Large-Scale Synthesis and Comprehensive Validation of 2-Hydroxypropyl-β-Cyclodextrin Equivalent to Commercial Product
DOI:
https://doi.org/10.48048/tis.2026.12144Keywords:
β-cyclodextrin, Characterization, Hydroxypropyl-β-cyclodextrin, Structure, SynthesisAbstract
This study was a development from a previous study, which established the method for synthesizing 2-hydroxypropyl-β-cyclodextrin (HPβCD) from β-cyclodextrin and successfully applied it in large-scale production. The large-scale synthesis of HPβCD produced 1.38 ± 0.05 kg of 2-Hydroxypropyl-β-cyclodextrin, yielding 86.77 ± 0.68%, and a degree of substitution of 5.91. The synthesis was monitored using thin-layer chromatography. The synthesized product was then structurally characterized using infrared spectroscopy, nuclear magnetic resonance spectroscopy, and mass spectroscopy, along with the commercial HPβCD. As a comparative analysis, the spectroscopic data revealed no significant structural differences between the synthesized HPβCD and a commercially available counterpart. These findings validate the developed synthesis method, highlighting its potential for industrial application and future research into the uses of HPβCD in drug development.
HIGHLIGHTS
- Successful Large-Scale Synthesis: A previously established method for synthesizing 2-hydroxypropyl-β-cyclodextrin (HPβCD) from β-cyclodextrin was successfully scaled up to achieve 1.38 ± 0.05 kg, yielding a high yield of 86.77 ± 0.68% and a degree of substitution of 5.91.
- Comprehensive Characterization: The synthesized HPβCD was rigorously monitored using thin-layer chromatography and structurally characterized by infrared spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectroscopy.
- Validation for Industrial Application: Comparative spectroscopic analysis against a commercial standard confirmed no significant structural differences, thereby validating the potential of this synthesis method for industrial application and future pharmaceutical research into HPβCD’s utility.
GRAPHICAL ABSTRACT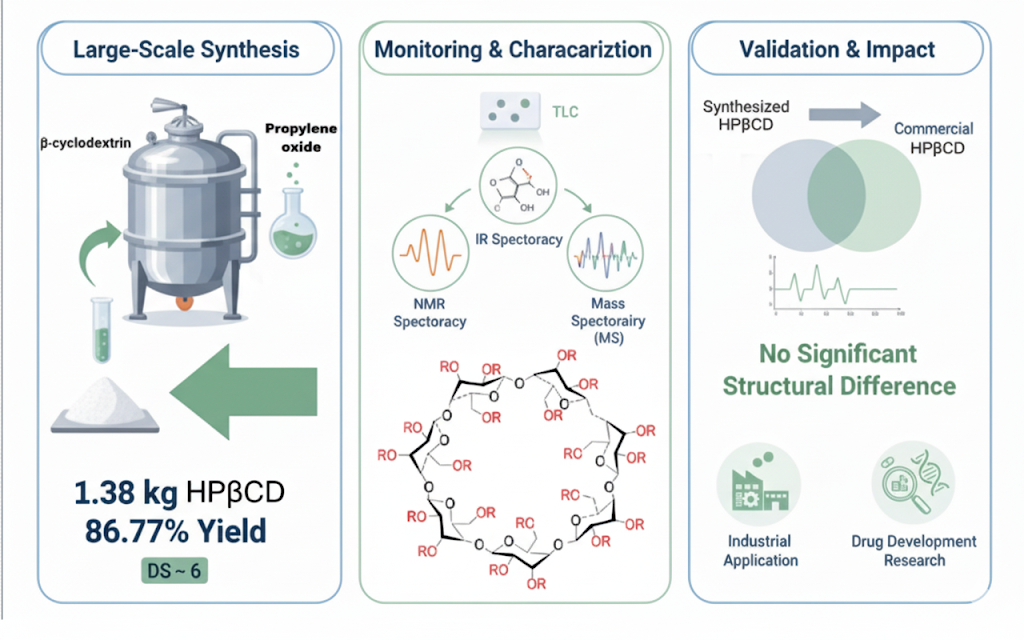
Downloads
References
V Saharan, V Kukkar, M Kataria, M Gera and PK Choudhury. Dissolution enhancement of drugs. Part I: Technologies and effect of carriers. International Journal of Health Research 2010; 2(2), 107-124.
VA Saharan, V Kukkar, M Kataria, M Gera and PK Choudhury. Dissolution enhancement of drugs part II: Effect of carriers. International Journal of Health Research 2009; 2(2), 207-223.
