Effect of Solid-State Fermentation (SSF) on the Antioxidant and Hypoglycemic Activities of Jackfruit (Artocarpus Heterophyllus Lam.) By-Products using Aspergillus Niger, Aspergillus Oryzae and Rhizopus Oryzae
DOI:
https://doi.org/10.48048/tis.2023.5648Keywords:
Jackfruit rags, Solid-state fermentation, Filamentous fungi, Antioxidant activity, Hypoglycemic activityAbstract
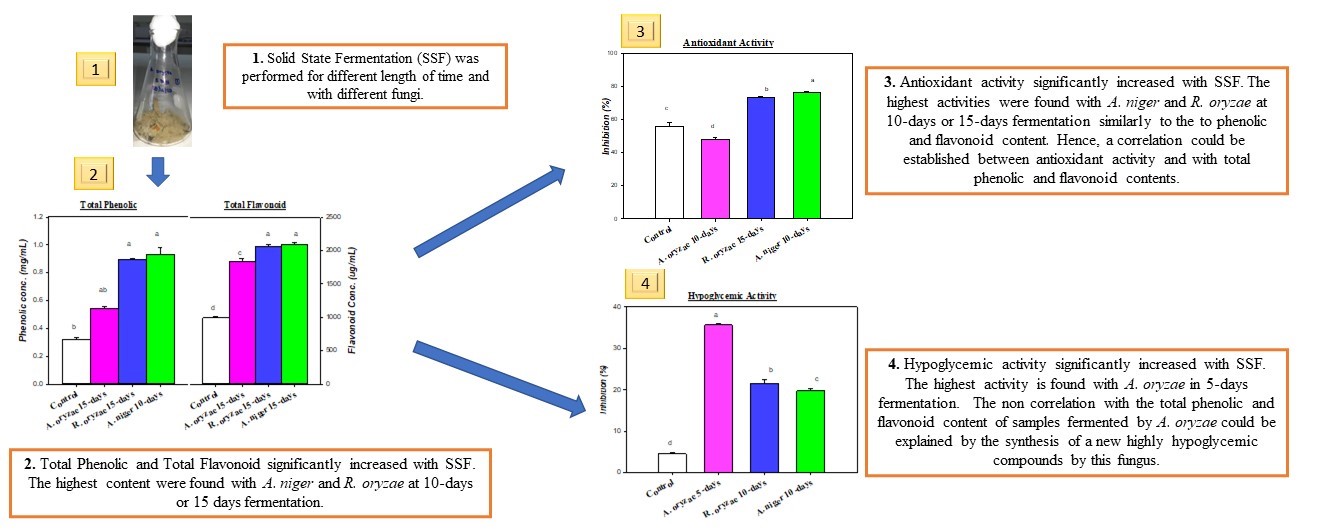
The objective of this experiment was to study the effect of Solid-State Fermentation (SSF) on jackfruit by-products using filamentous fungi. Rags from a ripen jackfruit were fermented for 5, 10 and 15 days by Aspergillus niger, Aspergillus oryzae and Rhizopus oryzae at 30 °C. Then the bioactive compounds were extracted using methanol. Total phenolic content, total flavonoid content, antioxidant activity and hypoglycemic activity of each liquid extract were estimated. The results showed significant increase and improvement in the samples in which SSF was applied. The highest total phenolic contents were found in fermented products with A. niger and R. oryzae with an average of 14.16 mg GAE/g D.S which was almost 3 times over the concentration found in unfermented products (4.83 mg GAE/g D.S). The same fermented products showed a significant increase in total flavonoid content which was 2 times higher than unfermented products with an average of 31.79 mg QuE/g D.S for fermented products and 15.08 for unfermented. The antioxidant activity was correlated to the total phenolic and flavonoid contents with the highest activity of around 76 % found in fermented products against 55 % for the unfermented ones. Finally, hypoglycemic activity was also found to be higher in fermented products than the unfermented with the best activity obtained in 5-days fermented product with A. oryzae (35.62 %) which was 8 times higher than non-fermented products (4.52 %). The present study showed that Solid-State Fermentation has a great potentiality to improve and enhance antioxidant and hypoglycemic activities of plant by-products.
