Optimization of Polyphenol Extraction Conditions from Rhizomes of Curcuma zedoaria with Antioxidant, Anti-Inflammatory and Anti-Diabetic Activities In Vitro
DOI:
https://doi.org/10.48048/tis.2024.8544Keywords:
Anti-diabetic, Anti-inflammatory, Antioxidant, Curcuma zedoaria rhizomes, Extract, Response surface methodology, PolyphenolAbstract
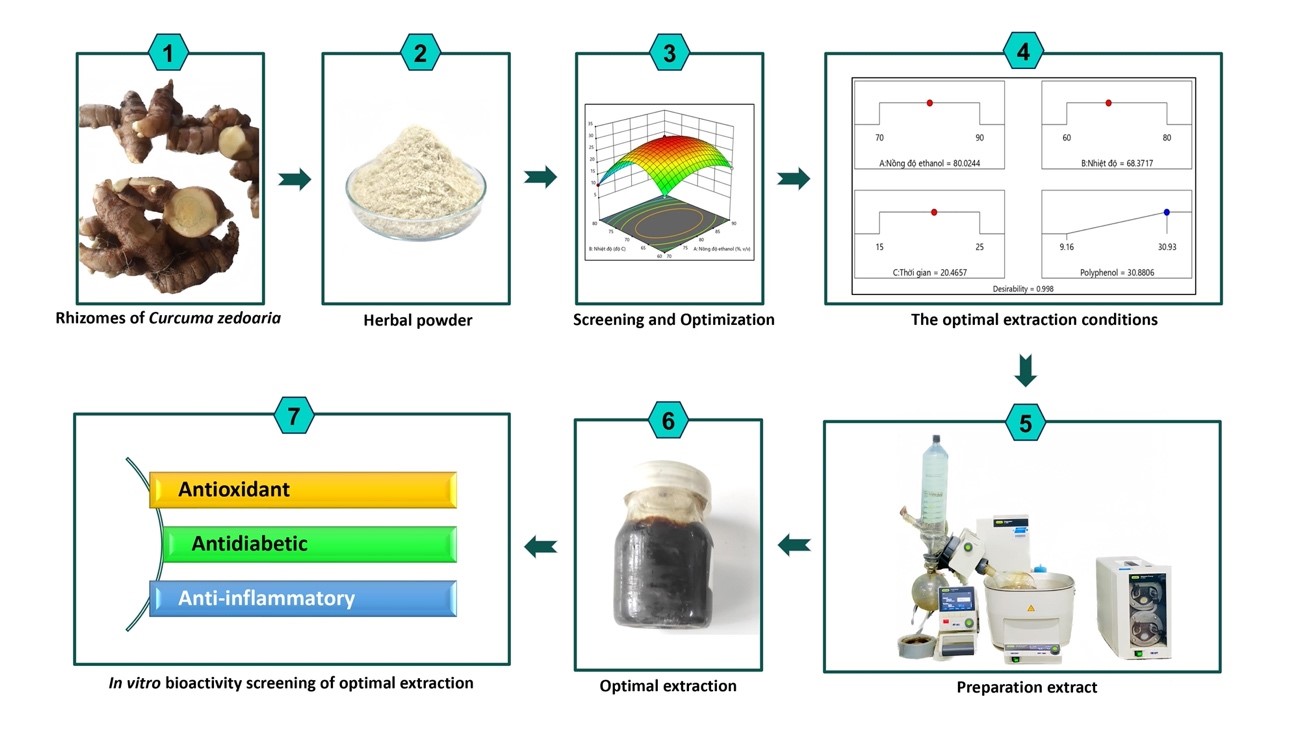
The recovery of polyphenol compounds is seen as an arduous task because polyphenol compounds are available as free aglycones, as sugar or ester conjugates, or as polymers with several monomeric components. The response surface method (RSM) as a tool to optimize the factors that affect extraction efficacy as well as to obtain maximum recovery of the compounds of interest. This study optimizes ultrasound-assisted polyphenol extraction from Curcuma zedoaria rhizomes using response surface methodology (RSM). Three variables were considered: Ethanol concentration (70 - 90 %, v/v), temperature (60 - 80 °C), and ultrasound time (15 - 25 min). Data were analyzed by ANOVA, yielding an R2 of 0.9993, a significant interaction effect (p < 0.0001), and an insignificant lack-of-fit test (p = 0.6684). Optimal conditions for maximum polyphenol content (TPC = 31.05 ± 0.53 mg GAE/g powder) were 80.02 % ethanol, 68 °C, 20.47 min ultrasound time, and 1/10 (w/v) raw material/solvent ratio. Experimental values matched RSM predictions, confirming successful extraction optimization from Curcuma zedoaria rhizomes. The polyphenol-rich optimum extract from Curcuma zedoaria rhizomes has been studied for its antioxidant, anti-inflammatory and anti-inflammatory properties in vitro. The results were found, the optimum extract of Curcuma zedoaria rhizomes could perform effective neutralization activities of free radicals performed in DPPH test (IC50 = 5.89 ± 0.23 µg/mL), NO· (IC50 = 5.89 ± 0.23 µg/mL) and ABTS·+ (IC50 = 8.28 ± 0.12 µg/mL). Besides, this optimum extract had the ability to protect red blood cell membranes and inhibit protein denaturation due to heat with IC50 times values such as 29.06 ± 0.35 