Protective Effect of DHQ-11 against Hypoxia-induced Vasorelaxation
DOI:
https://doi.org/10.48048/tis.2024.8192Keywords:
Smooth muscle, Endothelium, Nitric oxide, Ca-channels, K channels, HypoxiaAbstract
Hypoxia, or the lack of oxygen, has multiple impacts on the vascular system. The major molecular sensors for hypoxia at the cellular level are hypoxia inducible factor and heme oxygenase. Hypoxia also acts on the vasculature directly conveying its damaging effects through disruption of the control of vascular tone, particularly in the coronary circulation, enhancement of inflammatory responses and activation of coagulation pathways. These effects could be particularly detrimental under pathological conditions such as obstructive sleep apnea and other breathing disorders. Introduction: The effect of conjugate 2-(3,4-Dihydroxyphenyl)-6-{[1-(2’-bromo-3’,4’-dimethoxyphenyl)-6,7-dimethoxy-3,4-dihydroisoquinolin-2(1H )-yl]methyl}-3,5,7-trihydroxychroman-4-one (DHQ-11) on hypoxia-induced vasorelaxation was investigated in rat aortic rings using standard organ bath techniques. Materials and methods: Hypoxia was stimulated by a superfusion of aortic rings with a glucose-free Krebs solution bubbled with 95 % N2/5 % CO2. The effect of conjugate DHQ-11 was assessed after a 60- min period of hypoxia on aortic rings precontracted with 50 mm KCl or 1µM phenylephrine (PE). The conjugate DHQ-11 significantly attenuated hypoxia-induced vasorelaxation in the endothelium-intact aortic rings precontracted with KCl or PE in a concentration-dependent manner. Results and discussion: This effect of conjugate DHQ-11 was more potent in aortic rings precontracted with PE than those with KCl where it maximally reduced hypoxia-induced vasorelaxation from 44.7 ± 3.7 to 5.4 ± 3.7 and 33.9 ± 3.4 to 10.8 ± 4.2 %, respectively. The removal of the endothelium attenuated the effect of conjugate DHQ-11 on hypoxia-induced vasorelaxation. Similarly, pretreatment of endothelium-intact aortic rings with L-NAME and methylene blue also attenuated the effect of conjugate DHQ-11 on hypoxia-induced vasorelaxation. Furthermore, the blockade of the ATP-sensitive KATP channel with glibenclamide and the calcium-activated large conductance BKCa channel with TEA also significantly attenuated the effect of conjugate DHQ-11 on hypoxia-induced vasorelaxation. Collectively, these results indicated that conjugate DHQ-11 attenuated the hypoxia-induced vasorelaxation suggesting that it alleviated the oxidative damage of vasculature. Conclusions: This effect of conjugate DHQ-11 possible is mediated through several mechanisms including the blockage of the extracellular Ca2+ entry via the voltage-dependent and receptor-operative Ca2+ channels, as well as inhibition of sarcoplasmic reticulum Ca2+ release via the inositol triphosphate pathway. In addition, endothelium and NO/sGC/cGMP/PKG pathway, as well as KATP and BKCa channels most likely participate in protection by conjugate DHQ-11 against hypoxia-induced vasorelaxation.
HIGHLIGHTS
- Conjugate DHQ-11 (1-(2´-bromo-4´,5´-dimethoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline alkaloid and Dihydroquercetin flavonoid) was formed by chemical connection.
- Conjugate DHQ-11 exerts potent vasorelaxant and vasoprotective effects on KCl- and PE-induced contractions in rat aortic vascular SMCs.
- The vasoprotective effect of conjugate DHQ-11 effectively protects blood vessels from hypoxia-induced damage through the modification of the function of Ca2+L channels and IP3 cascade, as well as the activation of NOS, NO/sGC/cGMP/PKG signaling pathways, KATP and KCa
GRAPHICAL ABSTRACT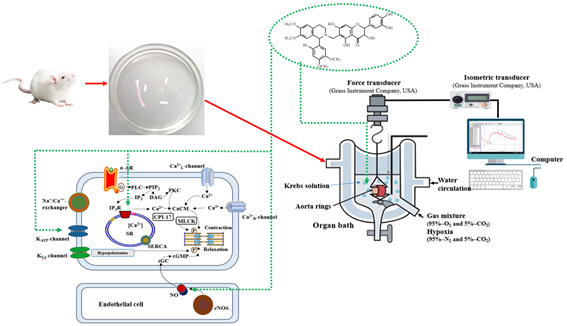
Downloads
References
FDR Hobbs. Prevention of cardiovascular diseases. BMC Med. 2015; 13, 261.
VV Uzbekov, BF Abdullaev, IZ Jumayev, YI Oshchepkova, PB Usmanov and SI Salikhov. Comparative study of the antiarrhythmic activity of liposomal forms of lappaconitine hydrobromide and its complex with glycyrrhizic acid monoammonium salt in the aconitine arrhythmia model. Pharmaceut. Chem. J. 2023; 56, 1327-32.
G Csányi and FJ Miller. Oxidative stress in cardiovascular disease. Int. J. Mol. Sci. 2014; 15, 6002-8.
YI Oshchepkova, VV Uzbekov, IZ Jumayev, SY Rustamov, PB Usmanov and SI Salikhov. Comparative study of antiarrhythmic and inotropic activity of amiodarone hydrochloride and its complexes with glycyrrhizic acid and monoammonium salt of glycyrrhizic acid. Eksperimental'naya i Klinicheskaya Farmakologiya 2023; 86, 15-22.
