Comparative Analysis of Glucomannan Properties from Wet and Dry-Extractions of Porang (Amorphophallus muelleri Blume) and Commercial Konjac Glucomannan
DOI:
https://doi.org/10.48048/tis.2025.8278Keywords:
Amorphophallus muelleri, Dry extraction, Molecular properties, Wet extraction, Konjac glucomannanAbstract
Porang tubers (Amorphophallus muelleri Blume) are known as konjac in Indonesian prominent glucomannan sources. The research objective is to compare the dry and wet extraction methods. Two extraction methods (dry and wet) were carried out. First, porang flour was obtained from a centrifugal grinder and purified twice with 70 % ethanol solution for 2 h, respectively. Next, the sample was milled with ethanol solution and precipitated twice with 70 % ethanol solution. The 2 samples were compared to a konjac commercial glucomannan for physicochemical and molecular properties. The study revealed that the physical characteristics of glucomannan extracted through the wet method significantly differed from those obtained via dry extraction and the commercially available Konjac glucomannan. Specifically, the glucomannan from dry extraction demonstrated poorer physical qualities compared to its wet-extracted counterpart, and even more so when compared to commercial Konjac. The wet-extracted porang glucomannan’s viscosity (33,000 cPs) and whiteness level (70.04) were markedly different from the commercial Konjac, which had a viscosity of 39,000 cPs and a whiteness level of 83.17. In contrast, the dry extraction method yielded lower values, with a viscosity of 22,000 cPs and a whiteness level of 63.43. While FTIR, XRD and DSC analyses produced similar results across the board, the thermal properties of the wet-extracted porang glucomannan displayed a higher resistance to decomposition compared to the other 2 samples.
HIGHLIGHTS
- Reliable and economical technique production of glucomannan
- Glucomannan obtained through wet extraction methods is of higher quality compared to that obtained via dry extraction.
- The molecular characteristics of konjac glucomannan and glucomannan extracted from wet porang are remarkably similar.
- Konjac glucomannan and glucomannan from wet porang exhibit comparable solubility profiles.
- The viscosity levels of glucomannan derived from wet porang align with the food industry’s standards.
GRAPHICAL ABSTRACT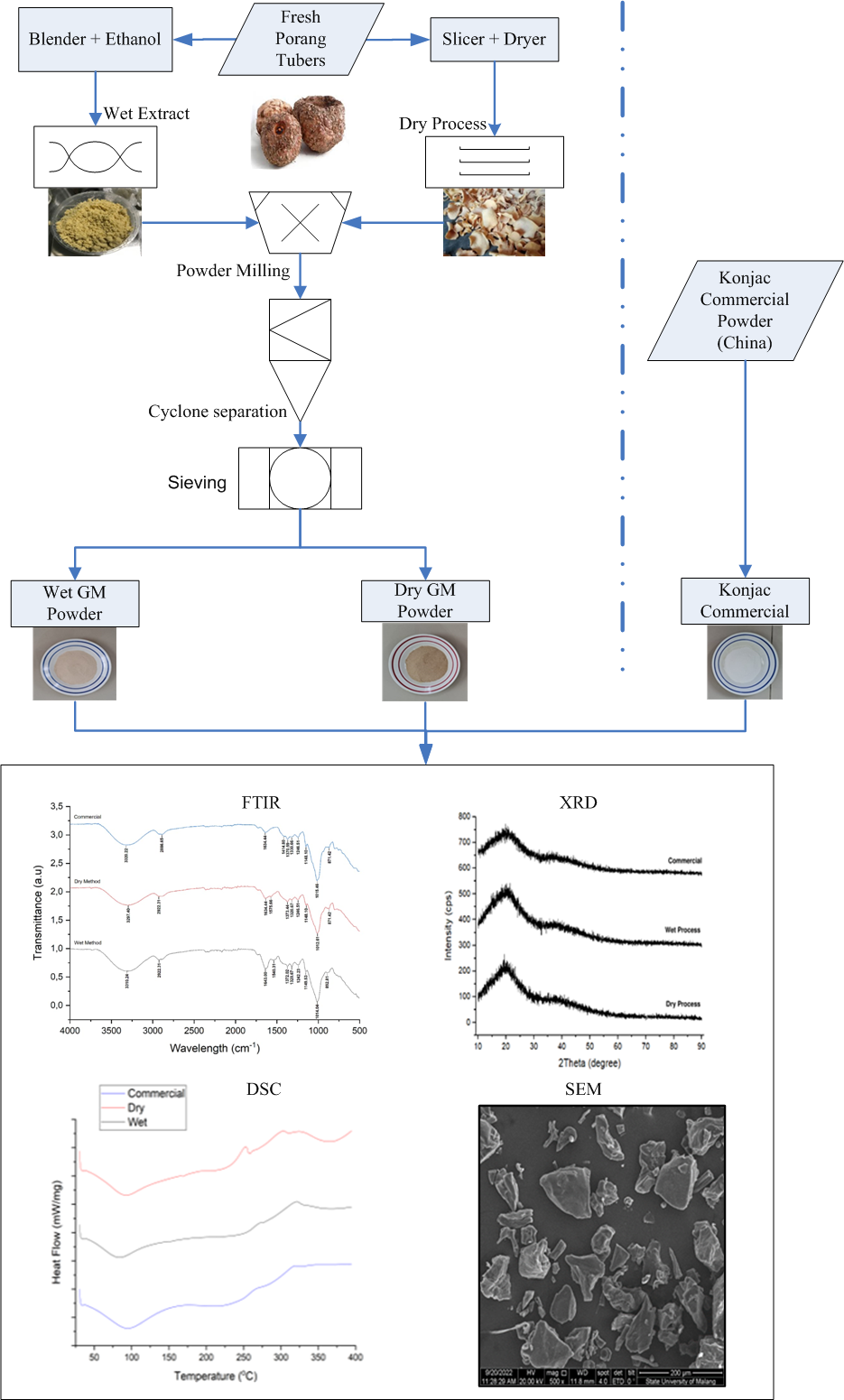
Downloads
References
M Alonso-Sande, D Teijeiro-Osorio, C Remuñán-López and MJ Alonso. Glucomannan, a promising polysaccharide for biopharmaceutical purposes. Eur. J. Pharm. Biopharm. 2009; 72, 453-62.