and 30.24 ± 0.32 µg/mL. In addition, it also significantly inhibited α-amylase, α-glucosidase enzyme activities with IC50 values of 5.89 ± 0.23 and 9.62 ± 0.11 μg/mL, respectively. This investigation showed that the polyphenol-rich optimum extract from Curcuma zedoaria rhizomes was a promising antioxidant, anti-inflammatory and anti-diabetic agent.
HIGHLIGHTS
- Using ultrasound and the Box-Behnken design to extract polyphenols from Curcuma zedoaria rhizomes can speed up and improve extraction efficiency.
- The goal of this study is to maximize the extraction parameters for polyphenols from the rhizomes of Curcuma zedoaria.
- There is strong agreement between the experimental data and the model that predicts the polyphenol content in the Box-Behnken design.
- In vitro biological activities such as anti-inflammatory, antidiabetic and antioxidant properties are significantly correlated with polyphenol concentration.
- The findings of our investigation show a noteworthy enhancement in the bioavailability of polyphenol compounds through increased extraction efficiency and bioactivity.
GRAPHICAL ABSTRACT

Downloads
References
YE Hadisaputri, T Miyazaki, S Suzuki, N Kubo, A Zuhrotun, T Yokobori, R Abdulah, S Yazawa and H Kuwano. Molecular characterization of antitumor effects of the rhizome extract from Curcuma zedoaria on human esophageal carcinoma cells. Int. J. Oncol. 2015; 47, 2255-63.
EB Jung, TA Trinh, TA Lee, N Yamabe, KS Kang, JH Song, S Choi, S Lee, TS Jang and KH Kim. Curcuzedoalide contributes to the cytotoxicity of Curcuma zedoaria rhizomes against human gastric cancer AGS cells through induction of apoptosis. J. Ethnopharmacol. 2018; 213, 48-55.
S Mou, Z Zhou, Y He, F Liu and L Gong. Curcumin inhibits cell proliferation and promotes apoptosis of laryngeal cancer cells through Bcl-2 and PI3K/Akt, and by upregulating miR-15a. Oncol. Lett. 2017; 14, 4937-42.
S Tariq, M Imran, Z Mushtaq and N Asghar. Phytopreventive antihypercholesterolmic and antilipidemic perspectives of zedoary (Curcuma Zedoaria Roscoe.) herbal tea. Lipids Health Dis. 2016; 15, 39.
