Metabolomics Profiles of Solid State and Submerge Fermentation of Corn Silk using Mixed Microbes
DOI:
https://doi.org/10.48048/tis.2024.7483Keywords:
Corn silk, Heterofermentative microbe, Heatmaps, LC-MS/MS, Xanthine oxidase inhibitionAbstract
Corn silk is a waste product when harvesting corn, but it contains many bioactive compounds. This research characterized and assessed the antioxidant bioactivity of compounds isolated from corn silk. Two different biological fermentation processes were compared: Solid-state fermentation (SSF) and submerged fermentation (SmF). Fresh corn silk (Zea mays L.) from both sweet corn and feed corn were fermented separately with various microorganisms (Bacillus subtilis, Lactobacillus sp. and Saccharomyces cerevisiae). Biological compounds were identified using LC-MS/MS. The extract was then tested for total phenolic content (TPC) and total flavonoid content (TFC). Next, the extract was tested for xanthine oxidase inhibition and antioxidant inhibition using the ABTS method. Potential contamination with the heavy metals As, Pb, and Hg was checked. The results show that fermented corn silk extract contains numerous bioactive compounds and amino acids. Among the different extract process variations, the highest TPC and TFC were 6.1 ± 0.7 µg GAE/g dry wt. and 68.3 ± 1.1 µg QE/g dry wt., respectively, and the highest xanthine oxidase inhibition was 76.5 ± 6.4 %. The above highest results were obtained using SmF of sweet corn for 60 days. In contrast, the highest antioxidant activity of 66.9 % was found using SSF of sweet corn for 20 days. Neither sweet corn nor feed corn were found to contain heavy metal ions. In summary, fermented corn silk extract samples contain numerous bioactive compounds, including both flavonoid and phenolic compounds, and the extract demonstrates antioxidant activity. Corn silk extracts from fermentation thus have potential for utilization in health products.
HIGHLIGHTS
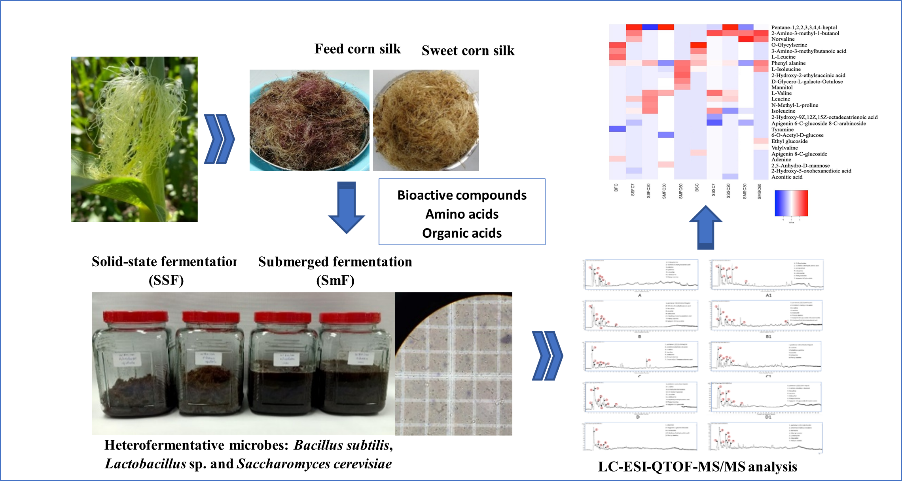
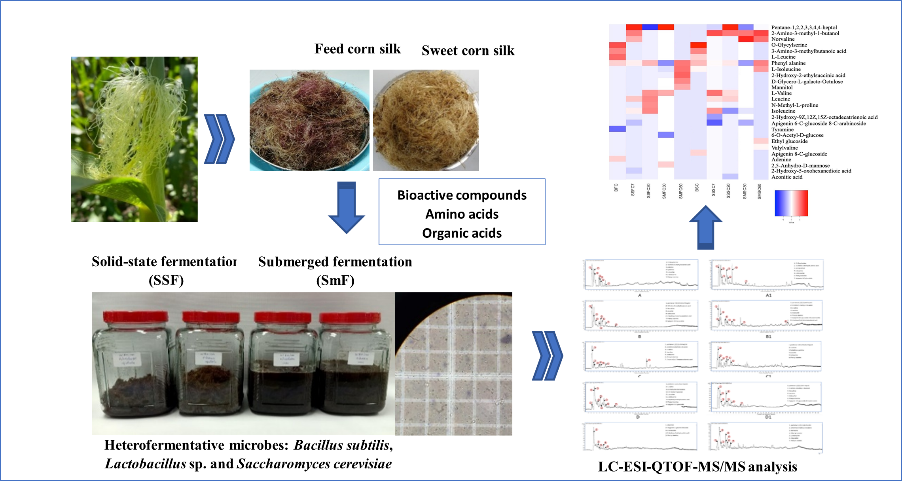
- Feed corn silk and sweet corn silk underwent solid-state fermentation (SSF) or submerged fermentation (SmF) for extraction using heterofermentative microbes of Bacillus subtilis, Lactobacillus and Saccharomyces cerevisiae to produce the high bioactive compounds, amino acids, and organic acids
- Metabolomic analysis of the fermentation extracts was carried out by LC-ESI-QTOF-MS/MS (Liquid Chromatography-Mass Spectrometry) using the heatmaps generated metabolites profiles
- The main amino acids found in the extractions were phenylalanine, leucine, and their derivatives
- SmF of sweet corn silk yielded the highest xanthine oxidase inhibitory activity (76 %)
- SSF of feed corn silk and sweet corn silk displayed the high antioxidant activity
GRAPHICAL ABSTRACT
Downloads
References
E Sarepoua, R Tangwongchai, B Suriharn and K Lertrat. Relationships between photochemical and antioxidant activity in corn silk. Int. Food 2013; 20, 2073-9.
