Effect of CuO/rGO and ZnO/rGO Hybrid Additional Layers on Supercapacitor Performance
DOI:
https://doi.org/10.48048/tis.2022.5603Keywords:
Reduced graphene oxide, Copper(II) oxide/reduced graphene oxide, Zinc oxide/reduced graphene oxide, Supercapacitor, Cyclic voltammetryAbstract
Increasing the specific electrical capacitance of supercapacitors has been received great attention from both researchers and industry. Herein, how to achieve this by coating the surface of reduced graphene oxide (rGO) with copper(II) oxide (CuO) and zinc oxide (ZnO) is reported. The CuO/rGO and ZnO/rGO hybrid layers were prepared via chemical reactions between graphene oxide (GO) and salts of copper and zinc, respectively. The crystallographic structures and surface morphologies of composite materials were studied by using X-ray diffraction (XRD) and scanning electron microscopy (SEM), respectively. Cyclic voltammetry (CV) and electrical capacitance measurements were used to analyze the electrochemical properties of the composites. The results show that CuO and ZnO increased the specific electrical capacitance of rGO, while the composite CuO/rGO and ZnO/rGO materials have good chemical stability with a higher specific electrical capacity (465.73 F×g−1) than CuO/rGO (167.52 F×g−1), ZnO/rGO (185.48 F×g−1), rGO (113.50 F×g−1), and annealed graphite (53.12 F×g−1). The mechanism of increasing the specific capacitance depending on whether the composite CuO/rGO and ZnO/rGO materials act as a pseudocapacitor and/or an electrical double-layer capacitor is elucidated.
HIGHLIGHTS
- One challenging problem for supercapacitor improvement is increasing its specific electrical capacitance
- CuO/rGO and ZnO/rGO hybrid layers were prepared using graphene oxide and copper and zinc salts
- The composite CuO/rGO and ZnO/rGO materials have better chemical stability with a higher specific electrical capacity than CuO/rGO, ZnO/rGO, rGO, and annealed graphite
GRAPHICAL ABSTRACT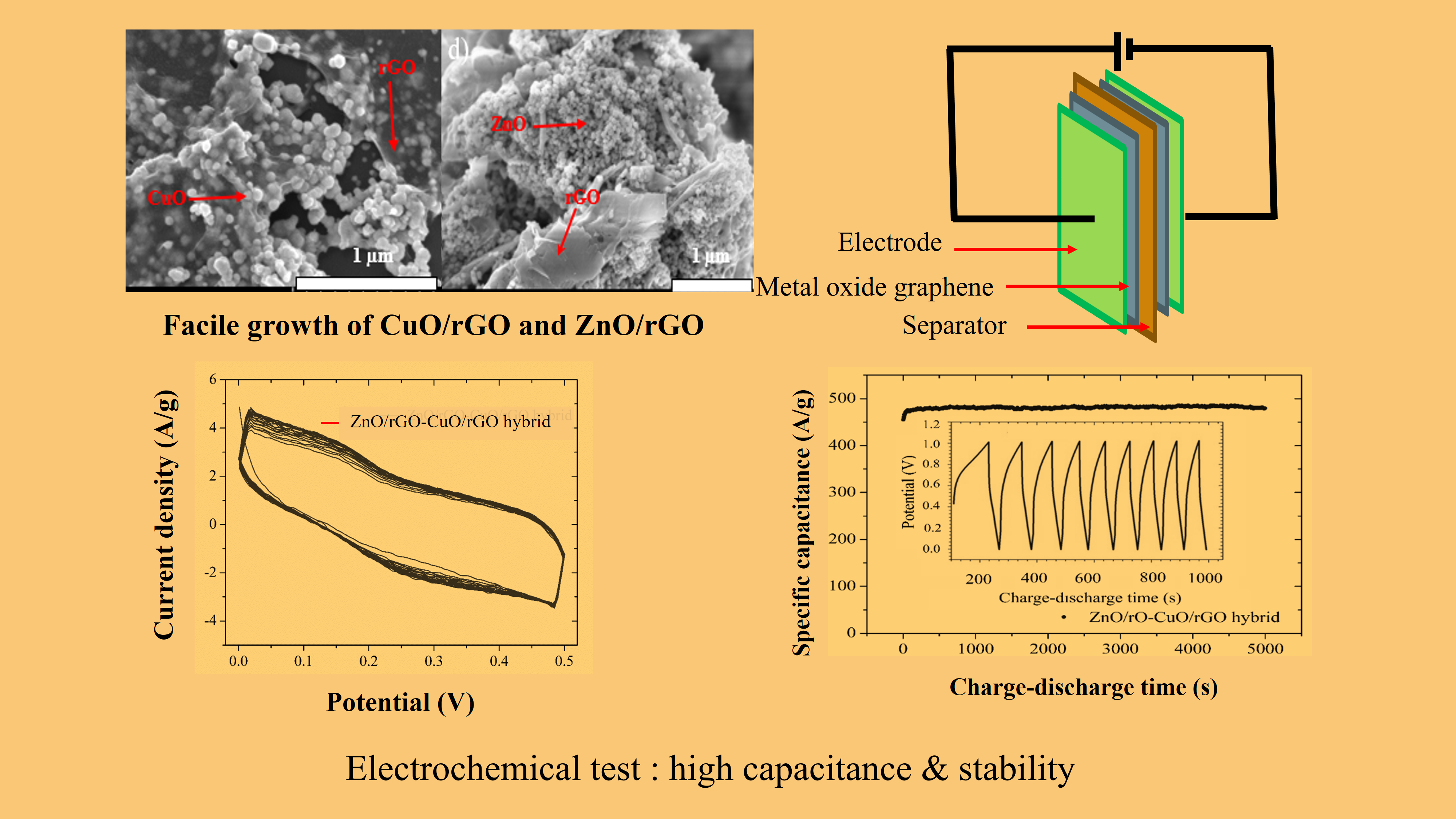
Downloads
References
V Singh, D Joung, L Zhai, S Das, SI Khondaker and S Seal. Graphene based materials: past, present and future. Progr. Mater. Sci. 2011; 56, 1178-271.
