Cardiospermum halicacabum Extract Inhibits the Production of Nitric Oxide and Reactive Oxygen Species in LPS-Stimulated RAW 264.7 Cells through Erk/p38 Signalling Pathways
DOI:
https://doi.org/10.48048/tis.2024.8690Keywords:
Anti-inflammation, Cardiospermum halicacabum, Nitric oxide, Reactive oxygen species, Erk1/2, NF-BAbstract
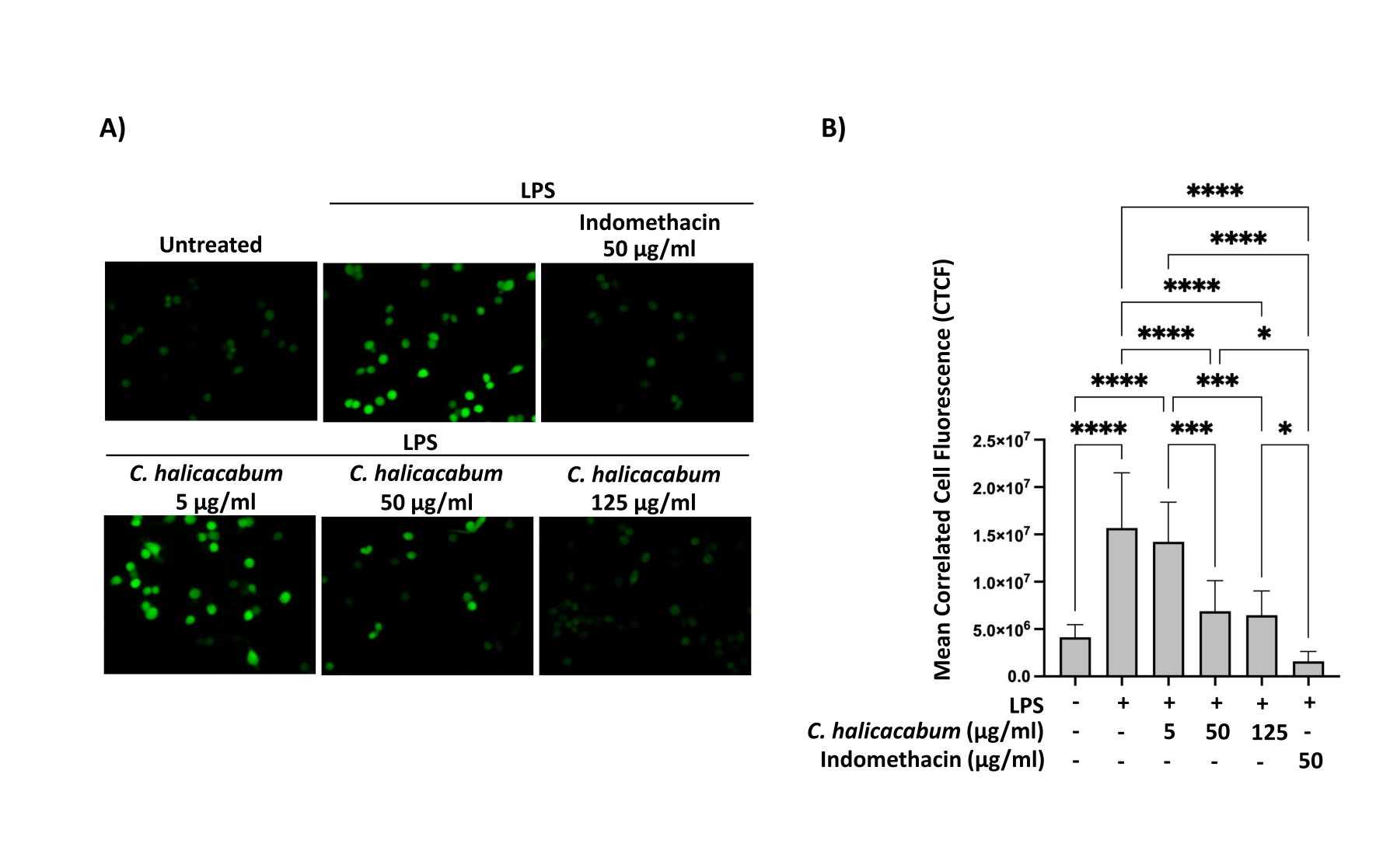
The medicinal plant Cardiospermum halicacabum has been extensively studied for its potential in various therapeutic activities, including anti-inflammation, antioxidant, antibacterial, antifungal and antiparasitic effects. However, the specific molecular mechanisms responsible for its anti-inflammatory properties remain largely unknown. In order to shed light on this aspect, the current investigation was aimed at elucidating the underlying mechanisms through which C. halicacabum exerts its anti-inflammatory response. The different concentrations of C. halicacabum extract were tested for cytotoxicity on RAW264.6 murine macrophage cells by MTT assay. The inhibition of nitric oxide (NO) in LPS-induced cell pre-treated with the extract was evaluated by Griess assay using Griess reagent. Flow cytometry and fluorescent microscopy were used to examine the reactive oxygen species (ROS) in the treated cells. The expression of iNOS and the phosphorylation status of Erk1/2, p38 and NF-kB were determined by western blotting. The C. halicacabum extract at concentrations up to 125 µg/mL did not cause the cytotoxicity to the RAW264.6 murine macrophage cells. Incubation of LPS-induced cells with the extract significantly minimized the production of NO as compared with