Alpha(α)-mangostin (Xanthone of Garcinia mangostana L.): Augmenting Macrophages Activity for an Effective Diabetic Wound Healing
DOI:
https://doi.org/10.48048/tis.2024.8254Keywords:
Alpha(α)-mangostin, Diabetic wound healing, Macrophage cells, Protein expressionAbstract
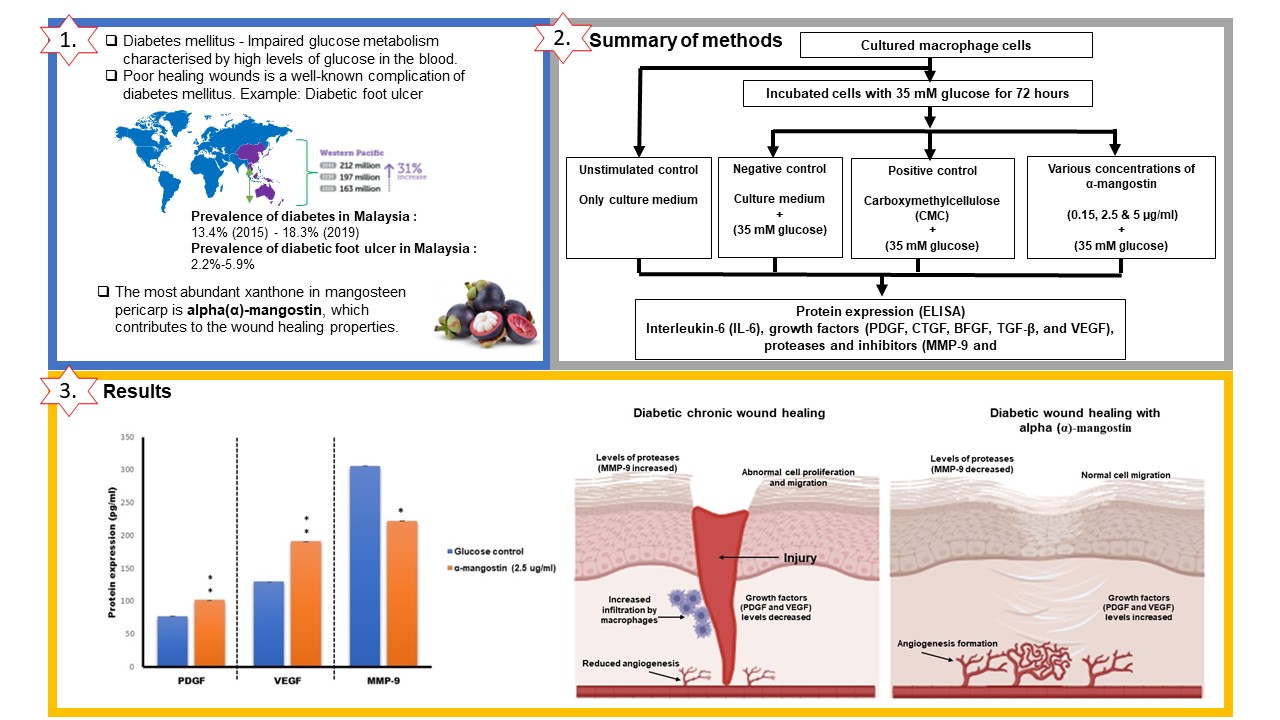
Diabetic wounds are particularly difficult to treat medically because they heal at a slower pace than regular wounds. Macrophages are essential in all stages of normal wound healing, including inflammation, proliferation and rem odelling. When wound healing is affected, macrophages can reduce the level of growth factor and increase the level of interleukin-6 (IL-6), disrupt the balance between tissue inhibitors of metalloproteinase (TIMPs) and matrix metalloproteinase (MMPs), which can slow down healing. Alpha(α)-mangostin, a natural xanthone derived from the pericarp of the mangosteen, has gained considerable attention due to its anti-inflammatory properties, suggesting its potential to promote wound healing. However, its exact role in healing diabetic foot ulcers, common in diabetes, remains unclear. Hence, this study aims to explore how α-mangostin might affect diabetic wound healing by evaluating its impact on PDGF, CTGF, BFGF, VEGF, TGF-β, MMP-9, TIMP-2 and IL-6 secretion in macrophage cells. Human monocytic macrophages (THP-1) were incubated with a 35 mM glucose solution for 72 h to create a glucose-enriched medium. The cells were then incubated with α-mangostin (0.15, 2.5 and 5 µg/mL) together with 35 mM glucose. Carboxymethyl cellulose (CMC) served as positive controls; glucose-enriched media and media-alone served as negative controls. Protein expression was measured using ELISA. α-mangostin (2.5 µg/mL) increased the levels of PDGF and VEGF and decreased the level of MMP-9 compared to glucose controls. There was no significant difference in other growth factors, TIMP-2 and IL-6 protein levels across any of the treatment groups compared to glucose controls. In conclusion, α-mangostin particularly at 2.5 µg/mL demonstrated a significant increase in PDGF and VEGF levels while simultaneously reducing MMP-9 in macrophage cells under glucose-induced conditions. These findings suggest that α-mangostin holds the potential for enhancing the healing of chronic wounds in diabetic conditions.
