In vitro Cultured Meat: Nutritional Aspects for the Health and Safety of Future Foods
DOI:
https://doi.org/10.48048/tis.2025.9060Keywords:
Cellular Agriculture, Alternative Protein, Nutrients, Future Foods, Serum Replacement, Food Safety, Cellular agriculture, Alternative protein, Nutrients, Future foods, Food safety, Serum replacementAbstract
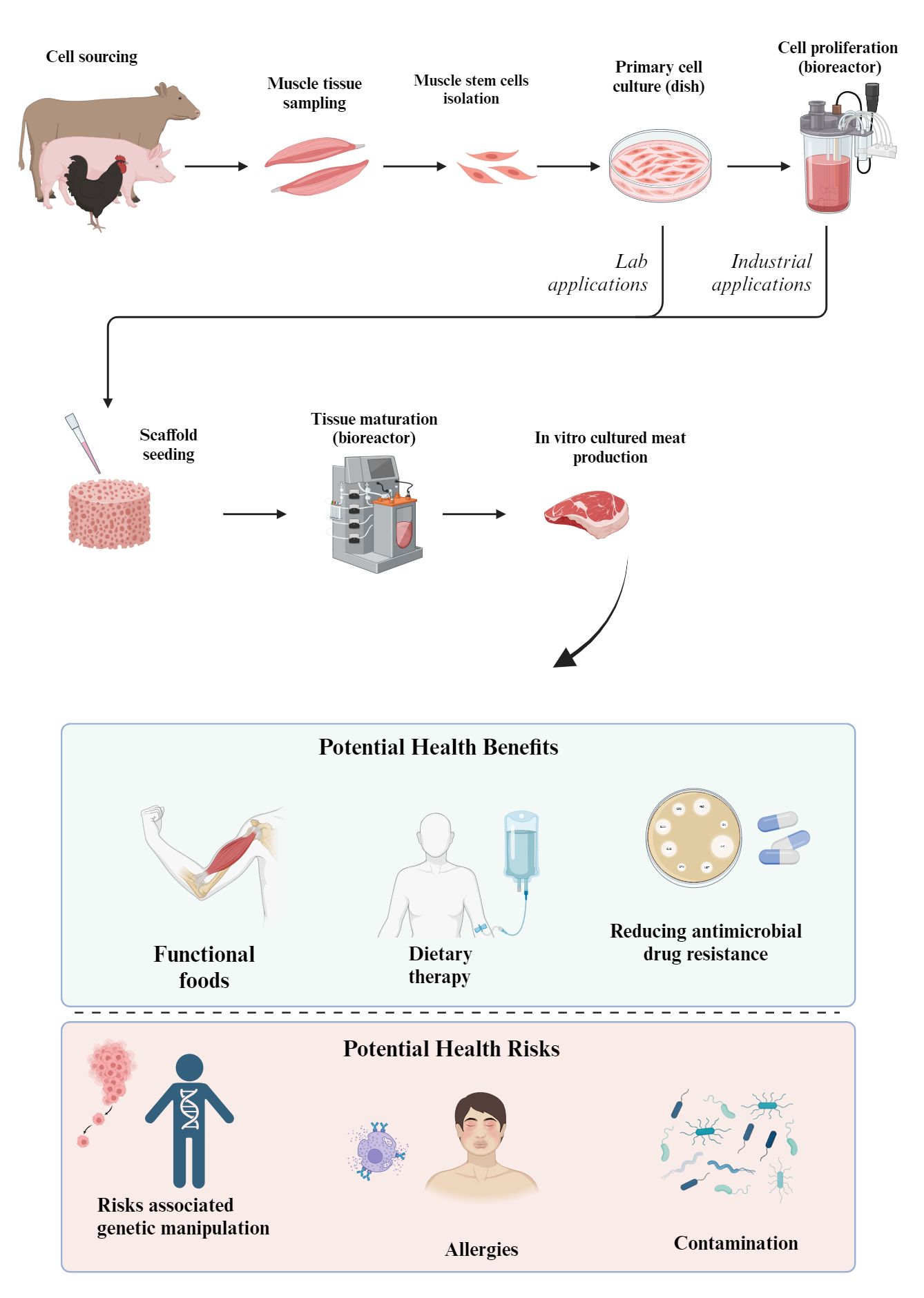
In vitro cultured meat is one of the future foods that might revolutionize meat production by allowing food manufacturers to sidestep traditional animal farming. Despite being in early development, in vitro cultured meat faces challenges such as technological limitations and consumer acceptance, hindering its market integration. Beyond being an ethical alternative to conventional meat, it resembles conventionally farmed meat, making it a focus on healthcare and food safety. From a health perspective, it theoretically presents intriguing possibilities in the fields of functional foods, tailored dietary therapy, and public health. In functional foods, its unique composition allows precise nutritional manipulation for specific dietary needs, aligning with personalized nutrition. In dietary therapy, in vitro cultured meat might offer customization for individuals with dietary restrictions or health conditions, providing a novel therapeutic avenue. From a public health standpoint, it holds promise in mitigating environmental and health challenges linked to conventional meat production, aligning with sustainability and disease prevention initiatives. However, the potential health risks of in vitro cultured meat include concerns about the use of genetically modified starting cells, fetal bovine serum, growth factors, scaffolding materials, and antibiotics during the production process. These additives may have unforeseen long-term health effects if not properly regulated. Additionally, in vitro cultured meat may lack some of the natural nutrients found in conventional meat, which could lead to nutritional imbalances. There are also uncertainties about how the texture and bioavailability of nutrients in in vitro cultured meat might affect digestion and absorption in the human body. Nevertheless, specific aspects of research in the field of in vitro cultured meat demand increased focus from researchers to guarantee the optimal safety standards. An in-depth study on safety-oriented research of in vitro cultured meat should be emphasized to ensure benefits for individual and public health.
HIGHLIGHTS