the LPS-stimulated cells. Further, the extract significantly reduced ROS generation in LPS-induced cells. In the present of C. halicacabum, LPS-treated cells had the reduced NOS expression as well as had low phosphorylation on Erk1/2, p38 and NF-kB. C. halicacabum attenuates NO and ROS production under the regulation of Erk1/2 and p38 pathway as well as NF-kB.
HIGHLIGHTS
- The underlying mechanisms of halicacabum on its anti-inflammatory response were investigated.
- halicacabum extract significantly reduced ROS and NO generation in LPS-induced cells.
- halicacabum attenuates NO and ROS production under the regulation of Erk1/2 and p38 pathway as well as NF-kB.
GRAPHICAL ABSTRACT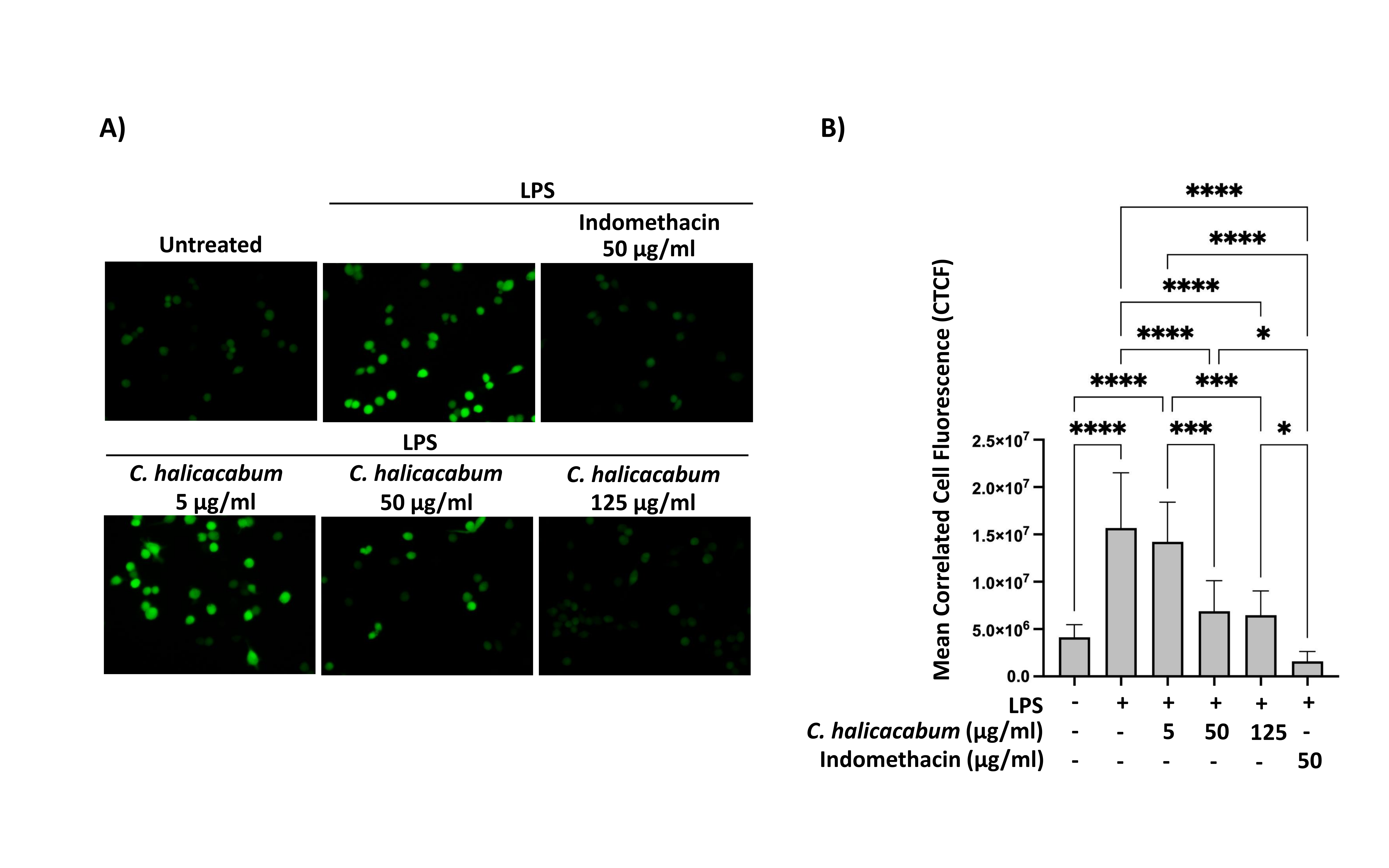
Downloads
References
F Menichini, L Losi, M Bonesi, A Pugliese, MR Loizzo and R Tundis. Chemical profiling and in vitro biological effects of Cardiospermum halicacabum L. (Sapindaceae) aerial parts and seeds for applications in neurodegenerative disorders. Journal of Enzyme Inhibition and Medicinal Chemistry 2014; 29(5), 677-685.
BKC Venkatesh and S Krishnakumari. Cardiospermum halicacabum suppresses the production of TNF-alpha and Nitric oxide by Human Peripheral Blood Mononuclear cells. African Journal of Biomedical Research 2006; 9(2), 95-99.
MM Shabi, R Dhevi, K Gayathri, U Subashini, GV Rajamanickam and GP Dubey. C. halicacabum (Linn): Investigations on anti-inflammatory and analgesic effect. Bulgarian Journal of Veterinary Medicine 2009; 12(3), 171-177.
K Mruthunjaya, AP Suresh, N Paramakrishnan and M Basavaraju. A comprehensive review on Cardiospermum halicacabum. Journal of Natural Remedies 2023; 23(2), 283-293.
