B Elya and R Sauriasari. Phytochemical screening, total flavonoid and total phenolic content and antioxidant activity of different parts of Caesalpinia bonduc (L.) Roxb. Pharmacognosy Journal 2017; 10, 123-127.
KS Kim, S Lee, YS Lee, SH Jung, Y Park, KH Shin and BK Kim. Anti-oxidant activities of the extracts from the herbs of Artemisia apiacea. Journal of Ethnopharmacology 2003; 85(1), 69-72.
E Barrantes and M Guinea. Inhibition of collagenase and metalloproteinases by aloins and aloe gel. Life Sciences 2003; 72(7), 843-850.
A Temraz and WH El-Tantawy. Characterization of antioxidant activity of extract from Artemisia vulgaris. Pakistan Journal of Pharmaceutical Sciences 2008; 21(4), 321-326.
N Babbar, HS Oberoi, SK Sandhu and VK Bhargav. Influence of different solvents in extraction of phenolic compounds from vegetable residues and their evaluation as natural sources of antioxidants. Journal of Food Science and Technology 2014; 51(10), 2568-2575.
H Tang, X Li, X Liu, Y Xu and J Shen. Rutin intake mitigates the injury of blue light irradiation by altering aging rates of mortality in Drosophila model. Photochemistry and Photobiology 2023; 100(3), 524-529.
I Karabegović, M Nikolova, D Veličković, S Stojičević, V Veljković and M Lazić. Comparison of antioxidant and antimicrobial activities of methanolic extracts of the Artemisia sp. recovered by different extraction techniques. Chinese Journal of Chemical Engineering 2011; 19(3), 504-511.
M Ahmed, M Ji, P Qin, Z Gu, Y Liu, A Sikandar, MF Iqbal, A Javeed, J Shafi and Y Du. Determination of phytochemicals, antioxidant activity and biochemical composition of Chinese mugwort (Artemisia argyi L.) leaf extract from Northeast China. Applied Ecology and Environmental Research 2019; 17(6), 15349-15362.
M Borkataky, BB Kakoty and LR Saikia. Influence of total phenolic content and total flavonoid content on the DPPH radical scavenging activity of Eclipta alba (L.) Hassk. International Journal of Pharmacy and Pharmaceutical Sciences 2013; 5(S1), 324-327.
KG Strothkamp, RL Jolley and HS Mason. Quaternary structure of mushroom tyrosinase. Biochemical and Biophysical Research Communications 1976; 70(2), 519-524.
E Amin, AA Saboury, H Mansuri-Torshizi and AA Moosavi-Movahedi. Potent inhibitory effects of benzyl and p-xylidine-bis dithiocarbamate sodium salts on activities of mushroom tyrosinase. Journal of Enzyme Inhibition and Medicinal Chemistry 2010; 25(2), 272-281.
K Ohguchi, M Koike, Y Suwa, S Koshimizu, Y Mizutani, Y Nozawa and Y Akao. Inhibitory effects of whisky congeners on melanogenesis in mouse B16 melanoma cells. Bioscience, Biotechnology, and Biochemistry 2008; 72(4), 1107-1110.
A Palumbo, M d’Ischia, G Misuraca and G Prota. Mechanism of inhibition of melanogenesis by hydroquinone. Biochimica et Biophysica Acta (BBA) - General Subjects 1991; 1073(1), 85-90.
K Maeda and M Fukuda. In vitro effectiveness of several whitening cosmetic components in human melanocytes. Journal of the Society of Cosmetic Chemists 1991; 42(6), 361-368.
FA Badria and MA elGayyar. A new type of tyrosinase inhibitors from natural products as potential treatments for hyperpigmentation. Bollettino Chimico Farmaceutico 2001; 140(4), 267-271.
I Kubo and I Kinst-Hori. Tyrosinase inhibitors from anise oil. Journal of Agricultural and Food Chemistry 1998; 46(4), 1268-1271.
I Kubo and I Kinst-Hori. Tyrosinase inhibitors from cumin. Journal of Agricultural and Food Chemistry 1998; 46(12), 5338-5341.
I Kubo and I Kinst-Hori. Tyrosinase inhibitory activity of the olive oil flavor compounds. Journal of Agricultural and Food Chemistry 1999; 47(11), 4574-4578.
TSA Thring, P Hili and DP Naughton. Anti-collagenase, anti-elastase and anti-oxidant activities of extracts from 21 plants. BMC Complementary and Alternative Medicine 2009; 9, 27.
M Chatatikun and A Chiabchalard. Thai plants with high antioxidant levels, free radical scavenging activity, anti-tyrosinase and anti-collagenase activity. BMC Complementary and Alternative Medicine 2017; 17, 487.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