RM Touyz, FJ Rios, R Alves-Lopes, KB Neves, LL Camargo and AC Montezano. Oxidative stress: A unifying paradigm in hypertension. Can. J. Cardiol. 2020; 36, 659-70
IZ Zhumaev, SN Boboev, PB Usmanov, SB Qurbonova, SY Rustamov, AT Esimbetov, GS Begdullaeva, AA Zaripov and SM Adizov. Mechanism of positive inotropic effect of vincamine on cardiac muscle contraction activity. Biomed. Pharmacol. J. 2022; 15, 2309-16
M Luo and ME Anderson. Mechanisms of altered Ca2+ handling in heart failure. Circ. Res. 2013; 113, 690-708.
A Horovitz, CB Menice, R Laporte and KG Morgan. Mechanism of smooth muscle contraction. Pharmacol. Rev. 1996; 76, 967-1003.
AV Somlyo, M Bond, AP Somlyo and A Scarpa. Inositol trisphosphate-induced calcium release and contraction in vascular smooth muscle. Proc. Natl. Acad. Sci. USA 1985; 82, 5231-5.
S Moreland, RF Coburn, CB Baron and RS Moreland. Mechanical and biochemical events during hypoxia-induced relaxations of rabbit aorta. Adv. Exp. Med. Biol. 1991; 304, 147-57.
ER Hedegaard, BD Nielsen, A Kun, AD Hughes, C Kroingaard, S Mogensen, VV Matchkov, O Frobert and U Simonsen. KV7 channels are involved in hypoxia-induced vasodilatation of porcine coronary arteries. Br. J. Pharmacol. 2014; 171, 69-82.
KM Gauthier. Hypoxia-induced vascular smooth muscle relaxation: increased ATP-sensitive K+ efflux or decreased voltage-sensitive Ca2+ influx? Am. J. Physio. Heart Circ. Physiol. 2006; 291, H24-H25.
H Cai and DG Harrison. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ. Res. 2000; 87, 840-4.
WJ Pearce, JM Williams, MW Hamade, MM Chang and CR White. Chronic hypoxia modulates endothelium-dependent vasorelaxation through multiple independent mechanisms in ovine cranial arteries. Adv. Exp. Med. Biol. 2006; 578, 87-92.
F Murad. Nitric oxide and cyclic GMP in cell signaling and drug development. N. Engl. J. Med. 2006; 355, 2003-11.
AM Gurney. Mechanisms of drug-induced vasodilation. J. Pharm. Pharmacol. 1994; 46, 242-51.
GM Herrera and BR Walker. Involvement of L-type calcium channels in hypoxic relaxation of vascular smooth muscle. J. Vasc. Res. 1998; 35, 265-73.
PB Usmanov, IZ Jumayev, Sh Yu Rustamov, AA Zaripov, AT Esimbetov, ShN Zhurakulov and VI Vinogradova. The Combined Inotropic and Vasorelaxant Effect of DHQ-11, a Conjugate of Flavonoid Dihydroquercetin with Isoquinoline Alkaloid 1-aryl-6,7-dimethoxy-1,2.3,4-tetrahydroisoquinoline. Biomedical & Pharmacology Journal. 2021; 14, 651-661.
CL Macêdo, LHC Vasconcelos, ACC Correia, IRR Martins, DPD Lira, BVDO Santos, FDA Cavalcante and BAD Silva. Mechanisms underlying vasorelaxation induced in rat aorta by galetin 3,6-dimethyl ether, a flavonoid from Piptadenia stipulacea (benth.) ducke. Molecules 2014; 19, 19678-95.
A Janaszak-Jasiecka, A Siekierzycka, A Płoska, IT Dobrucki and L Kalinowski. Endothelial dysfunction driven by Hypoxia - The influence of oxygen deficiency on NO bioavailability. Biomolecules 2021; 11, 982.
M Ottolini, K Hong and SK Sonkusare. Calcium signals that determine vascular resistance. Wiley Interdiscip. Rev. Syst. Biol. Med. 2019; 11, e1448.
I Eichler, J Wibawa, I Grgic, A Knorr, S Brakemeier, AR Pries, J Hoyer and R Köhler. Selective blockade of endothelial Ca2+-activated small- and intermediate-conductance K+-channels suppresses EDHF-mediated vasodilation. Br. J. Pharmacol. 2003; 138, 594-601.
CK Chan and PM Vanhoutte. Hypoxia, vascular smooth muscles and endothelium. Acta Pharm. Sin B 2013; 3, 1-7
MT Gewaltig and G Kojda. Vasoprotection by nitric oxide: mechanisms and therapeutic potential. Cardiovasc. Res. 2002; 55, 250-60.
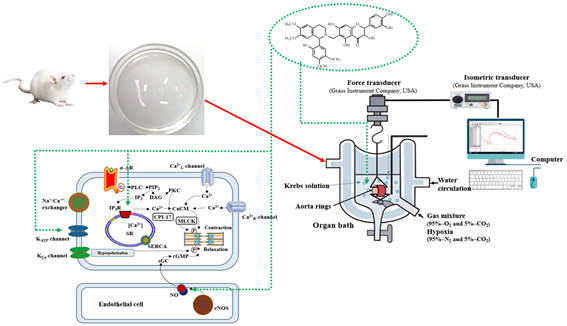
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