HIGHLIGHTS
- Treatment of macrophages (THP-1 cells) with 2.5 µg/mL Alpha (α)-mangostin in a high glucose medium led to a significant increase in PDGF and VEGF secretion compared to glucose control.
- Treatment of the cells with 2.5 µg/mL Alpha (α)-mangostin in a high glucose medium led to a significant reduction of MMP-9 secretion compared to glucose control.
- There is no significant effects of Alpha (α)-mangostin on IL-6 and TIMP secretion compared to controls
- This study suggests that Alpha (α)-mangostin may be beneficial in promoting diabetic wound healing via stimulation of PDGF and VEGF secretion and reducing MMP-9 levels.
GRAPHICAL ABSTRACT
Downloads
References
D Baltzis, I Elefheriadou and A Veves. Pathogenesis and treatment of impaired wound healing in diabetes mellitus: New insights. Adv. Ther. 2014; 31, 817-36.
A Hingorani, GM LaMuraglia, P Henke, MH Meissner, L Loretx, KM Zinszer, VR Driver, R Frykberg, TL Carman, W Marston, JL Mills and MH Murad. The management of diabetic foot: A clinical practice guideline by the society for vascular surgery in collaboration with the american podiatric medical association and the society for vascular medicine. J. Vasc. Surg. 2016; 63, 3S-21S.
SM Huang, CC Wu, MH Chiu, CH Wu, YT Chang, GS Chen and CC Elan. High glucose environment induces M1 macrophage polarization that impairs keratinocyte migration via TNF-α: An important mechanism to delay the diabetic wound healing. J. Dermatol. Sci. 2019; 96, 159-67.
TJ Koh and LA DiPieto. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011; 13, 23.
AE Louiselle, SM Niemiec, C Zgheib and KW Liechty. Macrophage polarization and diabetic wound healing. Transl. Res. 2021; 236, 109-16.
T Lucas, A Waisman, R Ranjan. J Roes, T Krieg, W Muller, A Roers and A Eming. Differential roles of macrophages in diverse phases of skin repair. J. Immunol. 2010; 184, 3964-77.
SA Eming, T Krieg and JM Davidson. Inflammation in wound repair: Molecular and cellular mechanisms. J. Invest. Dermatol. 2007; 127, 514-25.
SJ Wolf, WJ Melvin and K Gallagher. Macrophage-mediated inflammation in diabetic wound repair. Semin. Cell Dev. Biol. 2021; 119, 111-18.
R Yang, P Li, N Li, Q Zhang, X Bai, L Wang, Y Xiao, L Sun, Q Yang and J Yan. Xanthones from the pericarp of Garcinia mangostana. Molecules 2017; 22, 683.
W Suttirak and S Manurakchinakorn. In vitro antioxidant properties of mangosteen peel extract. J. Food Sci. Tech. 2014; 51, 3546-58.
JP Chaverri, NC Rodriquez, MO Ibarra and JMP Rojas. Medicinal properties of mangosteen (Garcinia mangostana). Food Chem. Toxicol. 2008; 46, 3227-39.
OA Hanafiah, T Abidin, S Ilyas, M Nainggolan and E Syamsudin. Wound healing activity of binahong (Anredera cordifolia (Ten.) steenis) leaves extract towards NIH-3T3 fibroblast cells. Int. Den. Med. Res. 2019; 12, 854-8.
DJ Jiang, Z Dai and YJ Li. Pharmacological effects of xanthones as cardiovascular protective agents. Cardiovas. Drug Rev. 2004; 22, 91-102.
KJ Zhang, QL Gu, K Yang, XJ Ming and JX Wang. Anticarcinogenic effects of α-mangostin: A review. Planta Med. 2017; 83, 188-202.
S Mohan, S Syam, SI Abdelwahab and N Thangavel. An anti-inflammatory molecular mechanism of action of α-mangostin, the major xanthone from the pericarp of Garcinia mangostana: An in silico, in vitro and in vivo approach. Food Funct. 2018; 9, 3860-71.
