The Effect of Light Intensity and Nutrient Formula on the Growth of Chlorella ellipsoidea in Batch Cultivation
DOI:
https://doi.org/10.48048/tis.2023.7009Keywords:
Batch cultivation, Biomass, Chlorella, Growth rate, Light intensity, Media, MicroalgaeAbstract
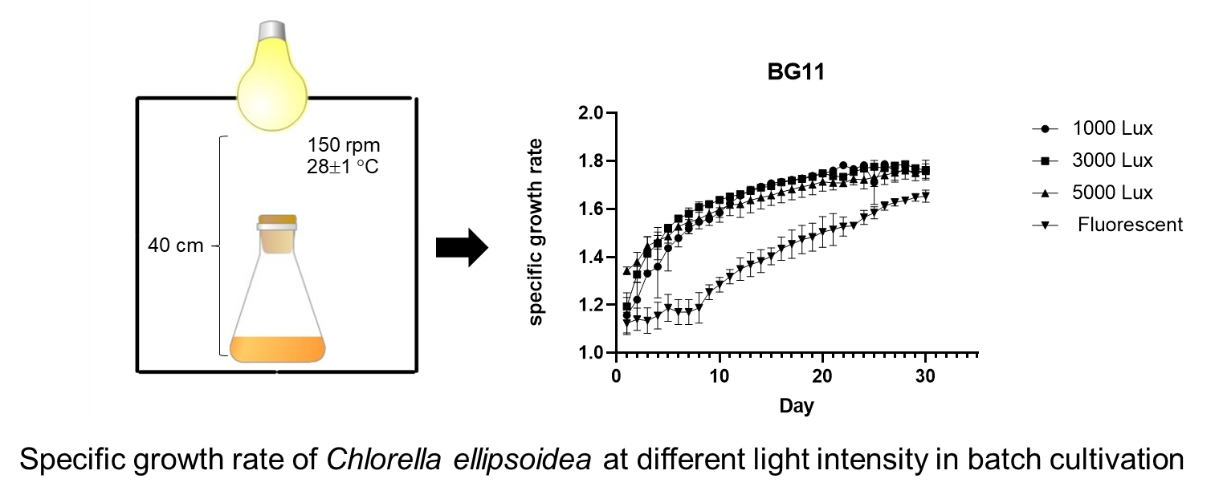
In the aquaculture feed industry, the batch cultivation of the alga Chlorella ellipsoidea is frequent because it allows the control of factors contributing to product quality. In this study, we conducted batch-cultivation experiments in a smart algae cabinet with 4 different light intensities, 1,000, 3,000 and 5,000 Lux, as well as a fluorescent lamp (8,000 Lux). The growth curves, specific growth rate and biomass productivity of C. ellipsoidea provided with the BG-11, Bold and Chu-13 nutrient formulae were evaluated. The results revealed biomasses of 1.315 ± 0.0087, 1.042 ± 0.0063 and 1.096 ± 0.0036 g/L/day and specific growth rates of 0.979 ± 0.0067, 0.7453 ± 0.0060 and 0.7781 ± 0.0026 per day for the 3 formulae, BG-11, Bold and Chu-13, respectively. The mean biomasses of C. ellipsoidea grown on BG-11 at all light intensities were significantly higher than those for other media (p-value = 0.0001). With the log phase achieved in 3 days, BG-11 was more suitable for rearing at all tested light intensities, while Chu-13 was only applicable at 5,000 Lux. Using all 3 light intensities, it was possible to reduce the log-phase of algae growth from 7 to 3 days and increase biomass by more than 1.2 times when compared to fluorescent light culture.
HIGHLIGHTS
- The batch cultivation was carried out in a smart algae cabinet that we are building and will publish soon.
- A smart algae network can control the factors that affect cultivation while also conducting all experiments at the same time.
- The 3 light intensities and BG-11 media used for cultivation in a smart algae cabinet can accelerate growth and biomass production.
GRAPHICAL ABSTRACT
Downloads
References
P Feng, Z Deng, L Fan and Z Hu. Lipid accumulation and growth characteristics of Chlorella zofingiensis under different nitrate and phosphate concentrations. J. Biosci. Bioeng. 2012; 114, 405-10.
S Otleş and R Pire. Fatty acid composition of Chlorella and Spirulina microalgae species. J. AOAC Int. 2001; 84, 1708-14.
W Zhao, M Duan, X Zhang and T Tan. A mild extraction and separation procedure of polysaccharide, lipid, chlorophyll and protein from Chlorella spp. Renew. Energ. 2018; 118, 701-8.
LBSD Moraes, RFB Santos, GFG Junior, GCP Mota, DMDM Dantas, RDS Bezerra and AO Gálvez. Microalgae for feeding of penaeid shrimp larvae: An overview. Aquaculture Int. 2022; 30, 1295-313.
MA Hesni, A Hedayati, A Qadermarzi, M Pouladi, S Zangiabadi and N Naqshbandi. Using Chlorella vulgaris and iron oxide nanoparticles in a designed bioreactor for aquaculture effluents purification. Aquacultural Eng. 2020; 90, 102069.
W Sawaengsak, T Silalertruksa, A Bangviwat and SH Gheewala. Life cycle cost of biodiesel production from microalgae in Thailand. Energ. Sustain. Dev. 2014; 18, 67-74.
