The Dynamics of Immature Rubber Photosynthetic Capacities Under Macronutrients Deficiencies
DOI:
https://doi.org/10.48048/tis.2023.4527Keywords:
Immature rubber, Macronutrient deficiencies, Photosynthetic capacities, Photosynthesis, RRIM 600Abstract
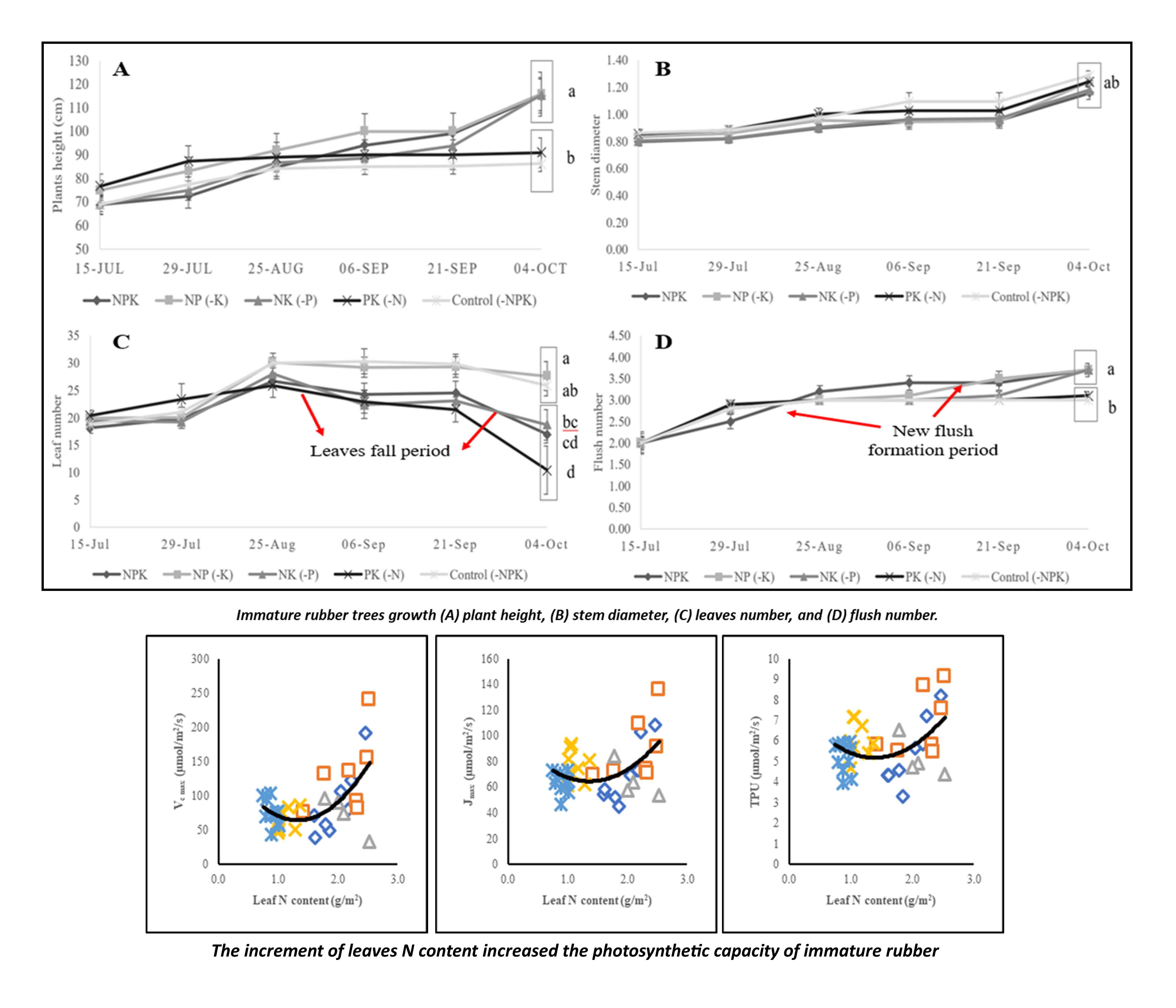
Pará rubber produces natural latex which is essential for the industries. Rubber plant in immature phase is prone to macronutrient deficiencies due to improper management practices in the field and the nature of immature plants that have sensitive physiological responses under stress conditions. The study aimed to assess the effect of macronutrient limitation on immature rubber trees’ photosynthetic capacity and growth. The immature rubber was pot-grown inside the greenhouse with a completely randomized design experiment and nutrient limitations used as the treatments. The treatments consisted of 5 levels, namely, NPK; NP (-K); NK (-P); PK (-N); Control (-NPK). Photosynthetic capacity parameters (Vc max: maximum rate RuBisCO carboxylation, Jmax: RuBP regeneration rate, and TPU: Triose Phosphate Utilization), tree growth (plant height, flush number, leaf number, stem diameter), and leaf macronutrient (N, P, and K) concentrations were periodically measured. Welsch’s test (α = 0.05) continued with Games-Howell pairwise comparison, followed by Pearson’s correlation test and polynomial regressions were performed to describe the nutrient limitation and photosynthetic capacity relationships. Results showed that the leaf nutrient concentration corresponds with the given treatments, even though it was above the critical level for immature rubber. The limitation of N fertilization slightly reduced plant development and growth such as height, leaf number, flush number, relative growth rate, and photosynthetic capacities. However, the P and K limitation effect could not be observed clearly in the observation periods on growth and photosynthetic capacity parameters. Furthermore, the mobility rate of nutrients from the soil to the plants and its translocation inside plant organs played more essential role in plant growth and photosynthetic capacities. Prolonged observation periods on various rubber clones have to be performed to deeply understand the effects of nutrient deficiencies on immature rubber tree morphophysiological activities.
