Identification of Antioxidants and Acetylcholinesterase Inhibitors from Hapaline benthamiana Schott. Using at Line LC-ESI-QTOF MS/MS
DOI:
https://doi.org/10.48048/tis.2023.6202Keywords:
Hapaline benthamiana schott, Acetylcholinesterase, At line-LC-ESI-QTOF-MS/MS, Antioxidant, Phenolics, Flavonoids, GlycolipidAbstract
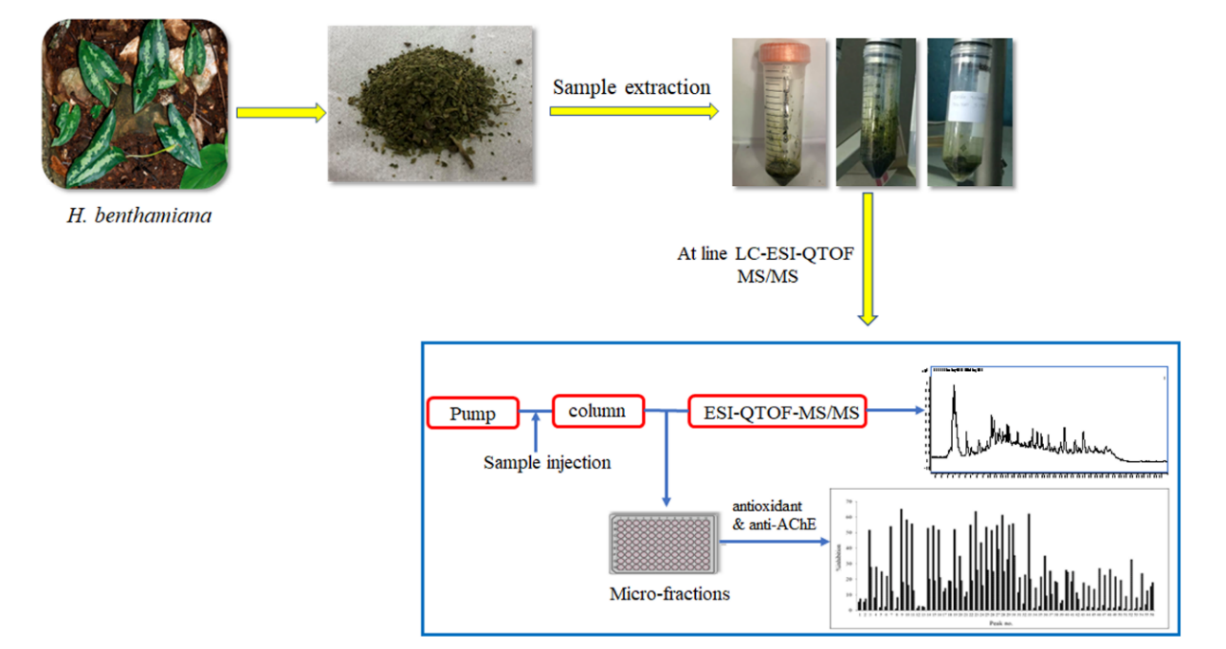
In this work, total phenolics, total flavonoids, antioxidant activity, and acetylcholinesterase inhibition potency of Hapaline benthamiana schott are reported. In addition, the investigation of antioxidant and anti-acetylcholinesterase compounds of this plant coupled to the at line-LC-ESI-QTOF-MS/MS technique is introduced for the first time. The results show that the methanolic extract contains the highest amount of total phenolics, total flavonoids, antioxidant activity, and acetylcholinesterase inhibition activity. The at line-LC-ESI-QTOF-MS/MS technique has provided information on the biological activity of each compound while mass spectrometry provided information on their molecular weights. Based on their mass spectra, 53 compounds were identified with most of them being phenolic, flavonoid, and glycolipid compounds. Seventeen compounds exhibited a strong antioxidant activity ranging between 51.52 - 63.58 % inhibition whereas one compound, namely septentriodine, showed a strong acetylcholinesterase inhibitory effect of more than 50 %. This study indicates that Hapaline benthamiana schott is a source of active compounds containing antioxidant and anti-acetylcholinesterase activities with health benefits. In addition, at line-LC-ESI-QTOF-MS/MS has been shown to be an effective technique for the rapid and simultaneous analyses of antioxidant and anti-acetylcholinesterase compounds in plant extracts.
HIGHLIGHTS
- Investigation of antioxidants and acetylcholinesterase inhibitors from Hapaline benthamiana Schott coupled to at line-LC-ESI-QTOF-MS/MS technique is reported for the first time
- Fifty-three compounds from methanolic extract were successfully identified
- Eighteen compounds exhibited strong biological activity
- This technique is a rapid and valuable tool for the identification of lead compounds in complex extracts and for the investigation of their bioactivity
GRAPHICAL ABSTRACT
Downloads
References
X Dong-Ping, L Ya, M Xiao, Z Tong, Z Yue, Z Jie, Z Jiao-Jiao and L Hua-Bin. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017; 18, 96.