J Akter, MA Hossain, K Takara, MZ Islam and DX Hou. Antioxidant activity of different species and varieties of turmeric (Curcuma spp): Isolation of active compounds. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019; 215, 9-17.
J Pérez-Jiméne, V Neveu, F Vos and A Scalbert. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010; 64, S112-S120.
A Singh, S Holvoet and A Mercenier. Dietary polyphenols in the prevention and treatment of allergic diseases. Clin. Exp. Allergy 2011; 41, 1346-59.
GEP Box and KB Wilson. On the experimental attainment of optimum conditions. In: S Kotz and NL Johnson (Eds.). Breakthroughs in statistics. Springer, New York, 1992, p. 270-310.
C Agarwal, K Máthé, T Hofmann and L Csóka. Ultrasound‐assisted extraction of cannabinoids from Cannabis sativa L. optimized by response surface methodology. J. Food Sci. 2018; 83, 700-10.
V Briones-Labarca, C Giovagnoli-Vicuña and R Cañas-Sarazúa. Optimization of extraction yield, flavonoids and lycopene from tomato pulp by high hydrostatic pressure-assisted extraction. Food Chem. 2019; 278, 751-9.
VL Singleton and JA Rossi. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965; 16, 144-58.
AK Esmaeili, RM Taha, S Mohajer and B Banisalam. Antioxidant activity and total phenolic and flavonoid content of various solvent extracts from in vivo and in vitro grown Trifolium pratense L. (red clover). Biomed. Res. Int. 2015; 2015, 643285.
LL Mensor, FS Menezes, GG Leitão, AS Reis, TCD Santos, CS Coube and SG Leitão. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001; 15, 127-30.
M Ebrahimzadeh, S Nabavi and S Nabavi. Antioxidant activities of methanol extract of Sambucus ebulus L. flower. Pak. J. Biol. Sci. 2009; 12, 447-50.
EAH Mohamed, MJA Siddiqui, LF Ang, A Sadikun, SH Chan, SC Tan, MZ Asmawi and MF Yam. Potent α-glucosidase and α-amylase inhibitory activities of standardized 50 % ethanolic extracts and sinensetin from Orthosiphon stamineus Benth as anti-diabetic mechanism. BMC Complement Altern. Med. 2012; 12, 176.
T Chipiti, MA Ibrahim, M Singh and MS Islam. In vitro α-amylase and α-glucosidase inhibitory effects and cytotoxic activity of Albizia antunesiana extracts. Pharmacogn. Mag. 2015; 11, S231-S236.
D Banani, DC Manabendra, D Amitabha, DT Anupam, K Nongalleima and D Lokesh. Anti-oxidant and anti-inflammatory activity of aqueous and methanolic extracts of rhizome part of Drynaria quercifolia (L.) j. Smith. Int. J. Pharm. Pharm. Sci. 2014; 6, 43-9.
S Sakat, P Tupe and A Juvekar. Gastroprotective effect of methanol extract of Oxalis corniculata Linn (whole plant) experimental animals. Planta Med. 2010; 76, P090.
R Chirinos, H Rogez, D Campos, R Pedreschi and Y Larondelle. Optimization of extraction conditions of antioxidant phenolic compounds from mashua (Tropaeolum tuberosum Ruíz & Pavón) tubers. Sep. Purif. Tech. 2007; 55, 217-25.
L Zhang, Y Jiang, X Pang, P Hua, X Gao, Q Li and Z Li. Simultaneous optimization of ultrasound-assisted extraction for flavonoids and antioxidant activity of Angelica keiskei using response surface methodology (RSM). Molecules 2019; 24, 3461.
Z Wissam, B Ghada, A Wassim and K Warid. Effective extraction of polyphenols and proanthocyanidins from pomegranate’s peel. Int. J. Pharm. Pharm. Sci. 2012; 4, 675-82.