A Yanuriati, DW Marseno, Rochmadi and E Harmayani. Characteristics of glucomannan isolated from fresh tuber of Porang (Amorphophallus muelleri Blume). Carbohydr. Polym. 2017; 156, 56-63.
R Tester and F Al-Ghazzewi. Beneficial health characteristics of native and hydrolysed konjac (Amorphophallus konjac) glucomannan. J. Sci. Food Agr. 2016; 96; 3283-91.
K Dwiyono and MA Djauhari. Indonesian konjac: Its benefits in industry and food security. Universitas Nasional, Jakarta, Indonesia, 2019.
S Behera and R Ray. Konjac glucomannan, a promising polysaccharide of Amorphophallus konjac K. Koch in health care. Int. J. Biol. Macromol. 2016; 92, 942-56.
A Faridah and SB Widjanarko. Optimization of multilevel ethanol leaching process of porang flour (Amorphophallus muelleri) using response surface methodology. Int. J. Adv. Sci. Eng. Inf. Tech. 2013; 3, 74-80.
O Tatirat and S Charoenrein. Physicochemical properties of konjac glucomannan extracted from konjac flour by a simple centrifugation process. LWT Food Sci. Tech. 2011; 44, 2059-63.
W Xu, S Wang, T Ye, W Jin, J Liu, J Lei, B Li and C Wang. A simple and feasible approach to purify konjac glucomannan from konjac flour - temperature effect. Food Chem. 2014; 158, 171-6.
M Chua, TC Baldwin, TJ Hocking and K Chan. Traditional uses and potential health benefits of Amorphophallus konjac K. Koch ex N.E.Br. J. Ethnopharmacol. 2010; 128, 268-78.
A Yanuriati and D Basir. Peningkatan kelarutan glukomanan porang (Amorphophallus muelleri Blume) dengan penggilingan basah dan kering (in Indonesian). Agritech 2020; 40, 223-31.
Nurlela, D Andriani and R Arizal. Extraction of glucomannan from porang (Amorphophallus muelleri Blume) flour using ethanol. J. Sains Terapan KImia 2020; 14, 88-98.
N Nurlela, N Ariesta, E Santosa and T Muhandri. Physicochemical properties of glucomannan isolated from fresh tubers of Amorphophallus muelleri Blume by a multilevel extraction method. Food Res. 2022; 6, 345-53.
A Kurt and T Kahyaoglu. Purification of glucomannan from salep: Part 2. Structural characterization. Carbohydr. Polym. 2017; 169, 406-16.
C Zhang, JD Chen and FQ Yang. Konjac glucomannan, a promising polysaccharide for OCDDS. Carbohydr. Polym. 2014; 104, 175-81.
L Guo, W Yokoyama, L Chen, F Liu, M Chen and F Zhong. Characterization and physicochemical properties analysis of konjac glucomannan: Implications for structure-properties relationships. Food Hydrocolloids 2021; 120, 106818.
SB Widjanarko, A Nugroho and T Estiasih. Functional interaction components of protein isolates and glucomannan in food bars by FTIR and SEM studies. Afr. J. Food Sci. 2011; 5, 12-21.
LJ Bellamy. The infra-red spectra of complex molecules. 3rd ed. Champan and Hall, London, 1975.
NT An, DT Thien, NT Dong, PL Dung and NV Du. Isolation and characteristics of polysaccharide from Amorphophallus corrugatus in Vietnam. Carbohydr. Polym. 2011; 84, 64-8.
C Wu, S Peng, C Wen, X Wang, L Fan, R Deng and J Pang. Structural characterization and properties of konjac glucomannan/curdlan blend films. Carbohydr. Polym. 2012; 89, 497-503.