- In theory, in vitro cultured meat offers potential as a novel solution for developing functional foods by allowing precise manipulation of nutritional content, enabling the creation of health-optimized products.
- In vitro cultured meat could help mitigate the risk of antimicrobial resistance by minimizing the need for antimicrobial agents, reducing overall exposure and the potential for the development and spread of antimicrobial-resistant microorganisms.
- Starting cells derived from genetically modified cells in the in vitro cultured meat may pose risks of allergenicity and contamination, as well as potential immune responses to novel proteins.
- Strict safety protocols and thorough testing are crucial to address these risks and ensure the final product is safe for consumption.
GRAPHICAL ABSTRACT
Downloads
References
PH Howard. Cellular agriculture will reinforce power asymmetries in food systems. Nature Food 2022; 3(10), 798-800.
S Krongdang, P Phokasem, K Venkatachalam and N Charoenphun. Edible insects in Thailand: An overview of status, properties, processing, and utilization in the food industry. Foods 2023; 12(11), 2162.
H Rischer, GR Szilvay and KM Oksman-Caldentey. Cellular agriculture - industrial biotechnology for food and materials. Current Opinion in Biotechnology 2020; 61, 128-134.
L Yart, AW Wijaya, JAC Lima, C Haller, EM Beek, RS Carvalho, MRC Kraus and O Mashinchian. Cellular agriculture for milk bioactive production. Nature Reviews Bioengineering 2023; 1(11), 858-874.
X Guan, Q Lei, Q Yan, X Li, J Zhou, G Du and J Chen. Trends and ideas in technology. Future Foods 2021; 3, 100032.
S Miretti, I Manenti, P Toschi, E Macchi, E Martignani, P Accornero and M Baratta. Bovine skeletal muscle satellite cells: Isolation, growth, and differentiation. In: M Baratta (Ed.). Epithelial cell culture. Humana, New York, 2024, p. 165-174.
L Pasitka, M Cohen, A Ehrlich, B Gildor, M Ayyash, G Wissotsky, A Herscovici, R Kaminker, A Niv, R Bitcover, O Dadia, A Rudik, A Voloschin, M Shimoni, Y Cinnamon and Y Nahmias. Spontaneous immortalization of chicken fibroblasts generates stable, high-yield cell lines for serum-free production of cultured meat. Nature Food 2023; 4(1), 35-50.
J Reiss, S Robertson and M Suzuki. Cell sources for cultivated meat: Applications and considerations throughout the production workflow. International Journal of Molecular Sciences 2021; 22(14), 7513.
E Zehorai, A Maor-Shoshani, N Molotski, A Dorojkin, N Marelly, T Dvash and N Lavon. From fertilised oocyte to cultivated meat - harnessing bovine embryonic stem cells in the cultivated meat industry. Reproduction, Fertility and Development 2023; 36(2), 124-132.