AS Deshmukh, KJ Tiwari and VR Mahajan. Solubility enhancement techniques for poorly water-soluble drugs. International Journal of Pharmaceutical Sciences and Nanotechnology 2017; 10(3), 3701-3708.
ME Brewster, P Neeskens and J Peeters. Solubilization of itraconazole as a function of cyclodextrin structural space. Journal of Inclusion Phenomena and Macrocyclic Chemistry 2007; 57(1-4), 561-566.
A Katdare and M Chaubal. Excipient development for pharmaceutical, biotechnology, and drug delivery systems. CRC Press, Boca Raton, United States, 2006.
S Gould and RC Scott. 2-Hydroxypropyl-β-cyclodextrin (HP-β-CD): A toxicology review. Food and Chemical Toxicology 2005; 43(10), 1451-1459.
H Jiang, R Sidhu, H Fujiwara, M De Meulder, R De Vries, Y Gong, M Kao, FD Porter, NM Yanjanin, N Carillo-Carasco, X Xu, E Ottinger, M Woolery, DS Ory and X Jiang. Development and validation of sensitive LC-MS/MS assays for quantification of HP-#x03B2;-CD in human plasma and CSF. Journal of Lipid Research 2014; 55(7), 1537-1548.
ME Brewster and T Loftsson. Cyclodextrins as pharmaceutical solubilizers. Advanced Drug Delivery Reviews 2007; 59(7), 645-666.
AA Sandilya, U Natarajan and MH Priya. Molecular view into the cyclodextrin cavity: Structure and hydration. ACS Omega 2020; 5(40), 25655-25667.
T Ishiguro, F Hirayama, D Iohara, H Arima and K Uekama. Crystallization and polymorphic transitions of chlorpropamide in aqueous 2-hydroxybutyl-β-cyclodextrin solution. European Journal of Pharmaceutical Sciences 2010; 39(4), 248-255.
T Tao, Y Zhao, J Wu and B Zhou. Preparation and evaluation of itraconazole dihydrochloride for the solubility and dissolution rate enhancement. International Journal of Pharmaceutics 2009; 367(1-2), 109-114.
D Truyen, Phung, TV Dang and HV Huynh. Development and optimization of the synthesis of 2-hydroxybutyl-β-cyclodextrin. Vietnam Journals Online 2012; 52(10), 42-47.
TD Phung, TNK Le and ATP Phung. Synthesis and structural characterization of hydroxy butyl-β-cyclodextrin. Asian Journal of Chemistry 2025; 37(6),1282-1288.
M Malanga, M Bálint, I Puskás, T Sohajda, L Jicsinszky, L Szente and É Fenyvesi. Synthetic strategies for the fluorescent labeling of epichlorohydrin-branched cyclodextrin polymers. Beilstein Journal of Organic Chemistry 2014; 10, 3007-3018.
Y Kondo, S Suzuki, M Watanabe, A Kaneta, P Albertini and K Nagamori. Temperature-dependent circularly polarized luminescence measurement using KBr pellet method. Frontiers in Chemistry 2020; 8, 527.
FM Dayrit and ACD Dios. 1H and 13C NMR for the profiling of natural product extracts: Theory and applications. In: E Sharmin and F Zafar (Eds.). Spectroscopic analyses - developments and applications. IntechOpen, London, 2017.
AA Andrianova, T DiProspero, C Geib, IP Smoliakova, EI Kozliak and A Kubátová. Electrospray ionization with high-resolution mass spectrometry as a tool for lignomics: Lignin mass spectrum deconvolution. Journal of the American Society for Mass Spectrometry 2018; 29(5), 1044-1059.
HY Sun, Y Bai, MG Zhao, AY Hao, GY Xu, J Shen, JY Li, T Sun and HC Zhang. New cyclodextrin derivative 6-O-(2-hydroxybutyl)-β-cyclodextrin: Preparation and its application in molecular binding and recognition. Carbohydrate Research 2009; 344(15), 1999-2004.
J Pitha, CT Rao, B Lindberg and P Seffers. Distribution of substituents in 2-hydroxypropyl ethers of cyclomaltoheptaose. Carbohydrate Research 1990; 200, 429-435.
K Trainor, JA Palumbo, DWS MacKenzie and EM Meiering. Temperature dependence of NMR chemical shifts: Tracking and statistical analysis. Protein Science: A Publication of the Protein Society 2020; 29(1), 306-314.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