N Aukkanit, T Kemngoen and N Ponharn. Utilization of corn silk in low fat meatballs and its characteristics. Proc. Soc. Behav. Sci. 2014; 197, 1403-10.
S Zilic, M Jankovic, Z Basic, J Vancetovic and V Maksimovic. Antioxidant activity, phenolic profile, chlorophyll and mineral matter content of corn silk (Zea mays L.): Comparison with medicinal herbs. J. Cereal Sci. 2016; 69, 363-70.
S Saheed, AE Oladipipo, AA Abdulazeez, SA Olarewaju, NO Ismaila, IA Emmanuel, QD Fatimah and AY Aisha. Toxicological evaluations of Stigma maydis (corn silk) aqueous extract on hematological and lipid parameters in Wistar rat. Toxicol. Rep. 2015; 2, 638-44.
J Singh, P Rasane, V Nanda and S Kaur. Bioactive compounds of corn silk and their role in management of glycaemic response. J. Food Sci. Tech. 2023; 60, 1695-710.
P Li, G Ren, Y Sun, D Jiang and C Liu. Extraction optimization, preliminary identification and bioactivities in corn silk. Evid. Base. Compl. Alternative Med. 2023; 2023, 5685174.
P Lertkaeo, A Limmongkon, M Srikummool, T Boonsong, W Supanpaiboon and D Surangkul. Antioxidative and neuroprotective activities of peanut sprout extracts against oxidative stress in SK-N-SH cells. Asian Pac. J. Trop. Biomed. 2017; 7, 64-9.
G Selo, M Planini´c, M Tiˇsma, S Tomas, DK Komleni´c and A Buci´c-Koji´c. A comprehensive review on valorization of agro-food industrial residues by solid-state fermentation. Foods 2021; 10, 927.
N Nuengchamnong, T Saesong, K Ingkaninan and SW areekul. Antioxidant activity and chemical constituents identification by LC-MS/MS in Bio-fermented fruit drink of Morinda citrifolia L. Trends Sci. 2023; 20, 6498.
O Zannou, I Koca, TMS Aldawoud and CM Galanakis. Recovery and stabilization of anthocyanins and phenolic antioxidants of roselle (Hibiscus sabdariffa L.) with hydrophilic deep eutectic solvents. Molecules 2020; 25, 3715.
A Grobelna, S Kalisz and M Kieliszek. Effect of processing methods and storage time on the content of bioactive compounds in blue honeysuckle berry purees. Agronomy 2019; 9, 860.
A Grobelna, S Kalisz and M Kieliszek. The effect of the addition of blue honeysuckle berry juice to apple juice on the selected quality characteristics, anthocyanin stability, and antioxidant properties. Biomolecules 2019; 9, 744.
Y Wang, G Zhang, J Pan and D Gong. Novel insights into the inhibitory mechanism of kaempferol on xanthine oxidase. J. Agr. Food Chem. 2015; 63, 526-34.
Thermo Fisher Scientific Inc. Inductively couple plasma- mass spectrometry (ICP-MS). Thermo Fisher Scientific Inc, Massachusetts, 2019.
Z Dai, Z Wu, W Zhu and G Wu. Amino acids in microbial metabolism and function. Adv. Exp. Med. Biol. 2022; 1354, 127-43.