G Zhu, D Gao, L Li, Y Yao, Y Wang, M Zhi, J Zhang, X Chen, Q Zhu, J Gao, T Chen, X Zhang, T Wang, S Cao, A Ma, X Feng and J Han. Generation of three-dimensional meat-like tissue from stable pig epiblast stem cells. Nature Communications 2023; 14(1), 8163.
JSH Seah, S Singh, LP Tan and D Choudhury. Scaffolds for the manufacture of cultured meat. Critical Reviews in Biotechnology 2022; 42(2), 311-323.
RI Tanaka, K Sakaguchi, A Yoshida, H Takahashi, Y Haraguchi and T Shimizu. Production of scaffold-free cell-based meat using cell sheet technology. NPJ Science of Food 2022; 6(1), 41.
DK Lee, M Kim, J Jeong, YS Lee, JW Yoon, MJ An, HY Jung, CH Kim, Y Ahn, KH Choi, C Jo and CK Lee. Unlocking the potential of stem cells: Their crucial role in the production of cultivated meat. Current Research in Food Science 2023; 7, 100551.
I Fraeye, M Kratka, H Vandenburgh and L Thorrez. Sensorial and nutritional aspects of cultured meat in comparison to traditional meat: Much to be inferred. Frontiers in Nutrition 2020; 7, 35.
GM Myers, KA Jaros, DS Andersen and DR Raman. Nutrient recovery in cultured meat systems: Impacts on cost and sustainability metrics. Frontiers in Nutrition 2023; 10, 1151801.
JF Hocquette, S Chriki, D Fournier and MP Ellies-Oury. Review: Will “cultured meat” transform our food system towards more sustainability? Animal 2024. https://doi.org/10.1016/j.animal.2024.101145
NR Rubio, N Xiang and DL Kaplan. Plant-based and cell-based approaches to meat production. Nature Communications 2020; 11(1), 6276.
MS Farvid, E Sidahmed, ND Spence, KM Angua, BA Rosner and JB Barnett. Consumption of red meat and processed meat and cancer incidence: A systematic review and meta-analysis of prospective studies. European Journal of Epidemiology 2021; 36(9), 937-951.
RJ Turesky. Mechanistic evidence for red meat and processed meat intake and cancer risk: A follow-up on the international agency for research on cancer evaluation of 2015. Chimia 2018; 72(10), 718-724.
I Benucci, C Lombardelli, C Mazzocchi and M Esti. Natural colorants from vegetable food waste: Recovery, regulatory aspects, and stability-A review. Comprehensive Reviews in Food Science and Food Safety 2022; 21(3), 2715-2737.
ED Salvo, GL Vecchio, RD Pasquale, LD Maria, R Tardugno, R Vadalà and N Cicero. Natural pigments production and their application in food, health and other industries. Nutrients 2023; 15(8), 1923.
S Chriki and JF Hocquette. The myth of cultured meat: A review. Frontiers in Nutrition 2020; 7, 7.
S Ong, L Loo, M Pang, R Tan, Y Teng, X Lou, SK Chin, MY Naik and H Yu. Decompartmentalisation as a simple color manipulation of plant-based marbling meat alternatives. Biomaterials 2021; 277, 121107.
CH Li, IH Yang, CJ Ke, CY Chi, J Matahum, CY Kuan, N Celikkin, W Swieszkowski and FH Lin. The production of fat-containing cultured meat by stacking aligned muscle layers and adipose layers formed from gelatin-soymilk scaffold. Frontiers in Bioengineering and Biotechnology 2022; 10, 875069.
FC Yen, J Glusac, S Levi, A Zernov, L Baruch, M Davidovich-Pinhas, A Fishman and M Machluf. Cultured meat platform developed through the structuring of edible microcarrier-derived microtissues with oleogel-based fat substitute. Nature Communications 2023; 14(1), 2942.
B Balasubramanian, W Liu, K Pushparaj and S Park. The epic of in vitro meat production - a fiction into reality. Foods 2021; 10(6), 1395.
M Henchion, M Hayes, AM Mullen, M Fenelon and B Tiwari. Future protein supply and demand: Strategies and factors influencing a sustainable equilibrium. Foods 2017; 6(7), 53.