A Elangovan, J Ramachandran, DK Lakshmanan, G Ravichandran and S Thilagar. Ethnomedical, phytochemical and pharmacological insights on an Indian medicinal plant: The balloon vine (Cardiospermum halicacabum Linn.). Journal of Ethnopharmacology 2022; 291, 115143.
M Król and M Kepinska. Human nitric oxide synthase - Its functions, polymorphisms, and inhibitors in the context of inflammation, diabetes and cardiovascular diseases. International Journal of Molecular Sciences 2021; 22(1), 56.
L Selloum, H Bouriche, C Tigrine and C Boudoukha. Anti-inflammatory effect of rutin on rat paw oedema, and on neutrophils chemotaxis and degranulation. Experimental and Toxicologic Pathology 2003; 54, 313-318.
AA Krishna, BL Abhirami, MB Bashi, R Jaice, AR Jasim and A Kumaran. Identification and anti-inflammatory activity of flavonoids from Cardiospermum halicacabum in murine RAW 264.7 macrophage-A bioassay-guided approach. Food Bioscience 2024; 61, 104510.
MH Huang, SS Huang, BS Wang, CH Wu, MJ Sheu, WC Hou, SS Lin and GJ Huang. Antioxidant and anti-inflammatory properties of Cardiospermum halicacabum and its reference compounds ex vivo and in vivo. Journal of Ethnopharmacology 2011; 133(2), 743-750.
SM Kumar, V Jiju and D Chamundeeswari. Investigation of phytoconstituents of Cardiospermum halicacabum and its efficacy as a potential anti-cancer drug candidate. Journal of Drug Delivery and Therapeutics 2019; 9, 252-257.
HY Hsu and MH Wen. Lipopolysaccharide-mediated reactive oxygen species and signal transduction in the regulation of interleukin-1 gene expression. Journal of Biological Chemistry. 2002; 277(25), 22131-22139.
