The Protective Role of Scoparia Dulcis Linn. in Alzheimer’s Disease
DOI:
https://doi.org/10.48048/tis.2022.2050Keywords:
Scoparia dulcis Linn., Phytochemical profile, Protein damage, Lipid damage, Acetylcholinesterase activityAbstract
Alzheimer’s disease (AD) is a neurodegenerative disorder that appears simultaneously with age. AD is caused by oxidative stress, which generates the oxidation of biomolecules such as DNA, proteins, and lipids. The present study aimed to evaluate protein and lipid protection against damage caused by the free radical and anticholinesterase properties of Scoparia dulcis, which are relevant to AD therapy. Furthermore, phytochemical profiling of S. dulcis extract was also observed. Quantitative phytochemical (phenolic, flavonoid and tannin contents) analysis of methanol, butanol, and ethyl acetate fraction substances in S. dulcis was performed by standard spectrophotometric methods. Butanolic extracts showed maximum amounts of phytochemicals, including phenolics, flavonoids, and tannins. The butanolic extract also showed the highest acetylcholinesterase potential inhibition and DPPH radical scavenging, with IC50 values of 93.24 and 22.8 μg/mL, respectively, in a dose-dependent manner. Additionally, the butanol fraction exhibited strong FeSO4-induced lipid peroxidation inhibition. The best free-radical-induced protein oxidation inhibitory activity was observed in methanol samples. In conclusion, this study suggests that S. dulcis is a potential agent for drug development against AD.
HIGHLIGHTS
- Butanolic Scoparia dulcis extract exhibited maximum amounts of phenolic, flavonoid, and tannins
- Butanolic fraction of dulcis extracts was the most active AChEI activity and DPPH radical scavenging
- Scoparia dulcis showed strong free-radical-induced lipid and protein damage inhibition
- Butanolic Scoparia dulcis warrant further investigation for Alzheimer's disease treatment
GRAPHICAL ABSTRACT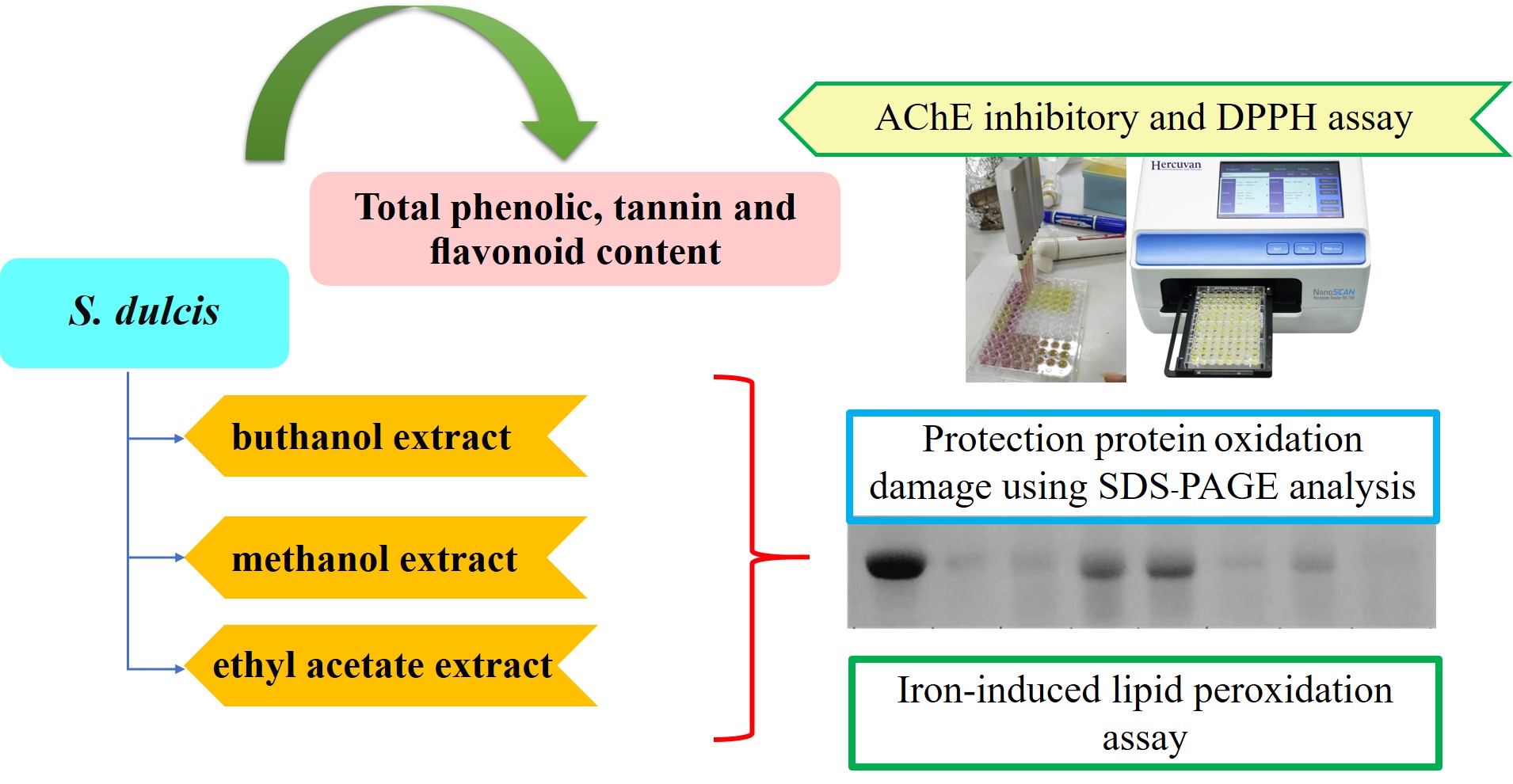
Downloads
References
P Scheltens, K Blennow, MMB Breteler, BD Strooper, GB Frisoni, S Salloway and WMVD Flier. Alzheimer’s disease. Lancet 2016; 388, 505-17.