M Zhou, Y Wei, Y Feng, S Zhang, N Ma, K Wang, P Tan, Y Zhao, J Zhao and X Ma. Arginine regulates skeletal muscle fiber type formation via mTOR signaling pathway. International Journal of Molecular Sciences 2024; 25(11), 6184.
AM Scholefield and KA Schuller. Cell proliferation and long chain polyunsaturated fatty acid metabolism in a cell line from southern bluefin tuna (Thunnus maccoyii). Lipids 2014; 49(7), 703-714.
M Taheri, PD Chilibeck and SM Cornish. A brief narrative review of the underlying mechanisms whereby omega-3 fatty acids may influence skeletal muscle: From cell culture to human interventions. Nutrients 2023; 15(13), 2926.
S Gutierrez, SL Svahn and ME Johansson. Effects of omega-3 fatty acids on immune cells. International journal of Molecular Sciences 2019; 20(20), 5028.
JJ Li, KF Dou, ZG Zhou, D Zhao, P Ye, JJ Zhao and LX Guo. Role of omega-3 fatty acids in the prevention and treatment of cardiovascular Diseases: A consensus statement from the Experts’ Committee Of National Society Of Cardiometabolic Medicine. Frontiers in Pharmacology 2022; 13, 1069992.
AJ Stout, AB Mirliani, EL Soule-Albridge, JM Cohen and DL Kaplan. Engineering carotenoid production in mammalian cells for nutritionally enhanced cell-cultured foods. Metabolic Engineering 2020; 62, 126-137.
EJ Johnson. The role of carotenoids in human health. Nutrition in Clinical Care 2002; 5(2), 56-65.
C Bomkamp, L Musgrove, DMC Marques, GF Fernando, FC Ferreira and EA Specht. Differentiation and maturation of muscle and fat cells in cultivated seafood: Lessons from developmental biology. Marine Biotechnology 2023; 25(1), 1-29.
S Levi, FC Yen, L Baruch and M Machluf. Scaffolding technologies for the engineering of cultured meat: Towards a safe, sustainable, and scalable production. Trends in Food Science & Technology 2022; 126(80), 13-25.
A Moeini, P Pedram, E Fattahi, P Cerruti and G Santagata. Edible polymers and secondary bioactive compounds for food packaging applications: Antimicrobial, mechanical, and gas barrier properties. Polymers 2022; 14(12), 2395.
Z Wei, S Dai, J Huang, X Hu, C Ge, X Zhang, K Yang, P Shao, P Sun and N Xiang. Soy protein amyloid fibril scaffold for cultivated meat application. ACS Applied Materials & Interfaces 2023; 15(12), 15108-15119.
N Xiang, Y Yao, JSK Yuen, AJ Stout, C Fennelly, R Sylvia, A Schnitzler, S Wong and DL Kaplan. Edible films for cultivated meat production. Biomaterials 2022; 287, 121659.
YY Zheng, Y Chen, HZ Zhu, CB Li, WJ Song, SJ Ding and GH Zhou. Production of cultured meat by culturing porcine smooth muscle cells in vitro with food grade peanut wire-drawing protein scaffold. Food Research International 2022; 159, 111561.
L Martinez, G Ros and G Nieto. Fe, Zn and Se bioavailability in chicken meat emulsions enriched with minerals, hydroxytyrosol and extra virgin olive oil as measured by Caco-2 cell model. Nutrients 2018; 10(8), 969.
DC Nwobodo, MC Ugwu, CO Anie, MTS Al-Ouqaili, JC Ikem, UV Chigozie and M Saki. Antibiotic resistance: The challenges and some emerging strategies for tackling a global menace. Journal of Clinical Laboratory Analysis 2022; 36(9), e24655.
TM Coque, R Canton, AE Perez-Cobas, MD Fernandez-de-Bobadilla and F Baquero. Antimicrobial resistance in the global health network: Known unknowns and challenges for efficient responses in the 21st century. Microorganisms 2023; 11(4), 1050.
World Health Organization. Food safety aspects of cell-based food. World Health Organization, Switzerland, 2023.
MG Bacanli. The two faces of antibiotics: An overview of the effects of antibiotic residues in foodstuffs. Archives of Toxicology 2024; 98(6), 1717-1725.