HIGHLIGHTS
- Rubber plant in immature phase have sensitive physiological responses under stress conditions, and it is prone to macronutrient deficiencies due to improper management practices in the field
- Assessment of macronutrient limitation effect on immature rubber trees’ photosynthetic capacity and growth is essential to understand how the plants strive under the nutrient scarcity and providing a perspective which nutrient is more essential
- The N fertilization played more essential role compared to P and K, for immature rubber growth and photosynthetic activity
GRAPHICAL ABSTRACT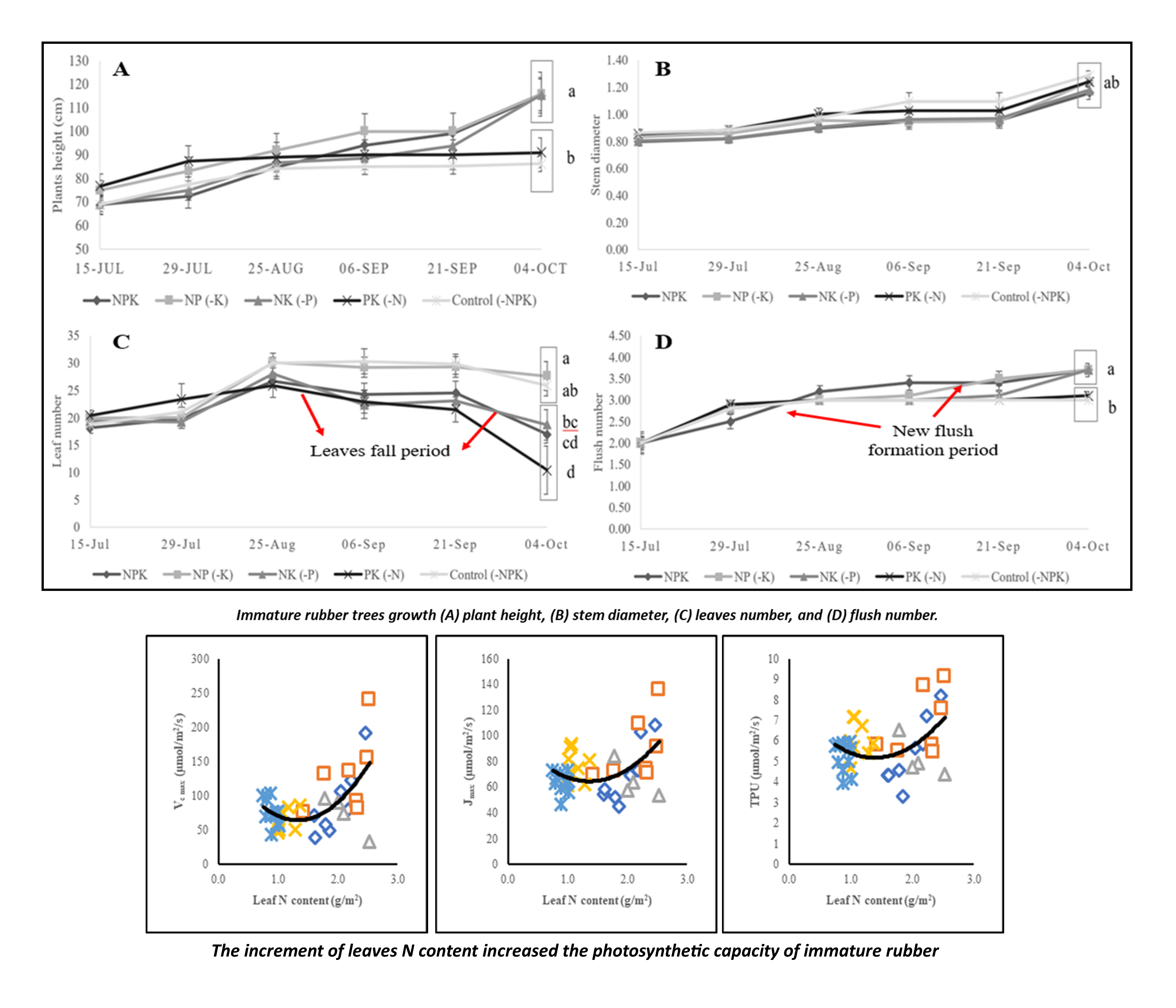
Downloads
References
I Ahmad, M Salisu and N Daud. Influence of fertilizer rates and soil series on growth performance of natural rubber (Hevea brasiliensis) latex timber clones. Aust. J. Crop Sci. 2013; 13, 1998-2004.