DDW Tsai, R Ramaraj and PH Chen. Growth condition study of algae function in ecosystem for CO2 bio-fixation. J. Photochem. Photobiol. B Biol. 2012; 107, 27-34.
C Yeesang and B Cheirsilp. Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresource Tech. 2011; 102, 3034-40.
EW Becker. Micro-algae as a source of protein. Biotechnol. Adv. 2007; 25, 207-10.
A Jehlee, P Khongkliang and S O-Thong. Biogas production from Chlorella sp. TISTR 8411 biomass cultivated on biogas effluent of seafood processing wastewater. Energ. Procedia 2017; 138, 853-7.
G Dragone. Challenges and opportunities to increase economic feasibility and sustainability of mixotrophic cultivation of green microalgae of the genus Chlorella. Renew. Sustain. Energ. Rev. 2022; 160, 112284.
S Liang, X Liu, F Chen and Z Chen. Current microalgal health food R & D activities in China. Hydrobiologia 2004; 512, 45-48.
A Widjaja, CC Chien and YH Ju. Study of increasing lipid production from fresh water microalgae Chlorella vulgaris. J. Taiwan Inst. Chem. Eng. 2009; 40, 13-20.
D Tang, W Han, P Li, X Miao and J Zhong. CO2 biofixation and fatty acid composition of Scenedesmus obliquus and Chlorella pyrenoidosa in response to different CO2 levels. Bioresource Tech. 2011; 102, 3071-6.
Y Park, KW Je, K Lee, SE Jung and TJ Choi. Growth promotion of Chlorella ellipsoidea by co-inoculation with Brevundimonas sp. isolated from the microalga. Hydrobiologia 2008; 598, 219-28.
SH Cho, SC Ji, SB Hur, J Bae, IS Park and YC Song. Optimum temperature and salinity conditions for growth of green algae Chlorella ellipsoidea and Nannochloris oculate. Fish. Sci. 2007; 73, 1050-6.
GL Bhalamurugan, O Valerie and L Mark. Valuable bioproducts obtained from microalgal biomass and their commercial applications: A review. Environ. Eng. Res. 2018; 23, 229-41.
CK Wayne, CS Reen, PL Show, YY Jiun, LT Chuan and C Jo-Shu. Effects of water culture medium, cultivation systems and growth modes for microalgae cultivation: A review. J. Taiwan Inst. Chem. Eng. 2018; 91, 332-44.
P Asadi, HA Rad and F Qaderi. Lipid and biodiesel production by cultivation isolated strain Chlorella sorokiniana pa.91 and Chlorella vulgaris in dairy wastewater treatment plant effluents. J. Environ. Health Sci. Eng. 2020; 18, 573-85.
CF Yang, ZY Ding and KC Zhang. Growth of Chlorella pyrenoidosa in wastewater from cassava ethanol fermentation. World J. Microbiol. Biotechnol. 2008; 24, 2919-25.
A Baidya, T Akter, MR Islam, AKMA Shah, MA Hossain, MA Salam and SI Paul. Effect of different wavelengths of LED light on the growth, chlorophyll, β-carotene content and proximate composition of Chlorella ellipsoidea. Heliyon 2021; 7, e08525.
M Khalaji, SA Hosseini, R Ghorbani, N Agh, H Rezaei, M Kornaros and E Koutra. Treatment of dairy wastewater by microalgae Chlorella vulgaris for biofuels production. Biomass Convers. Biorefinery 2023; 13, 3259-65.
MB Mangesh and S Felix. Effect of light wavelengths on biomass production and pigment enhancement of Chlorella vulgaris in indoor system. Res. J. Biotechnol. 2019; 14, 111-7.
HS Yun, YS Kim and HS Yoon. Characterization of Chlorella sorokiniana and Chlorella vulgaris fatty acid components under a wide range of light intensity and growth temperature for their use as biological resources. Heliyon 2020; 6, e04447.
TJ Gunawan, Y Ikhwan, F Restuhadi and U Pato. Effect of light intensity and photoperiod on growth of Chlorella pyrenoidosa and CO2 Biofixation. In: Proceedings of the 2nd International Conference on Energy, Environmental and Information System, Semarang, Indonesia. 2018.
M Alariqi W Long and BI Musah. Algae biomass for biofuel production; yield analysis of Chlorella Vulgaris and Scenedesmus Meyen in different culture media. In: Proceedings of the 12th International Conference on Environment Science and Engineering, Beijing, China. 2023, p. 144-50.
K Chankhong, W Chotigeat and J Iewkittayakorn. Effects of culture medium on growth kinetics and fatty acid composition of Chlorella sp. T12. Songklanakarin J. Sci. Tech. 2018; 40, 1098-104.
R Chandra, Amit and UK Ghosh. Effects of various abiotic factors on biomass growth and lipid yield of Chlorella minutissima for sustainable biodiesel production. Environ. Sci. Pollut. Res. 2019; 26, 3848-61.
S Satthong, K Saego, P Kitrungloadjanaporn, N Nuttavut, S Amornsamankul and W Triampo. Modeling the effects of light sources on the growth of algae. Adv. Difference Equat. 2019; 2019, 170.
P Nawkarkar, AK Singh, MZ Abdin and S Kumar. Life cycle assessment of Chlorella species producing biodiesel and remediating wastewater. J. Biosci. 2019; 44, 89.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