NA Dafale, S Srivastava and HJ Purohit. Zoonosis: An emerging link to antibiotic resistance under “one health approach”. Indian Journal of Microbiology 2020; 60(2), 139-152.
MN Hayek. The infectious disease trap of animal agriculture. Science Advances 2022; 8(44), eadd6681.
WJ Underwood, R Blauwiekel, M Delano, R Gillesby, S Mischler and A Schoell. Biology and diseases of ruminants (sheep, goats, and cattle). In: JG Fox, GM Otto, MT Whary, LC Anderson and KR Pritchett-Corning (Eds.). Laboratory animal medicine. Academic Press, Massachusetts, 2015, p. 623-694.
S Manzoor, Z Syed and M Abubabakar. Global perspectives of intensive animal farming and its applications. In: S Manzoor and M Abubakar (Eds.). Intensive animal farming - a cost-effective tactic. IntechOpen, London, 2023.
World Health Organization. Stop using antibiotics in healthy animals to prevent the spread of antibiotic resistance. World Health Organization, Switzerland, 2024.
R Helliwell, C Morris and S Raman. Antibiotic stewardship and its implications for agricultural animal-human relationships: Insights from an intensive dairy farm in England. Journal of Rural Studies 2020; 78, 447-456.
MJ Martin, SE Thottathil and TB Newman. Antibiotics overuse in animal agriculture: A call to action for health care providers. American Journal of Public Health 2015; 105(12), 2409-2410.
B Clark, LA Panzone, GB Stewart, I Kyriazakis, JK Niemi, T Latvala, R Tranter, P Jones and LJ Frewer. Consumer attitudes towards production diseases in intensive production systems. PLoS One 2019; 14(1), e0210432.
PW Barone, ME Wiebe, JC Leung, ITM Hussein, FJ Keumurian, J Bouressa, A Brussel, D Chen, M Chong, H Dehghani, L Gerentes, J Gilbert, D Gold, R Kiss, TR Kreil, R Labatut, Y Li, J Müllberg, L Mallet, C Menzel, …, SL Springs. Viral contamination in biologic manufacture and implications for emerging therapies. Nature Biotechnology 2020; 38(5), 563-572.
F Cobo, JL Cortes, C Cabrera, A Nieto and A Concha. Microbiological contamination in stem cell cultures. Cell Biology International 2007; 31(9), 991-995.
M Janghorban, S Kazemi, R Tormon, PN Makobore and R Pandey. Methods and analysis of biological contaminants in the biomanufacturing industry. Chemosensors 2023; 11(5), 298.
I Kuhlmann. The prophylactic use of antibiotics in cell culture. Cytotechnology 1995; 19(2), 95-105.
A Skubis, J Gola, B Sikora, J Hybiak, M Paul-Samojedny, U Mazurek and MJ Łos. Impact of antibiotics on the proliferation and differentiation of human adipose-derived mesenchymal stem cells. International Journal of Molecular Sciences 2017; 18(12), 2522.
S Weiskirchen, SK Schroder, EM Buhl and R Weiskirchen. A beginner’s guide to cell culture: Practical advice for preventing needless problems. Cells 2023; 12(5), 682.
L Nikfarjam and P Farzaneh. Prevention and detection of Mycoplasma contamination in cell culture. Cell Journal 2012; 13(4), 203-212.
K Wehbe, M Vezzalini and G Cinque. Detection of mycoplasma in contaminated mammalian cell culture using FTIR microspectroscopy. Analytical and Bioanalytical Chemistry 2018; 410(12), 3003-3016.
OW Merten. Virus contaminations of cell cultures - a biotechnological view. Cytotechnology 2002; 39(2), 91-116.
AM Kolkmann, MJ Post, MAM Rutjens, ALM van Essen and P Moutsatsou. Serum-free media for the growth of primary bovine myoblasts. Cytotechnology 2020; 72(1), 111-120.
S Gangaram, Y Naidoo, YH Dewir, M Singh, J Lin and HN Murthy. Phytochemical composition and antibacterial activity of barleria albostellata C.B. clarke leaf and stem extracts. Plants 2023; 12(13), 2396.
N Nik Mohamad Nek Rahimi, I Natrah, JY Loh, FKE Ranzil, M Gina, SHE Lim, KS Lai and CM Chong. Phytocompounds as an alternative antimicrobial approach in aquaculture. Antibiotics 2022; 11(4), 469.