B Chambon, XL Dao, U Tongkaemkaew and F Gay. What determine smallholders’ fertilization practices during the mature period of rubber plantations in Thailand? Exp. Agr. 2018; 54, 824-41.
S Mak, S Chinsathit, A Pookpakdi and P Kasemsap. The effect of fertilizer and irrigation on yield and quality of rubber (Hevea brasiliensis) grown in Chanthaburi province of Thailand. Kasetsart J. Nat. Sci. 2018; 42, 226-37.
JY Alle, EA Dick, EF Soumahin, RO Gabla, JZ Keli and S Obouayeba. Effect of mineral fertilization on agrophysiological parameters and economic viability of clone PB 235 of Hevea brasiliensis in the region of GO in south western Côte d’Ivoire. J. Plant Anim. Sci. 2015; 24, 3768-80.
SJ Mokhatar, NW Daud and CF Ishak. Response of Hevea Brasiliensis (RRIM 2001) planted on an oxisol to different rates of fertilizer application. Malays. J. Soil Sci. 2012; 16, 57-69.
O Boussadia, K Steppe, H Zgallai, SBE Hadj, M Braham, R Lemeur and MCV Labeke. Effects of nitrogen deficiency on leaf photosynthesis, carbohydrate status and biomass production in two olive cultivars ‘Meski’ and ‘Koroneiki’. Sci. Horticulturae 2010; 123, 336-42.
M Hawkesford, W Horst, T Kichey, H Lambers, J Schjoerring, IS Møller and P White. Functions of macronutrients. In: Marschner’s Mineral Nutrition of Higher Plants (Ed.). Academic Press, Massachusetts, 2012, p. 135-89.
SC Pasquini and LS Santiago. Nutrients limit photosynthesis in seedlings of a lowland tropical forest tree species. Oecologia 2012; 168, 311-9.
P Marschner and Z Rengel. Nutrient Availability in Soils. In: Marschner’s Mineral Nutrition of Higher Plants (Ed.). Academic Press, Massachusetts, 2012, p. 315-30.
MAR Correia, DDC Maranhão, RA Flores, SFDS Júnior, MAD Araujo and RLDL Leite. Growth, nutrition and production of dry matter of rubber tree (Hevea brasiliensis) in function of K fertilization. Aust. J. Crop Sci. 2017; 11, 95-101.
D Epron, JP Laclau, JC Almeida, JL Goncalves, S Ponton, CR Sette and Y Nouvellon. Do changes in carbon allocation account for the growth response to potassium and sodium applications in tropical Eucalyptus plantations? Tree Physiol. 2012; 32, 667-79.
V Römheld. Diagnosis of deficiency and toxicity of nutrients. In: Marschner’s Mineral Nutrition of Higher Plants (Ed.). Academic Press, Massachusetts, 2012, p. 299-312.
S Vrignon-Brenas, F Gay, S Ricard, D Snoeck, T Perron, L Mareschal and P Malagoli. Nutrient management of immature rubber plantations. A review. Agron. Sustain. Dev. 2019; 39, 11.
P Battie-Laclau, JP Laclau, C Beri, L Mietton, MR Muniz, BC Arenque and Y Nouvellon. Photosynthetic and anatomical responses of eucalyptus grandis leaves to potassium and sodium supply in a field experiment. Plant Cell Environ.2014; 37, 70-81.
C Zörb, M Senbayram and E Peiter. Potassium in agriculture - status and perspectives. J. Plant Physiol. 2014; 171, 656-69.
SP Long and CJ Bernacchi. Gas exchange measurements, what can they tell us about the underlying limitations to photosynthesis? procedures and sources of error. J. Exp. Bot. 2003; 54, 2393-401.
TD Sharkey, CJ Bernacchi, GD Farquhar and EL Singsaas. Fitting photosynthetic carbon dioxide response curves for C(3) leaves. Plant Cell Environ. 2007; 30, 1035-40.
WJ Baulkwill and CC Webster. Rubber. John Wiley & Sons Inc, New York, 1989, p. 34-63.