HIGHLIGHTS
- SSF improved the Antioxidant and hypoglycemic properties of jackfruit rags
- niger and R. oryzae had better capacity in improving the antioxidant activity
- Phenolic Content, Flavonoid Content and Antioxidant Activity were correlated
- Better hypoglycemic activity was seen in fermented products with oryzae
- oryzae could produce a new bioactive compound with high hypoglycemic activity
GRAPHICAL ABSTRACT
Downloads
References
American Cancer Society. Global cancer facts & figures. 4th eds. American Cancer Society, Atlanta, Georgia, 2018, p. 76.
P Saeedi, P Salpea, S Karuranga, I Petersohn, B Malanda, EW Gregg and SN Unwin. Mortality attributable to diabetes in 20 - 79 years old adults, 2019 estimates: Results from the international diabetes federation diabetes atlas, 9th edition. Diabetes Res. Clin. Pract. 2020; 162, 108086.
K Jamwal, S Bhattacharya and S Puri. Plant growth regulator mediated consequences of secondary metabolites in medicinal plants. J. Appl. Res. Med. Aromat. Plants 2018; 9, 26-38.
D Twilley and N Lall. The role of natural products from plants in the development of anticancer agents. In: C Subhash, Mandal, V Mandal and T Konishi (Eds.). Natural products and drug discovery, an integrated approach. Elsevier, Amsterdam, Netherlands, 2018, p. 139-78.
KR Bonta. Dietary phenolic acids and flavonoids as potential anti-cancer agents: Current state of the art and future perspectives. Anti-Cancer Agents Med. Chem. 2020; 20, 29-48.
L Zhang, ZC Tu, X Xie, Y Lu, ZX Wang, H Wang and XM Sha. Antihyperglycemic, antioxidant activities of two Acer palmatum cultivars, and identification of phenolics profile by UPLC-QTOF-MS/MS: New natural sources of functional constituents. Ind. Crop. Prod. 2016; 89, 522-32.
L Zhang, ZC Tu, X Xie, Y Lu, ZX Wang, H Wang and XM Sha. Jackfruit (Artocarpus heterophyllus Lam.) peel: A better source of antioxidants and a-glucosidase inhibitors than pulp, flake and seed, and phytochemical profile by HPLC-QTOF-MS/MS. Food Chem. 2017; 234, 303-13.
H Abedul, B Daniel and RA Arm. Perspectives on natural compounds in chemoprevention and treatment of cancer: An update with new promising compounds. Eur. J. Cancer 2021; 149, 165-83.
Y Wang and J Zhiguo. Rapid large-scale preparation of polysaccharides from jackfruit peel waste by high-speed countercurrent chromatography and their antioxidant and hypoglycemic activities. J. Sep. Sci. 2022; 45, 771-9.
E Suchithra and S Subramanian. Biochemical evaluation of antioxidant properties of artocarpus heterophyllus lamn. rags extract studied in high fat diet-low dose streptozotocin induced experimental type 2 diabetes in rats. Diabetes Mellit. Type 2 2016; 20, 12-23.
VV Michela Verni and CG Rizello. How fermentation affects the antioxidant properties of cereals and legumes. Foods 2019; 8, 362.
E Topakas, P Katapodis, D Kekos, BJ Macris and P Christakopoulos. Production and partial characterization of xylanase by Sporotrichum thermophile under solid-state fermentation. World J. Microbiol. Biotechnol. 2003; 19, 195-8.
CN Aguilar, A Aguilera-Carbo, A Robledo, J Ventura, R Belmares and D Martinez. Production of antioxidants nutraceuticals by solid-state cultures of pomegranate (Punica granatum) peel and creosote bush (Larrea tridentata) leaves. Food Technol. Biotechnol. 2008; 46, 218-20.
PS Nigam, N Gupta and A Anthwal. Pre-treatment of agro-industrial residues. In: PS Nigam and A Pandey (Eds.). Biotechnology for agro-industrial residues utilization. Springer, Heidelberg, Germany, 2009, p. 13-33.
S Huang, Y Ma, D Sun, J Fan and S Cai. In vitro DNA damage protection and anti-inflammatory effects of Tartary buckwheats (Fagopyrum tataricum L. Gaertn) fermented by filamentous fungi. Int. J. Food Sci. 2017; 52, 2006-17.