P Sharma, J Kaur, G Sharma and P Kashyap. Plant derived antimicrobial peptides: Mechanism of target, isolation techniques, sources and pharmaceutical applications. Journal of Food Biochemistry 2022; 46(10), e14348.
MM Cowan. Plant products as antimicrobial agents. Clinical Microbiology Reviews 1999; 12(4), 564-582.
HA Hemeg, IM Moussa, S Ibrahim, TM Dawoud, JH Alhaji, AS Mubarak, SA Kabli, RA Alsubki, AM Tawfik and SA Marouf. Antimicrobial effect of different herbal plant extracts against different microbial population. Saudi Journal of Biological Sciences 2020; 27(12), 3221-3227.
U Friedlein, S Dorn-In and K Schwaiger. Antimicrobial effects of plant extracts against clostridium perfringens with respect to food-relevant influencing factors. Journal of Food Protection 2021; 84(10), 1809-1818.
ML Heymich, U Friedlein, M Trollmann, K Schwaiger, RA Bockmann and M Pischetsrieder. Generation of antimicrobial peptides Leg1 and Leg2 from chickpea storage protein, active against food spoilage bacteria and foodborne pathogens. Food Chemistry 2021; 347, 128917.
S Manandhar, S Luitel and RK Dahal. In vitro antimicrobial activity of some medicinal plants against human pathogenic bacteria. Journal of Tropical Medicine 2019; 2019, 1895340.
YM Cheng, PC Hong, MM Song, HN Zhu, J Qin, ZD Zhang, H Chen, XZ Ma, MY Tian, WY Zhu and Z Huang. An immortal porcine preadipocyte cell strain for efficient production of cell-cultured fat. Communications Biology 2023; 6, 1202.
R Verma, Y Lee and DF Salamone. iPSC technology: An innovative tool for developing clean meat, livestock, and frozen ark. Animals 2022; 12(22), 3187.
M Lee, S Park, B Choi, W Choi, H Lee, JM Lee, ST Lee, KH Yoo, D Han, G Bang, H Hwang, WG Koh, S Lee and J Hong. Cultured meat with enriched organoleptic properties by regulating cell differentiation. Nature Communications 2024; 15, 77.
MJ Post. Cultured meat from stem cells: Challenges and prospects. Meat Science 2012; 92(3), 297-301.
P Weeratunga, RM Harman and GR van de Walle. Induced pluripotent stem cells from domesticated ruminants and their potential for enhancing livestock production. Frontiers in Veterinary Science 2023; 10, 1129287.
K Wuputra, CC Ku, DC Wu, YC Lin, S Saito and KK Yokoyama. Prevention of tumor risk associated with the reprogramming of human pluripotent stem cells. Journal of Experimental & Clinical Cancer Research 2020; 39, 100.
CT Charlesworth, I Hsu, AC Wilkinson and H Nakauchi. Immunological barriers to haematopoietic stem cell gene therapy. Nature Reviews Immunology 2022; 22(12), 719-733.
JI Pearl, LS Kean, MM Davis and JC Wu. Pluripotent stem cells: Immune to the immune system? Science Translational Medicine 2012; 4(164), 164ps25.
L Ketelings, S Kremers and A de Boer. The barriers and drivers of a safe market introduction of cultured meat: A qualitative study. Food Control 2021; 130(4), 108299.
P Hanlon and V Sewalt. GEMs: Genetically engineered microorganisms and the regulatory oversight of their uses in modern food production. Critical Reviews in Food Science and Nutrition 2021; 61(6), 959-970.
GS Ladics. Assessment of the potential allergenicity of genetically-engineered food crops. Journal of Immunotoxicology 2019; 16(1), 43-53.
EFSA Panel on Genetically Modified Organisms, H Naegeli, JL Bresson, T Dalmay, IC Dewhurst, MM Epstein, LG Firbank, P Guerche, J Hejatko, FJ Moreno, E Mullins, F Nogué, N Rostoks, JJS Serrano, G Savoini, E Veromann, F Veronesi, F Álvarez, M Ardizzone, AF Dumont, …, K Paraskevopoulos. Assessment of genetically modified soybean SYHT0H2 for food and feed uses, import and processing, under Regulation (EC) No 1829/2003 (application EFSA-GMO-DE-2012-111). EFSA Journal 2020; 18(1), e05946.