W Xu, Y Wang, W Jin, S Wang, B Zhou, J Li, B Li and L Wang. A one-step procedure for elevating the quality of konjac flour: Azeotropy-assisted acidic ethanol. Food Hydrocolloids 2014; 35, 653-60.
A Kurt and T Kahyaoglu. Purification of glucomannan from salep: Part 2. Structural characterization. Carbohydr. Polym. 2017; 169, 406-16.
B Li, J Xia, Y Wang and B Xie. Structure characterization and its antiobesity of ball-milled konjac flour. Eur. Food Res. Tech. 2005; 221, 814-20.
JE Witoyo, BD Argo, SS Yuwono and SB Widjanarko. A pilot plant scale of yellow konjac (Amorphophallus muelleri Blume) flour production by a centrifugal mill using response surface methodology. Slovak J. Food Sci. 2021; 15, 199-209.
L Kumalla, SH Surmarlan and MB Hermanto. Uji performansi pengering semprot tipe Buchi B-290 pada proses pembuatan tepung santan (in Indonesian). J. Bioproses Komoditas Tropis 2013; 1, 44-53.
X Du, J Li, J Chen and B Li. Effect of degree of deacetylation on physicochemical and gelation properties of konjac glucomannan. Food Res. Int. 2012; 46, 270-8.
T Ye, L Wang, W Xu, J Liu, Y Wang, K Zhu, S Wang, B Li and C Wang. An approach for prominent enhancement of the quality of konjac flour: Dimethyl sulfoxide as medium. Carbohydr. Polym. 2014; 99, 173-9.
LX Wang, AR Lee, Y Yuan, XM Wang and TJ Lu. Preparation and FTIR, Raman and SEM characterizations of konjac glucomannan-KCl electrogels. Food Chem. 2019; 331, 127289.
B Li and B Xie. Synthesis and characterization of konjac glucomannan/poly(vinyl alcohol) interpenetrating polymer networks. J. Appl. Polym. Sci. 2004; 93, 2775-80.
S Popović and Ž Skoko. X-ray diffraction broadening analysis. Maced. J. Chem. Chem. Eng. 2015; 34, 39-49.
C Wang, M Xu, W Lv, P Qiu, Y Gong and D Li. Study on rheological behavior of konjac glucomannan. Phys. Procedia 2012; 33, 25-30.
Y Fang, C Selomulya and XD Chen. On measurement of food powder reconstitution properties. Drying Tech. 2008; 26, 3-14.
K Nishinari, PA Williams and GO Phillips. Review of the physico-chemical characteristics and properties of konjac mannan. Food Hydrocolloids 1992; 6, 199-222.
SB Widjanarko, A Nugroho and T Estiasih. Functional interaction components of protein isolates and glucomannan in food bars by FTIR and SEM studies. Afr. J. Food Sci. 2011; 5, 12-21.
J Li, T Ye, X Wu, J Chen, S Wang, L Lin and B Li. Preparation and characterization of heterogeneous deacetylated konjac glucomannan. Food Hydrocolloids 2014; 40, 9-15.
LJ Bellamy. Amides, proteins and polypeptides. In: LJ Bellamy (Ed.). The infra-red spectra of complex molecules. Springer, Dordrecht, Netherlands, 1975, p. 231-62.
Y Hua, M Zhang, C Fu, Z Chen and GYS Chan. Structural characterization of a 2-O-acetylglucomannan from Dendrobium officinale stem. Carbohydr. Res. 2004; 339, 2219-24.
M Nasrollahzadeh, M Atarod, M Sajjadi, SM Sajadi and Z Issaabadi. Plant-mediated green synthesis of nanostructures: Mechanisms, characterization, and applications. Interface Sci. Tech. 2019; 28, 199-322.
K Wang, K Wu, M Xiao, Y Kuang, H Corke, X Ni and F Jiang. Structural characterization and properties of konjac glucomannan and zein blend films. Int. J. Biol. Macromol. 2017; 105, 1096-104.
A Kurt and T Kahyaoglu. The physicochemical and structural characteristics of cultivated sahlep. Int. J. Second. Metab. 2017; 4, 488-98.
CG Biliaderis. Differential scanning calorimetry in food research - a review. Food Chem. 1983; 10, 239-65.
A Rohman and YB Che-Man. Analysis of lard in food products for halal authentication study. Agritechnology 2008; 28, 192-201.
B Menard and K Menard. Detecting the adulteration of extra virgin olive oil by controlled cooling DSC. PerkinElmer, Waltham, 2013.
J Li, T Ye, X Wu, J Chen, S Wang, L Lin and B Li. Preparation and characterization of heterogeneous deacetylated konjac glucomannan. Food Hydrocolloids 2014; 40, 9-15.
M Kugimiya and F Ohno. Surface deposits on glucomannan granules in polished konjac flours (in Japanish). Nippon Shokuhin Kagaku Kogaku Kaishi 2021; 68, 360-7.
N Nurlela, N Ariesta, E Santosa and T Muhandri. Physicochemical properties of glucomannan isolated from fresh tubers of Amorphophallus muelleri Blume by a multilevel extraction method. Food Res. 2022; 6, 345-53.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