UV Karpiuk, VS Kyslychenko, IS Cholak and OI Yemelianova. Determination of free and bound amino acids in plant raw materials of zea mays L. by the method of high‐performance liquid chromatography. Pharmacognosy Res. 2020; 12, 143.
LK Feizi, J Seifdavati, H Rafiee, F Rezazadeh, JH Meléndez, OM Molina and AZ Salem. Biotechnological valorization of fermented soybean meal for sustainable ruminant and non-ruminant feeding: Modulating ruminal fermentation, gut or ruminal microflora, immune system, and growth performance. Biomass Convers. Biorefinery 2022, https://doi.org/10.1007/s13399-022-02971-7.
X Cui, Y Yang, M Zhang, F Jiao, T Gan, Z Lin and Y Qian. Optimized ensiling conditions and microbial community in mulberry leaves silage with inoculants. Front. Microbiol. 2022; 13, 813363.
H Jiang, F Yu, LI Qin, N Zhang, Q Cao, W Schwab and C Song. Dynamic change in amino acids, catechins, alkaloids, and gallic acid in six types of tea processed from the same batch of fresh tea (Camellia sinensis L.) leaves. J. Food Compos. Anal. 2019; 77, 28-38.
F Garavand, DF Daly and LG Gómez-Mascaraque. The consequence of supplementing with synbiotic systems on free amino acids, free fatty acids, organic acids, and some stability indexes of fermented milk. Int. Dairy J. 2023; 137, 105477.
JL Kim, C Hidalgo-Shrestha, ND Bonawitz, RB Franke and C Chapple. Spatio-temporal control of phenylpropanoid biosynthesis by inducible complementation of a cinnamate 4-hydroxylase mutant. J. Exp. Bot. 2021; 72, 3061-73.
K Yonekura-Sakakibara, Y Higashi and R Nakabayashi. The origin and evolution of plant flavonoid metabolism. Front. Plant Sci. 2019; 10, 943.
A Aires and R Carvalho. Compositional study and antioxidant potential of polyphenol extracted from corn by-product, using ultrasound extraction method. Austin Chrom. 2016; 3, 1043.
O Santín and G Moncalian. Loading of malonyl-CoA onto tandem acyl carrier protein domains of polyunsaturated fatty acid synthases. J. Biol. Chem. 2018; 293, 12491-501.
K Bhuvaneshwari and S Sridevi. Analysis of nutrients and phytochemical contents in corn silk (Zea Mays). Int. J. Sci. Res. 2015; 78, 79-81.
ADT Phan, BM Flanagan, BR D’Arcy and MJ Gidley. Binding selectivity of dietary polyphenols to different plant cell wall components: Quantification and mechanism. Food Chem. 2017; 233, 216-27.
R Yucharoen, P Srisuksomwong, J Julsrigival, L Mungmai, T Kaewkod and Y Tragoolpua. Antioxidant, anti-tyrosinase, and anti-skin pathogenic bacterial activities and phytochemical compositions of corn silk extracts, and stability of corn silk facial cream product. Antibiotics 2023, 12, 1443.
HN Thai, JV Camp, G Smagghe and K Raes. Improved release and metabolism of flavonoids by steered fermentation processes: A review. Int. J. Mol. Sci. 2014; 15, 19369-88.
D Zhang, Y Wang and H Liu. Corn silk extract inhibit the formation of Nε-carboxymethyllysine by scavenging glyoxal/methyl glyoxal in a casein glucose-fatty acid model system. Food Chem. 2020; 309, 125708.
MM Alajlani. Characterization of subtilosin gene in wild type Bacillus spp. and possible physiological role. Sci. Rep. 2022; 12, 10521.
SJ Hur, SY Lee, YC Kim, I Choi and GB Kim. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chem. 2014; 160, 346-56.
S Lin, G Zhang, Y Liao, J Pan and D Gong. Dietary flavonoids as xanthine oxidase inhibitors: Structure-affinity and structure-activity relationships. J. Agr. Food Chem. 2015; 63, 7784-94.
LM Valdés-Velasco and E Favela-Torres, A Théatre, A Arguelles-Arias, JG Saucedo-Castañeda and P Jacques. Relationship between lipopeptide biosurfactant and primary metabolite production by Bacillus strains in solid-state and submerged fermentation. Bioresource Tech. 2022; 345, 126556.
V Kumar, V Ahluwalia, S Saran, J Kumar, AK Patel and RR Singhania. Recent developments on solid-state fermentation for production of microbial secondary metabolites: Challenges and solutions. Bioresource Tech. 2021; 323, 124566.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