JD Macdougall, KO Thomas and OI Iweala. The meat of the matter: Understanding and managing alpha-gal syndrome. ImmunoTargets and Therapy 2022; 11, 37-54.
AS Carson, A Gardner and OI Iweala. Where’s the beef ? Understanding allergic responses to red meat in alpha-gal syndrome. The Journal of Immunology 2022; 208(2), 267-277.
SP Commins. Diagnosis & management of alpha-gal syndrome: Lessons from 2,500 patients. Expert Review of Clinical Immunology 2020; 16(7), 667-677.
D Brunner, J Frank, H Appl, H Schoffl, W Pfaller and G Gstraunthaler. Serum-free cell culture: The serum-free media interactive online database. Altex 2010; 27(1), 53-62.
CR Pilgrim, KA McCahill, JG Rops, JM Dufour, KA Russell and TG Koch. A review of fetal bovine serum in the culture of mesenchymal stromal cells and potential alternatives for veterinary medicine. Frontiers in Veterinary Science 2022; 9, 859025.
JVD Valk, D Brunner, KD Smet, AF Svenningsen, P Honegger, LE Knudsen, T Lindl, J Noraberg, A Price, ML Scarino and G Gstraunthaler. Optimization of chemically defined cell culture media - replacing fetal bovine serum in mammalian in vitro methods. Toxicology in Vitro 2010; 24(4), 1053-1063.
M Chruszcz, K Mikolajczak, N Mank, KA Majorek, PJ Porebski and W Minor. Serum albumins-unusual allergens. Biochimica et Biophysica Acta (BBA)-General Subjects 2013; 1830(12), 5375-5381.
LH Ulfman, JHW Leusen, HFJ Savelkoul, JO Warner and RJJV Neerven. Effects of bovine immunoglobulins on immune function, allergy, and infection. Frontiers in Nutrition 2018; 5, 52.
RD Silva, W Dasanayake, GD Wickramasinhe, C Karunatilake, N Weerasinghe, P Gunasekera and GN Malavige. Sensitization to bovine serum albumin as a possible cause of allergic reactions to vaccines. Vaccine 2017; 35(11), 1494-1500.
J Wang and HA Sampson. Food allergy: Recent advances in pathophysiology and treatment. Allergy, Asthma & Immunology 2009; 1(1), 19-29.
AJ Stout, AB Mirliani, ML Rittenberg, M Shub, EC White, JSK Yuen and DL Kaplan. Simple and effective serum-free medium for sustained expansion of bovine satellite cells for cell cultured meat. Communications Biology 2022; 5, 466.
A Singh, V Kumar, SK Singh, J Gupta, M Kumar, DK Sarma and V Verma. Recent advances in bioengineered scaffold for in vitro meat production. Cell and Tissue Research 2023; 391(2), 235-247.
P Kumar, N Sharma, S Sharma, N Mehta, AK Verma, S Chemmalar and AQ Sazili. In-vitro meat: A promising solution for sustainability of meat sector. Journal of Animal Science and Technology 2021; 63(4), 693-724.
F Bajerski, M Nagel and J Overmann. Microbial occurrence in liquid nitrogen storage tanks: A challenge for cryobanking? Applied Microbiology and Biotechnology 2021; 105(20), 7635-7650.
F Houston and O Andreoletti. Animal prion diseases: The risks to human health. Brain Pathology 2019; 29(2), 248-262.
F Houston, S McCutcheon, W Goldmann, A Chong, J Foster, S Siso, L González, M Jeffrey and N Hunter. Prion diseases are efficiently transmitted by blood transfusion in sheep. Blood, The Journal of the American Society of Hematology 2008; 112(12), 4739-4745.
MR Scott, R Will, J Ironside, HOB Nguyen, P Tremblay, SJ DeArmond and SB Prusiner. Compelling transgenetic evidence for transmission of bovine spongiform encephalopathy prions to humans. Proceedings of the National Academy of Sciences 1999; 96(26), 15137-15142.
H Hara, J Chida, K Uchiyama, AD Pasiana, E Takahashi, H Kido and S Sakaguchi. Neurotropic influenza A virus infection causes prion protein misfolding into infectious prions in neuroblastoma cells. Scientific Reports 2021; 11, 10109.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