B Kositsup, P Montpied, P Kasemsap, P Thaler, T Améglio and E Dreyer. Photosynthetic capacity and temperature responses of photosynthesis of rubber trees (Hevea brasiliensis Müll. Arg.) acclimate to changes in ambient temperatures. Trees 2009; 23, 357-65.
MF Morgan. Chemical soil diagnosis by the universal soil testing system. Bulletin 1941; 450, 579-626.
G Schuman, M Stanley and D Knudsen. Automated total nitrogen analysis of soil and plant samples. Soil Sci. Soc. Am. J. 1973; 37, 480-1.
CO Plank. Determination of phosphorus in plant tissue by colorimetry. Plant analysis reference procedures for the Southern region of the United States. Soutern Cooper. Bull. 1992; 368, 29-31.
CO Plank. Determination of potassium, calcium, and magnesium in plants by atomic absorption techniques. Plant analysis reference procedures for the southern region of the United States. Soutern Cooper. Bull. 1992; 368, 33-36.
T Chatzistathis and I Therios. How soil nutrient availability influences plant biomass and how biomass stimulation alleviates heavy metal toxicity in soils: The cases of nutrient use efficient genotypes and phytoremediators, respectively. IntechOpen, London, 2013.
J Onthong, K Khawmee and C Keawmano. Growth of immature rubber trees planted in abandoned paddy field and upland areas in relation to soil properties and leaf nutrients. Songklanakarin J. Sci. Tech. 2017; 39, 565-695.
R Ardika, PB Sanchez, RB Badayos and PCS Cruz. Growth of PB 260 clone (Hevea brasiliensis (Willd. ex A. Juss.) Muell-Arg.) in different potting media and fertilization scheme. Agrivita J. Agr. Sci. 2017; 39, 956.
I Damrongrak, J Onthong and C Nilnond. Effect of fertilizer and dolomite applications on growth and yield of tapping rubber trees. Songklanakarin J. Sci. Tech. 2015; 37, 643-50.
S Thitithanakul, N Ma, S Sukkawong and B Jaikrajang. Determination of nitrogen and phosphorus requirement of the RRIM 600 and RRIT 251 young rubber trees. Walailak J. Sci. Tech. 2017; 14, 571-80.
T Balemi and N Kefyalew. Management of soil phosphorus and plant adaptation mechanisms to phosphorus stress for sustainable crop production: A review. J. Soil Sci. Plant Nutr. 2012; 12, 547-62.
Z Yan, A Eziz, D Tian, X Li, X Hou, H Peng, W Han, Y Guo and J Fang. Biomass allocation in response to nitrogen and phosphorus availability: insight from experimental manipulations of Arabidopsis thaliana. Front. Plant Sci. 2019; 10, 598.
KR Kramer-Walter and DC Laughlin. Root nutrient concentration and biomass allocation are more plastic than morphological traits in response to nutrient limitation. Plant Soil 2017; 416, 539-50.
B Yan, Z Ji, B Fan, X Wang, G He, L Shi and G Liu. Plants adapted to nutrient limitation allocate less biomass into stems in an arid-hot grassland. New Phytologist 2016; 211, 1232-40.
DL Achat, N Pousse, M Nicolas and L Augusto. Nutrient remobilization in tree foliage as affected by soil nutrients and leaf life span. Ecol. Monogr. 2018; 88, 408-28.
L Taiz and E Zeiger. Plant physiology. Sinauer Associates, Massachusetts, 2012.
WH Loescher, T Mccamant and JD Keller. Carbohydrate reserves, translocation, and storage in woody plant roots. Horticultural Sci. 1990; 25, 274-81.
PM Schleuss, M Widdig, A Heintz-Buschart, K Kirkman and M Spohn. Interactions of nitrogen and phosphorus cycling promote P acquisition and explain synergistic plant-growth responses. Ecology 2020; 101, e03003
VD Fageria. Nutrient interactions in crop plants. J. Plant Nutr. 2001; 24, 1269-90.
D Reuter and JB Robinson. Plant analysis: An interpretation manual. CSIRO Publishing, Clayton, Australia, 1997.
JL Bubier, R Smith, S Juutinen, TR Moore, R Minocha, S Long and S Minocha. Effects of nutrient addition on leaf chemistry, morphology, and photosynthetic capacity of three bog shrubs. Oecologia 2011; 167, 355-68.
K Qiu, Y Xie, D Xu, T Qi and R Pott. Photosynthesis-related properties are affected by desertification reversal and associated with soil N and P availability. Braz. J. Bot. 2018; 41, 329-36.
AP Walker, AP Beckerman, L Gu, J Kattge, LA Cernusak, TF Domingues and FI Woodward. The relationship of leaf photosynthetic traits-Vcmax and Jmax-to leaf nitrogen, leaf phosphorus, and specific leaf area: A meta‐analysis and modeling study. Ecol. Evol. 2014; 4, 3218-35.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