A Sanabria-Castro, I Alvarado-Echeverría and C Monge-Bonilla. Molecular pathogenesis of
Alzheimer’s disease: An Update. Ann Neurosci. 2017; 24, 46-54.
WJ Deardorff and GT Grossberg. Pharmacotherapeutic strategies in the treatment of severe Alzheimer's disease. Expet. Opin. Pharmacother. 2016; 17, 1789-800.
CC Tan, JT Yu, HF Wang, MS Tan, XF Meng, C Wang, T Jiang, XC Zhu and L Tan. Efficacy and safety of donepezil, galantamine, rivastigmine, and memantine for the treatment of Alzheimer's disease: A systematic review and meta-analysis. J. Alzheimers Dis. 2014; 41, 615-31.
S Elangovan and RMD Holsinger. Cyclical amyloid beta-astrocyte activity induces oxidative stress in Alzheimer’s disease. Biochimie 2020; 171-172, 38-42.
ASM Ali Reza, MS Hossain, S Akhter, MR Rahman, MS Nasrin, MJ Uddin, G Sadik and AHM Khurshid Alam. In vitro antioxidant and cholinesterase inhibitory activities of Elatostema papillosum leaves and correlation with their phytochemical profiles: A study relevant to the treatment of Alzheimer’s disease. BMC Compl. Alternative Med. 2018; 18, 123.
AK Sahoo, J Dandapat, UC Dash and S Kanhar. Features and outcomes of drugs for combination
therapy as multi-targets strategy to combat Alzheimer’s disease. J. Ethnopharmacol. 2018; 215,
-73.
M Penumala, RB Zinka, JB Shaik and DA Gangaiah. In vitro screening of three indian medicinal plants for their phytochemicals, anticholinesterase, antiglucosidase, antioxidant, and neuroprotective effects. BioMed Res. Int. 2017; 2017, 5140506.
JE Beh, J Latip, MP Abdullah, A Ismail and M Hamid. Scoparia dulcis (SDF7) endowed with glucose uptake properties on L6 myotubes compared insulin. J. Ethnopharmacol. 2010; 129, 23-33.
W Wu, T Chen, R Lu, S Chen and C Chang. Benzoxazinoids from Scoparia dulcis (sweet broomweed) with antiproliferative activity against the DU-145 human prostate cancer cell line. Phytochemistry 2012; 83, 110-5.
N Chana, P Supaphon and A Phongdara. Evaluation of antibacterial, antioxidant activity and calmodulin gene expression of Scoparia dulcis Linn. Songklanakarin J. Sci. Tech. 2019; 41, 246-53.
W Wankhar, S Srinivasan, R Rajan and R Sheeladevi. Antioxidant mediated response of Scoparia
dulcis in noise-induced redox imbalance and immunohistochemical changes in rat brain. J. Biomed.
Res. 2017; 31, 143-53.
MVV Lima, ADO Freire, ELF Sousa, AAM Vale, AJO Lopes, CC Vasconcelos, MVV Lima-Aragão, HO Serra, RNMG Liberio, APSDAD Santos, GEB Silva, CQD Rocha, FCVM Lima, MDSDS Cartágenes and JBS Garcia. Therapeutic use of Scoparia dulcis reduces the progression of experimental osteoarthritis. Molecules 2019; 24, 3474.
W Wankhar, S Srinivasan, L Sundareswaran, D Wankhar, R Rajan and R Sheeladevi. Role of Scoparia dulcis linn on noise-induced nitric oxide synthase (NOS) expression and neurotransmitter assessment on motor function in Wistar albino rats. Biomed. Pharmacother. 2017; 86, 475-81.
D Kim, SW Jeong and CY Lee. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 2003; 81, 321- 6.
A Rebaya, SL Belghith, B Baghdikian, VM Leddet, F Mabrouki, E Olivier, JK Cherif and MT Ayadi. Total phenolic, total flavonoid, tannin content, and antioxidant capacity of Halimium halimifolium (Cistaceae). J. Appl. Pharm. Sci. 2015; 5, 52-7.