K Pyrzynska and A Pękal. Application of free radical diphenylpicrylhydrazyl (DPPH) to estimate antioxidant capacity of food samples. Anal. Meth. 2013; 5, 4288-95.
VL Singleton and JA Rossi. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965; 16, 144-58.
I Mujic, S Dudaš, J Zivkovic, J Tomin, Z Zekovic, V Alibabic, H Jukić and R Šahinovic. Determination of total phenolic compounds in cultivars of Castanea Sativa Mill. Acta Hortic. 2009; 815, 63-8.
S Wiesneth and G Jürgenliemk. Total phenolic and tannins determination: a modification of Ph. Eur. 2.8.14 for higher throughput. Die Pharmazie - Int. J. Pharm. Sci. 2017; 72, 195-6.
S Chandra, S Khan, B Avula, H Lata, MH Yang, MA Elsohly and I A Khan. Assessment of total phenolic and flavonoid content, antioxidant properties, and yield of aeroponically and conventionally grown leafy vegetables and fruit crops: A comparative study. Evid. Based Complement. Altern. Med. 2014; 2014, 253875.
M Asadi-Samani, M Rafieian-kopaei, Z Lorigooini and H Shirzad. A screening to determine total phenol and flavonoid content of some iran’s medicinal plants grown in chaharmahal va Bakhtyari province. Indian J. Nat. Prod. Resour. 2018; 9, 296-302.
KY Kim, KA Nam, H Kurihara and SM Kim. Potent alpha-glucosidase inhibitors purified from the red alga Grateloupia elliptica. Phytochemistry 2008; 69, 2820-5.
MT Nguyen, N Nguyen, H Nguyen, T Huynh and B Min. Screening of α-glucosidase inhibitory activity of vietnamese medicinal plants: isolation of active principles from oroxylum indicum. Nat. Prod. Sci. 2012; 18, 47-51.
Y Xiao, B Zhang, Y Chen, J Miao, Q Zhang, X Rui and M Dong. Solid-state bioprocessing with Cordyceps militaris enhanced antioxidant activity and DNA damage protection of red bean (Phaseolus angularis). Cereal Chem. 2016; 94, 177-84.
AEA Magro, LC Silva, GB Rasera and RJSD Castro. Solid-state fermentation as an efficient strategy for the biotransformation of lentils: Enhancing their antioxidant and antidiabetic potentials. Bioresour. Bioprocess. 2019; 6, 9.
TB Dey and RC Kuhad. Enhanced production and extraction of phenolic compounds from wheat by solid-state fermentation with Rhizopus oryzae RCK2012. Biotechnol. Rep. 2014; 4, 120-7.
MFA Bakar, M Mohamed, A Rahmat and J Fry. Phytochemicals and antioxidant activity of different parts of bambangan (Mangifera pajang) and tarap (Artocarpus odoratissimus). Food Chem. 2009; 113, 479-83.
R Romero-Díez, S Rodríguez-Rojo, MJ Cocero, C Duarte, AA Matias and MR Bronze. Phenolic characterization of aging wine lees: Correlation with antioxidant activities. Food Chem. 2018; 259, 188-95.
A Biworo, E Tanjung, I Thalib and E Suhartono. Antidiabetic and antioxidant activity of jackfruit (artocarpus heterophyllus) extract. J. Med. Biol. Eng. 2015; 4, 318-23.
E Karimi, E Oskoueian and R Hze. Solid state fermentation effects on pistachio hulls antioxidant activities. KKU Res. J. 2010; 15, 366.
MG Kang, SH Yi and JS Lee. Production and characterization of a new α-glucosidase inhibitory peptide from aspergillus oryzae N159-1. Mycobiology 2013; 41, 149-54.
MN Sarian, QU Ahmed, SZM So’ad, AM Alhassan, S Murugesu, V Perumal, SNAS Mohamad, A Khatib and J Latip. Antioxidant and antidiabetic effects of flavonoids: A structure-activity relationship based study. Biomed. Res. Int. 2017; 14, 2017.
F Adetuyi and T Ibrahim. Effect of fermentation time on the phenolic, flavonoid and vitamin C contents and antioxidant activities of okra (abelmoschus esculentus) seeds. Niger. Food J. 2014; 3, 128-37.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