ML Gabriel, RD Méndez, H Sophie, CEA Julio, FB Yilan, OP Ania, GD Jesús, JMQ Humberto, FD Albys, IA Elizabeth, B Natalie, EMS Isidro, OR Teresa, C Paul and C Ann. Antioxidants in plants: A valorization potential emphasizing the need for the conservation of plant biodiversity in Cuba. Antioxidants 2020; 9, 1048.
N Chaves, A Santiago and JC Alías. Quantification of the antioxidant activity of plant extracts: Analysis of sensitivity and hierarchization based on the method used. Antioxidants 2020, 9, 76.
A Phaniendra, DB Jestadi and L Periyasam. Free radicals: Properties, sources, targets, and their implication in various diseases. Indian J. Clin. Biochem. 2015; 30, 11-26.
SB Nimse and D Pal. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015; 5, 27986-8006.
G Martemucci , C Costagliola, M Mariano, L D’andrea, P Napolitano and AG D’Alessandro. Free radical properties, source and targets, antioxidant consumption and health. Oxygen 2022; 2, 48-78.
S Li, Y Zhao, L Zhang, X Zhang, L Huang, D Li, C Niu, Z Yang and Q Wang. Antioxidant activity of lactobacillus plantarum strains isolated from traditional Chinese fermented foods. Food Chem. 2012; 135, 1914-9.
TC Dos Santos, TM Gomes, BA Serra Pinto, AL Camara and AM de Andrade Paes. Naturally occurring acetylcholinesterase inhibitors and their potential use for alzheimer’s disease therapy. Front. Pharmacol. 2018; 9, 1192.
B Sarkar, S Alam, TK Rajib, SS Islam, Y Araf and A Ullah. Identification of the most potent acetylcholinesterase inhibitors from plants for possible treatment of Alzheimer’s disease: A computational approach. Egypt. J. Med. Hum. Genet. 2021; 22, 10.
M Mathew and S Subramanian. In vitro screening for anti-cholinesterase and antioxidant activity of methanolic extracts of ayurvedic medicinal plants used for cognitive disorders. PLos One 2014; 9, e86804.
N Nuengchamnong and K Ingkaninan. An on-line LC-MS/ DPPH approach towards the quality control of antioxidative ingredients in Sahastara. Songklanakarin J. Sci. Tech. 2017; 39, 123-9.
L Swiatek, E Sieniawska, KI Sinan, M Maciejewska-Turska, A Boguszewska, M Polz-Dacewicz, I Senkardes, GO Guler, NB Sadeer, MF Mahomoodally and G Zengin. LC-ESI-QTOF-MS/MS analysis, cytotoxic, antiviral, antioxidant, and enzyme inhibitory properties of four extracts of Geranium pyrenaicum Burm. f.: A good gift from the natural treasure. Int. J. Mol. Sci. 2021; 22, 7621.
N Nuengchamnong and K Ingkaninan. On-line HPLC-MS-DPPH assay for the analysis of phenolic antioxidant compounds in fruit wine: Antidesma thwaitesianum Muell. Food Chem. 2010; 118, 147-52.
S Bhandari, N Nuengchamnong, N Chaichamnong, T Seasong, K Ingkaninan and P Temkitthawon. At‐line LC‐QTOF‐MS micro‐fractionation of derris scandens (Roxb.) benth, coupled to radioassay for the early identification of PDE5A1 inhibitors. Phytochemical Anal. 2020; 31, 297-305.
ZH Su, GA Zou, A Alfred Preiss, HW Zhanga and ZM Zoua. Online identification of the antioxidant constituents of traditional Chinese medicine formula Chaihu-Shu-Gan-San by LC-LTQ-Orbitrap mass spectrometry and microplate spectrophotometer. J. Pharmaceut. Biomed. Anal. 2010; 53, 454-61.
M Mladic, DJ Scholten, WM Niessen, GW Somsen, MJ Smit and J Kool. At‐line coupling of LC-MS to bioaffinity and selectivity assessment for metabolic profiling of ligands towards chemokine receptors CXCR1 and CXCR2. J. Chrom. B 2015; 1002, 42‐53.
M Mladic, TD Waal, L Burggraaff, J Slagboom, GW Somsen, WMA Niessen, RM Kini and J Kool. Rapid screening and identification of ACE inhibitors in snake venoms using at‐line nanofractionation LCMS. Anal. Bioanalytical Chem. 2017; 409, 5987‐97.
HM Puteri-Adiba, M Arifullah, AA Nazahatul, T Sirikitputtisak, S Klaiklay, P Chumkaew, S Chewchanwuttiwong, MZ Norhazlini and H Zulhazman. Total phenolic content and antioxidant activity of limestone endemic araceae species, Alocasia farisii. IOP Conf. Earth Environ. Sci. 2021; 842, 012064.
HT Van, NT Le, NTA Huynh, HS Vo, TT Le, HV Chu, HAV Truong and QH Nguyen. Chemical composition and antibacterial activities of essential oils from Homalomena pierreana (Araceae). Nat. Prod. Res. 2022; 36, 3129-32.