I Tomaz, L Maslov, D Stupić, D Preiner, D Ašperger and JK Kontić. Multi‐response optimisation of ultrasound‐assisted extraction for recovery of flavonoids from red grape skins using response surface methodology. Phytochem. Anal. 2016; 27, 13-22.
R Tabaraki and A Nateghi. Optimization of ultrasonic-assisted extraction of natural antioxidants from rice bran using response surface methodology. Ultrason. Sonochem. 2011; 18, 1279-86.
A Bucić-Kojić, M Planinić, S Tomas, M Bilić and D Velić. Study of solid-liquid extraction kinetics of total polyphenols from grape seeds. J. Food Eng. 2007; 81, 236-42.
SH Mohd-Setapar, SN Mohamad-Aziz, CS Chuong, MAC Yunus, MAA Zaini and MJ Kamaruddin. A review of mixed reverse micelle system for antibiotic recovery. Chem. Eng. Commun. 2014; 201, 1664-85.
G Martemucci, C Costagliola, M Mariano, L D’andrea, P Napolitano and AG D’Alessandro. Free radical properties, source and targets, antioxidant consumption and health. Oxygen 2022; 2, 48-78.
AM Dirir, M Daou, AF Yousef and LF Yousef. A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochem. Rev. 2022; 21, 1049-79.
EO Ayua, SG Nkhata, SJ Namaumbo, EH Kamau, TN Ngoma and KO Aduol. Polyphenolic inhibition of enterocytic starch digestion enzymes and glucose transporters for managing type 2 diabetes may be reduced in food systems. Heliyon 2021; 7, e06245.
H Ullah, S Zaman, F Juhara, L Akter, SM Tareq, EH Masum and R Bhattacharjee. Evaluation of antinociceptive, in-vivo & in-vitro anti-inflammatory activity of ethanolic extract of Curcuma zedoaria rhizome. BMC Complement Med. Ther. 2014; 14, 346.
T Ozdal, E Capanoglu and F Altay. A review on protein-phenolic interactions and associated changes. Food Res. Int. 2013; 51, 954-70.
M Ali, T Homann, J Kreisel, M Khalil, R Puhlmann, HP Kruse and H Rawel. Characterization and modeling of the interactions between coffee storage proteins and phenolic compounds. J. Agr. Food Chem. 2012; 60, 11601-8.
MJ Podolsk, MH Biermann, C Maueröder, J Hahn and M Herrmann. Inflammatory etiopathogenesis of systemic lupus erythematosus: An update. J. Inflammation Res. 2015; 8, 161-71.
J Vallecillo-Hernández, MD Barrachina, D Ortiz-Masiá, S Coll, JV Esplugues, S Calatayud and C Hernández. Indomethacin disrupts autophagic flux by inducing lysosomal dysfunction in gastric cancer cells and increases their sensitivity to cytotoxic drugs. Sci. Res. 2018; 8, 3593.
M Derouich, EDT Bouhlal, A Hmidani, M Bammou, B Bourkhis, K Sellam and C Alem. Assessment of total polyphenols, flavonoids and anti-inflammatory potential of three Apiaceae species grown in the Southeast of Morocco. Sci. Afr. 2020; 9, e00507.
MS Blois. Antioxidant determinations by the use of a stable free radical. Nature 1958; 181, 1199-200.
S Shehna, S Sreelekshmi, P Remani, G Padmaja and S Lakshmi. Anti-cancer, anti-bacterial and anti-oxidant properties of an active fraction isolated from Curcuma zedoaria rhizomes. Phytomed. Plus 2022; 2, 100195.
G Göger, M Allak, A Şen, F Göger, M Tekin and G Özek. Assessment of Cota altissima (L.) J. Gay for phytochemical composition and antioxidant, anti-inflammatory, antidiabetic and antimicrobial activities. Z. Naturforsch. C A J. Biosci. 2021; 76, 317-27.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