H Wang, J Lin and ZX Shen. Polyaniline (PANi) based electrode materials for energy storage and conversion. J. Sci. Adv. Mater. Dev. 2016; 1, 225-55.
M Harilal, B Vidyadharan, II Misnon, GM Anilkumar, Adrian Lowe, J Ismail, MM Yusoff and R Jose. One dimensional assembly of conductive and capacitive metal oxide electrodes for high-performance asymmetric supercapacitors. ACS Appl. Mater. Interface. 2017; 9, 10730-42.
Y Zhang, X Sun, L Pan, H Li, Z Sun, C Sun and BK Tay. Carbon nanotube-ZnO nanocomposite electrodes for supercapacitors. Solid State Ionics. 2009; 180, 1525-8.
E Frackowiak. Carbon materials for supercapacitor application. Phys. Chem. Chem. Phys. 2007; 9, 1774-85.
R Madhu, V Veeramani, SM Chen, P Veerakumar and SB Liu. Functional porous carbon/nickel oxide nanocomposites as binder-free electrodes for supercapacitors. Chem. Eur. J. 2015; 21, 8200-6.
D Majumdar, T Maiyalagan and Z Jiang. A review on recent progress in ruthenium oxide-based composites for supercapacitor applications. ChemElectroChem. 2019; 6, 4343-72.
W Xiao, H Xia, JYH Fuh and L Lu. Growth of single-crystal -MnO2 nanotubes prepared by a hydrothermal route and their electrochemical properties. J. Power Sources. 2009; 193, 935-8.
X Xu, Y Yang, M Wang, P Dong, R Baines, J Shen and M Ye. Straightforward synthesis of hierarchical Co3O4@CoWO4/rGO core-shell arrays on Ni as hybrid electrodes for asymmetric supercapacitors. Ceram. Int. 2016; 42, 10719-25.
CY Foo, A Sumboja, DJH Tan, J Wang and PS Lee. Flexible and highly scalable V2O5-rGO electrodes in an organic electrolyte for supercapacitor devices. Adv. Energy Mater. 2014; 4, 1400236.
X Zhang, H Fan, J Sun and Y Zhao. Structural and electrical properties of p-type ZnO films prepared by Ultrasonic Spray Pyrolysis. Thin Solid Films. 2007; 515, 8789-92.
Z Endut, W Basirun and M Abd Shukor. Pseudocapacitive performance of vertical copper oxide nanoflakes. Thin Solid Films. 2013; 528, 213-6.
K Bramhaiah and NS John. Facile synthesis of reduced graphene oxide films at the air-water interface and in situ loading of noble metal nanoparticles. Adv. Nat. Sci.: Nanosci. Nanotechnol. 2012; 3, 045002.
F Huang, Y Zhong, J Chen, S Li, Y Li, F Wang and S Feng. Nonenzymatic glucose sensor based on three different CuO nanomaterials. Anal. Methods. 2013; 5, 3050-5.
ER Ezeigwe, MTT Tan, PS Khiew and CW Siong. One-step green synthesis of graphene/ZnO nanocomposites for electrochemical capacitors. Ceram. Int. 2015; 41, 715-24.
R Kumar, P Rai and A Sharma. Facile synthesis of Cu2O microstructures and their morphology dependent electrochemical supercapacitors properties. RSC Adv. 2016; 6, 3815-22.
W Ly, DM Tang, YB He, CH You, ZQ Shi, XC Chen, CM Chen, PX Hou, C Liu and QH Yang. Low-temperature exfoliated graphenes: vacuum-promoted exfoliation and electrochemical energy storage. ACS nano. 2009; 3, 3730-6.
F Wang, S Xiao, Y Hou, C Hu, L Liu and Y Wu. Electrode materials for aqueous asymmetric supercapacitors. RSC Adv. 2013; 3, 13059-84.
S Liu, J Wu, J Zhou, G Fang and S Liang. Mesoporous NiCo2O4 nanoneedles grown on three dimensional graphene networks as binder-free electrode for high-performance lithium-ion batteries and supercapacitors. Electrochim. Acta. 2015; 176, 1-9.
E Mitchell, A Jimenez, RK Gupta, BK Gupta, K Ramasamy, M Shahabuddin and SR Mishra. Ultrathin porous hierarchically textured NiCo2O4/graphene oxide flexible nanosheets for high-performance supercapacitors. New J. Chem. 2015; 39, 2181-7.
ME McMahon, RJ Santucci and JR Scully. Advanced chemical stability diagrams to predict the formation of complex zinc compounds in a chloride environment. RSC Adv. 2019; 9, 19905-16.
I Puigdomenech and C Taxén. Thermodynamic data for copper: Implications for the corrosion of copper under repository conditions (No. SKB-TR--00-13). Swedish Nuclear Fuel and Waste Management, Stockholm, Sweden, 2000.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.