D Rizaldy, R Hartati, T Nadhifa and I Fidrianny. Chemical compounds and pharmacological activities of mangosteen (Garcinia mangostana L.)-updated review. Bio. Res. Appl. Chem. 2021; 12, 2503-16.
KL McFarland, JM Hahn, KA Combs and DM Supp. Antiproliferative, proapoptotic, and potential antifibrotic effects of α-mangostin in fibroblasts from keloid lesions and normal skin. Keloid Res. 2021; 1, 1-10.
M Abate, C Pagano, M Masullo, M Citro, S Pisanti, S Piacente and M Bifulco. Mangostanin, a xanthone derived from Garcinia mangostana fruit, exerts protective and reparative effects on oxidative damage in human keratinocytes. Pharmaceuticals 2022; 15, 84.
M Genin, F Clement A Fattaccioli, M Raes and C Michiels. M1 and M2 macrophages derived from THP-1 cells differentially modulate the response of cancer cells to etoposide. BMC Cancer 2015; 15, 577.
P Buranasin, K Mizutani, K Iwasaki, CPN Mahasarakham, D Kido, K Takeda and Y Izumi. High glucose-induced oxidative stress impairs proliferation and migration of human gingival fibroblasts. Plos One 2018; 13, e0201855.
KV Maltan and SB Pruett. ELISA assays and alcohol: Increasing carbon chain length can interfere with detection of cytokines. Alcohol 2011; 45, 1-9.
Y Lu, Y Yang, L Xiao, L., S Li, X Liao and H Liu. Autocrine and paracrine effects of vascular endothelial cells promote cutaneous wound healing. Biomed. Res. Int. 2021; 2021, 6695663.
ZX Zhu, WH Cai, T Wang, HB Ye, YT Zhu, LS Chi, YM Duan, CC Sun, YH Xuan and LT Jin. BFGF-regulating MAPKs are involved in high glucose-mediated ROS production and delay of vascular endothelial cell migration. Plos One 2015; 10, e0144495.
NIM Fadilah, M Maarof, A Motta, Y Tabata and MB Fauzi. The discovery and development of natural-based biomaterials with demonstrated wound healing properties: A reliable approach in clinical trials. Biomedicines 2022; 10, 2226.
OC Chijioke, RM Aliyu, OE Ohams, AD Onyebuchi, AI Fountain and NB Nwaforcha. Natural honey and diabetic wound healing: A review of literature. Magna Sci. Adv. Res. Rev. 2023; 7, 67-73.
M Patrick, WNWM Zohdi, SA Muid and E Omar. Alpha-mangostin (Garcinia mangostana Linn.) and its potential application in mitigating chronic wound healing. Malays. Appl. Biol. 2022; 51, 1-8.
GS Schultz, GA Chin, L Moldawer and RF Diegelmann. Handbook of mechanism of vascular disease. Adelaide, Australia, 2011, p. 423.
G Han and R Ceiley. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017; 34, 599-610.
FL Lesmana, A Rizqiawan, I Mulyawan, NPM Sumarta, DB Kamadjaja, DC Pramono, T Kei, GDS Rurus, NA Sativa and RA Fanddhy. Effect of the application of (Garcinia mangostana L.) towards PDGF-B expression on human gingival fibroblast cell culture after wound healing scratch test assay (in vitro study). J. Int. Dent. Med. Res. 2021; 14, 1413-8.
SM Ayuk, H Abrahamse and NN Houreld. The role of matrix metalloproteinases in diabetic wound healing in relation to photobiomodulation. J. Diabetes Res. 2016; 2016, 2897656.
AR IM, YM Kim, YW Chin and S Chae. Protective effects of compounds from Garcinia mangostana L.(mangosteen) against uvb damage in Hacat cells and hairless mice. Int. J. Mol. Med. 2017; 40, 1941-49.
BZ Johnson, AW Stevenson, CM Prele, MW Fear and FM Wood. The role of IL-6 in skin fibrosis and cutaneous wound healing. Biomedicines 2020; 8, 101.
M Zubair, A Malik and J Ahmad. Plasma adiponectin, IL-6, hsCRP, and TNF-α levels in subject with diabetic foot and their correlation with clinical variables in a North Indian tertiary care hospital. Indian J. Endocrinol. Metab. 2012; 16,769-76.
J Jin, Y Bao, Y Wang, H Zheng, H Guo, L Zhang, R Guo and L Yang. Protective activity of alpha-mangostin against uvb-induced injury in Hacat cells by modulating the ceramide and MAPK and NF-kB signaling pathways. J. Food Biochem. 2023; 2023, 4702866.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