SH Yang, AD Sharrocks and AJ Whitmarsh. MAP kinase signalling cascades and transcriptional regulation. Gene 2013; 513(1), 1-13.
S Papi, F Ahmadizar and A Hasanvand. The role of nitric oxide in inflammation and oxidative stress. Immunopathologia Persa 2019; 5(1), e08.
J Taira, H Nanbu and K Ueda. Nitric oxide-scavenging compounds in Agrimonia Pilosa Ledeb on LPS-induced RAW264.7 macrophages. Food Chemistry 2009; 115(4), 1221-1227.
T Joo, K Sowndhararajan, S Hong, J Lee, SY Park, S Kim and JW Jhoo. Inhibition of nitric oxide production in LPS-stimulated RAW 264.7 cells by stem bark of Ulmus pumila L. Saudi Journal of Biological Sciences 2014; 21, 427-435.
M Canton, R Sánchez-Rodríguez, I Spera, FC Venegas, M Favia, A Viola and A Castegna. Reactive oxygen species in macrophages: Sources and targets. Frontiers in Immunology 2021; 12, 734229.
H Sies and DP Jones. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nature Reviews Molecular Cell Biology 2020; 21, 363-383.
SH Baek, T Park, MG Kang and D Park. Anti-inflammatory activity and ROS regulation effect of Sinapaldehyde in LPS-stimulated RAW 264.7 macrophages. Molecules 2020; 25(18), 4089.
J Liu, X Han, T Zhang, K Tian, Z Li and F Luo. Reactive oxygen species (ROS) scavenging biomaterials for anti-inflammatory diseases: From mechanism to therapy. Journal of Hematology & Oncology 2023; 16, 116.
HJ Jeon, HS Choi, OH Lee, YJ Jeon and BY Lee. Inhibition of Reactive Oxygen Species (ROS) and Nitric Oxide (NO) by gelidium elegans using alternative drying and extraction conditions in 3T3-L1 and RAW 264.7 cells. Preventive Nutrition and Food Science 2012; 17(2), 122-8.
T Lawrence. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harbor Perspectives in Biology 2009; 1, a00165.
L Barnabei, E Laplantine, W Mbongo, F Rieux-Laucat and R Weil. NF-κB: At the borders of autoimmunity and inflammation. Frontiers in Immunology 2021; 12, 716469.
M Herb, A Gluschko, K Wiegmann, A Farid, A Wolf, O Utermöhlen, O Krut, M Krönke and M Schramm. Mitochondrial reactive oxygen species enable proinflammatory signaling through disulfide linkage of NEMO. Science Signaling 2019; 12, eaar5926.
T Liu, L Zhang, D Joo and SC Sun. NF-κB signaling in inflammation. Signal Transduction Targeted Ther. 2017; 2(568), 17023.
D Arias-Salvatierra, EK Silbergeld, LC Acosta-Saavedra and ES Calderon-Aranda. Role of nitric oxide produced by iNOS through NF-κB pathway in migration of cerebellar granule neurons induced by Lipopolysaccharide. Cellular Signalling 2011; 23, 425-435.
Y Lim, JW Park, OK Kwon, JW Lee, HS Lee, S Lee, S Choi, W Li, H Jin, SB Han and KS Ahn. Anti-inflammatory effects of a methanolic extract of Castanea seguinii Dode in LPS-induced RAW264.7 macrophage cells. International Journal of Molecular Medicine. 2017; 41, 391-398.
M Mittal, MR Siddiqui, K Tran, SP Reddy and AB Malik. Reactive oxygen species in inflammation and tissue injury. Antioxidants & Redox Signaling 2014; 20(7), 1126-1167.
G Shanker, JL Aschner, T Syversen and M Aschner. Free radical formation in cerebral cortical astrocytes in culture induced by methylmercury. Molecular Brain Research 2004; 128(1), 48-57.
K Bode, C Link, PH Krammer and H Weyd. Flow-cytometric detection of low-level reactive oxygen species in cell lines and primary immune cells. Bio-protocol 2020; 10, e3737.
ED Chan and DW Riches. IFN-gamma + LPS induction of iNOS is modulated by ERK, JNK/SAPK, and p38(mapk) in a mouse macrophage cell line. American Journal of Physiology-Cell Physiology 2001; 280, C441-C450.