R Upadhyay, JK Chaurasia, KN Tiwari and K Singh. Antioxidant property of aerial parts and root of Phyllanthus fraternus Webster, an important medicinal plant. Sci. World J. 2014; 24, 692392.
S Mohan, K Thiagarajan and R Chandrasekaran. In vitro protection of biological macromolecules against oxidative stress and in vivo toxicity evaluation of Acacia nilotica (L.) and ethyl gallate in rats. BMC Compl. Alternative Med. 2014; 14, 257.
GL Ellman, KD Courtney, V Andreas and RM Featherstone. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961; 7, 88-90.
H Khan, Marya, S Amin, MA Kamal and S Patel. Flavonoids as acetylcholinesterase inhibitors: current therapeutic standing and future prospects. Biomed. Pharmacother. 2018; 101, 860-70.
WMADB Fernando, G Somaratne, KG Goozee, S Williams, H Singh and RN Martins. Diabetes and Alzheimer’s disease: Can tea phytochemicals play a role in prevention? J. Alzheimers Dis. 2017; 59, 481-501.
E Wightman. Potential benefits of phytochemicals against Alzheimer's disease. Proc.Nutr. Soc. 2017; 76, 106-12.
G D’Onofrio, D Sancarlo, Q Ruan, Z Yu, F Panza, A Daniele, A Greco and D Seripa. Phytochemicals in the treatment of Alzheimer's disease: A systematic review. Curr. Drug Targets. 2017; 18, 1487-98.
MB Colović, DZ Krstić, TD Lazarević-Pašti, AM Bondžić and VM Vasić. Acetylcholinesterase
inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013; 11, 315-35.
M Mathew and S Subramanian. In vitro screening for anti-cholinesterase and antioxidant activity of methanolic extracts of ayurvedic medicinal plants used for cognitive disorders. PloS one 2014; 9, e86804.
AY Coulibaly, M Kiendrebeogo, PG Kehoe, PAED Sombié, CE Lamien, JF Millogo and OG Nacoulma. Antioxidant and anti-Inflammatory effects of Scoparia dulcis L. J. Med. Food. 2011; 14, 1576-82.
M Latha, L Pari, S Sitasawad and R Bhonde. Scoparia dulcis, a traditional antidiabetic plant, protects against streptozotocin induced oxidative stress and apoptosis in vitro and in vivo. J. Biochem. Mol. Toxicol. 2004; 18, 261-72.
N Chana and J Chaikaew. DNA damage protection activities of different solvents from Scoparia dulcis leaf and stem extracts. Khonkaen Agr. J. 2018; 46, 397-408.
Q Liu, Q Yang, H Hu, L Yang, Y Yang, G Chou and Z Wang. Bioactive diterpenoids and flavonoids from the aerial parts of Scoparia dulcis. J. Nat. Prod. 2014; 77, 1594-600.
J Wojsiat, KM Zoltowska, K Laskowska-Kaszub and U Wojda. Oxidant/antioxidant imbalance in Alzheimer’s disease: Therapeutic and diagnostic prospects. Oxid. Med. Cell. Longev. 2018; 2018, 6435861.
MA Lovell and WR Markesbery. Oxidative DNA damage in mild cognitive impairment and late-stage Alzheimer’s disease. Nucleic Acids Res. 2007; 35, 7497-504.
Q Kong and CG Lin. Oxidative damage to RNA: Mechanisms, consequences, and diseases. Cell. Mol. Life Sci. 2010; 67, 1817-29.
V Cecarini, J Gee, E Fioretti, M Amici, M Angeletti, AM Eleuteri and JN Keller. Protein oxidation and cellular homeostasis: Emphasis on metabolism. Biochim. Biophys. Acta. 2007; 1773, 93-104.
LJ Su, JH Zhang, H Gomez, R Murugan, X Hong, D Xu, F Jiang and ZY Peng. Reactive oxygen species-induced lipid peroxidation in apoptosis, autophagy, and ferroptosis. Oxidative Med. Cell. Longevity. 2019; 2019, 5080843.
Y Kao, P Ho, Y Tu, I Jou and K Tsai. Lipids and Alzheimer’s Disease. Int. J. Mol. Sci. 2020; 21, 1505.
DA Butterfield and D Boyd-Kimball. Oxidative stress, amyloid-β Peptide, and altered key molecular pathways in the pathogenesis and progression of Alzheimer’s disease. J. Alzheimers Dis. 2018; 62, 1345-67.
LJ Lizcano, M Viloria-Bernal, F Vicente, LA Berrueta, B Gallo, M Martínez-Cañamero, MB Ruiz-Larrea and JI Ruiz-Sanz. Lipid oxidation inhibitory effects and phenolic composition of aqueous extracts from medicinal plants of Colombian Amazonia. Int. J. Mol. Sci. 2012; 13, 5454-67.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