HT Van, THH Duong, VS Le, GB Tran and NTA Huynh. Chemical composition and antibacterial activities of ethanolic extract from rhizomes and aerial parts of Typhonium lineare Hett. & V.D. Nguyen (Araceae). Banat’s J. Biotechnol. 2020; 11, 60-6.
T Van, NT Vo, NT Nguyen, TN Luu, TV Pham, P Tan and Q Le. Chemical composition and antibacterial activities of ethanol extract of Arisaema langbiangense rhizome (Araceae). J. Biotechnol. 2020; 11, 12-1.
J Scapinello, LG Müller, MS Z Schindler, GS Anzollin, AM Siebel, AA Boligon, R Niero, TES Saraiva, NP Maus, AH Betti, JV Oliveira, JD Magro and DD Oliveira. Antinociceptive and anti-inflammatory activities of Philodendron bipinnatifidum Schott ex Endl (Araceae). J. Ethnopharmacol. 2019; 236, 21-30.
RC Ongpoy. The medicinal properties of the alocasia genus: A systematic review. J. Asian Assoc. Schools Pharm. 2017; 6, 25-33.
A Ferid, M Arifullah, K Fatimah, B Matcha, H Zulhazman, SK Podapati and LV Reddy. Xanthine oxidase inhibitory activity, chemical composition, antioxidant properties and GCMS analysis of Keladi Candik (Alocasia longiloba Miq.). Molecules 2020; 25, 2658.
SA Baba and SA Malik. Determination of total phenolic content and flavonoid content, antimicrobial and antioxidant activity of root extract of Arisaema jacquemontii Blume. J. Taibah Univ. Sci. 2015; 9, 449-54.
HT Van, NB Tran, TNT Vo, TT Trinh, NT Nguyen, VS Le and GB Tran. Antioxidant capacity and flavoinoids, triterpenoids, polysaccharides contents from aerial part and rhizome of Homalomena pierreana (Araceae). J. Appl. Biol. Sci. 2020; 14, 15-25.
G Frausin, RBS Lima, AF Hidalgo, LC Ming and AM Pohlit. Plants of the Araceae family for malaria and related diseases: A review. Rev. Bras. Plantas Med. 2015; 17, 657-66.
T Iwashina. Flavonoids in the species of the family Araceae: A review Sebuah tinjauan: Senyawa-senyawa flavonoid pada suku Araceae. Buletin Kebun Raya 2020; 23, 1-24.
FU Afifi, V Kasabri, S Litescu1, IF Abaza and K Tawaha. Phytochemical and biological evaluations of Arum hygrophilum Boiss. (Araceae). Phcog. Mag. 2017; 30, 275-80.
D Plaipol and W Kanchana. Antioxidant activity and antidiabetic activities of Northern Thai indigenous edible plant extracts and their phytochemical constituents. Heliyon 2022; 8, e10740.
LN Lucky, E Ekramy, T Jack, W Buddhika, W Anusha, T Rebecca, W Averil and GC Wayne. Anti-acetylcholinesterase activity and antioxidant properties of extracts and fractions of Carpolobia lutea. Pharmaceut. Biol. 2017; 55, 1875-83.
S Aryal, MK Baniya, K Danekhu, P Kunwar, R Gurung and N Koirala. Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from western Nepal. Plants 2019; 8, 96.
K Ingkaninan, P Temkitthawon, K Chuenchom, T Yuyaem and W Thongnoi. Screening for acetylcholinesterase inhibitory activity in plants used in Thai traditional rejuvenating and neurotonic remedies. J. Ethnopharmacol. 2003; 89, 261-4.
S Langyan, P Chaniad and J Puripattanavo. Acetylcholinesterase inhibitory and antioxidant properties of Thai vegetables. Int. J. Pharma Med. Biol. Sci. 2017; 6, 67-72.
S Dobrinas, A Soceanu, V Popescu, IC Popovici and D Jitariu. Relationship between total phenolic content, antioxidant capacity, Fe and Cu content from Tea plant samples at different brewing times. Processes 2021; 9, 1311.
MA Osman, GI Mahmoud and SS Shoman. Correlation between total phenols content, antioxidant power and cytotoxicity. Biointerface Res. Appl. Chem. 2021; 11, 10640-53.
JRN Nansseu, J Fokom-Domgue, JJN Noubiap, EV Balti, E Sobngwi and AP Kengne. Fructosamine measurement for diabetes mellitus diagnosis and monitoring: A systematic review and meta-analysis protocol. BMJ Open 2015; 5, e007689.
E Danese, M Montagnana, A Nouvenne and G Lippi. Advantages and pitfalls of fructosamine and glycated albumin in the diagnosis and treatment of diabetes. J. Diabetes Sci. Tech. 2015; 9, 169-76
I Ivanov, R Vrancheva, IY Aneva, IN Dincheva, I Badjakov and A Pavlov. Alkaloids profiling and acetylcholinesterase inhibitory activity of Corydalis species. Plant Cell Biotechnol. Mol. Biol. 2020; 21, 12-20.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