KCV Babu and S Krishnakumari. Anti-inflammatory and antioxidant compound, rutin in Cardiospermum halicacabum leaves. Ancient Science of Life 2005; 25, 47-49.
MS Sheeba and VV Asha. Cardiospermum halicacabum ethanol extract inhibits LPS induced COX-2, TNF-alpha and iNOS expression, which is mediated by NF-kappaB regulation, in RAW264.7 cells. Journal of Ethnopharmacology. 2009; 124(1), 39-44.
F Aktan. iNOS-mediated nitric oxide production and its regulation. Life Sciences 2004; 75(6), 639-653.
K Chang, SJ Lee, I Cheong, TR Billiar, HT Chung, JA Han, YG Kwon, KS Ha and YM Kim. Nitric oxide suppresses inducible nitric oxide synthase expression by inhibiting post-translational modification of IκB. Experimental & Molecular Medicine 2004; 36, 311-324.
ASJ Baldwin. The NF-kappa B and I kappa B proteins: New discoveries and insights. Annual Review of Immunology 1996; 14, 649-683.
HCF Oliveira, RG Cosso, LC Alberici, EN Maciel, AG Salerno, GG Dorighello, JA Velho, ECD Faria and AE Vercesi. Oxidative stress in atherosclerosis-prone mouse is due to low antioxidant capacity of mitochondria. The FASEB Journal 2005; 19(2), 278-280.
C Nathan and A Cunningham-Bussel. Beyond oxidative stress: An immunologist’s guide to reactive oxygen species. Nature Reviews Immunology 2013; 13, 349-361.
S Sanlioglu, CM Williams, L Samavati, NS Butler, G Wang, PBJ McCray, TC Ritchie, GW Hunninghake, E Zandi and JF Engelhardt. Lipopolysaccharide induces Rac1-dependent reactive oxygen species formation and coordinates tumor necrosis factor-α secretion through IKK regulation of NF-κB. Journal of Biological Chemistry 2001; 276(32), 30188-30198.
X Wu, H Gao, Y Hou, J Yu, W Sun, Y Wang, X Chen, Y Feng, QM Xu and X Chen. Dihydronortanshinone, a natural product, alleviates LPS-induced inflammatory response through NF-κB, mitochondrial ROS, and MAPK pathways. Toxicology and Applied Pharmacology 2018; 355, 1-8.
R Craig, A Larkin, AM Mingo, DJ Thuerauf, C Andrews, PM McDonough and CC Glembotski. p38 MAPK and NF-kappa B collaborate to induce interleukin-6 gene expression and release: Evidence for a cytoprotective autocrine signaling pathway in a cardiac myocyte model system. Journal of Biological Chemistry 2000; 275, 23814-23824.
WV Berghe, S Plaisance, E Boone, K D Bosscher, ML Schmitz, W Fiers and G Haegeman. p38 and extracellular signal-regulated kinase mitogen activate protein kinase pathways are required for nuclear factor-B p65 transactivation mediated by tumor necrosis factor. Journal of Biological Chemistry 1998; 273(6), 3285-3290.
MJ Morgan and ZG Liu. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Research 2011; 21, 103-115.
T Meng, J Yu, Z Lei, J Wu, S Wang, Q Bo, X Zhang, Z Ma and J Yu. Propofol reduces lipopolysaccharide-induced, NADPH oxidase (NOX2) mediated TNF-α and IL-6 production in macrophages. Journal of Immunology Research 2013; 2013, 325481.
S Merighi, A Travagli, P Tedeschi, N Marchetti and S Gessi. Antioxidant and antiinflammatory effects of Epilobium parviflorum, Melilotus officinalis and Cardiospermum halicacabum plant extracts in macrophage and microglial cells. Cells 2021; 10(10), 2691.
Y Chen, ZX Zhang, LP Zheng, L Wang, YF Liu, WY Yin, YY Chen, XS Wang, ST Hou, JF Chen and RY Zheng. The adenosine A(2A) receptor antagonist SCH58261 reduces macrophage/ microglia activation and protects against experimental autoimmune encephalomyelitis in mice. Neurochemistry International 2019; 129, 104490.
S Thakur, J Du, S Hourani, C Ledent and JM Li. Inactivation of adenosine A2A receptor attenuates basal and angiotensin II-induced ROS production by Nox2 in endothelial cells. Journal of Biological Chemistry 2010; 285(51), 40104-40113.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






