Effect of pH and Calcination Temperature on the Structural, Optical, Electrical, and Magnetic Properties of Co-Precipitated Iron Oxide Nanomaterials
DOI:
https://doi.org/10.48048/tis.2026.12091Keywords:
Iron oxide, Co-precipitation, Optical property, Energy band gap, Electrical resistance, Electrical conductivity, Magnetic properties, Coercivity, RemanenceAbstract
Iron oxide nanomaterials were synthesized by co-precipitation, with pH adjusted from 9 to 12, and subsequent calcination carried out between 300 and 900 °C. The as-synthesized samples were characterized for morphology, crystal structure, and functional properties. At pH 11, nearly spherical nanoparticles with uniform size and high crystallinity were obtained, giving the highest saturation magnetization of 121.97 emu/g and a narrow indirect optical band gap of 2.03 eV, beneficial for magnetic hyperthermia and visible-light photocatalysis. Calcination at 500 °C retained the γ-Fe2O3 phase and yielded peak electrical conductivity (98.62×10−9 (Ω·cm)−1) together with near-superparamagnetic behavior with low coercivity and remanence, making it suitable for spintronics and biomedical applications. In contrast, annealing above 500 °C triggered irreversible conversion to antiferromagnetic α-Fe2O3, which sharply reduced magnetization and increased electrical resistivity. Combined structural, optical, and magnetic data indicate that pH 11 with 500 °C provides the most favorable trade-off between defect density, phase stability, and interparticle connectivity. Therefore, this work demonstrates that the dual-parameter control of pH and calcination temperature allows the optimization of structural, optical, electrical, and magnetic properties, enabling the development of multifunctional iron oxide nanomaterials for catalysis, biomedicine, and next-generation electronic devices.
HIGHLIGHTS
- Iron oxide nanomaterials were synthesized by co-precipitation and tuned by varying pH (9 - 12) and calcination temperature (300 - 900 °C).
- pH 11 produced uniform spherical nanoparticles with reduced spin disorder, yielding the highest magnetization (121.97 emu/g).
- Calcination at 500 °C was identified as the “optimum temperature,” maximizing electrical conductivity and near-superparamagnetic behavior.
- Higher calcination temperatures (> 700 °C) triggered a phase transition from maghemite to hematite, causing loss of magnetism and higher resistivity.
- Optical band gap tuning was achieved: Stable 2.03 eV at pH 9 - 11 and widened to 2.12 eV at pH 12 due to reduced defect states.
- Dual optimization (pH 11 and 500 °C) provides a simple experimental strategy for multifunctional nanomaterials in spintronics, catalysis, and biomedicine.
GRAPHICAL ABSTRACT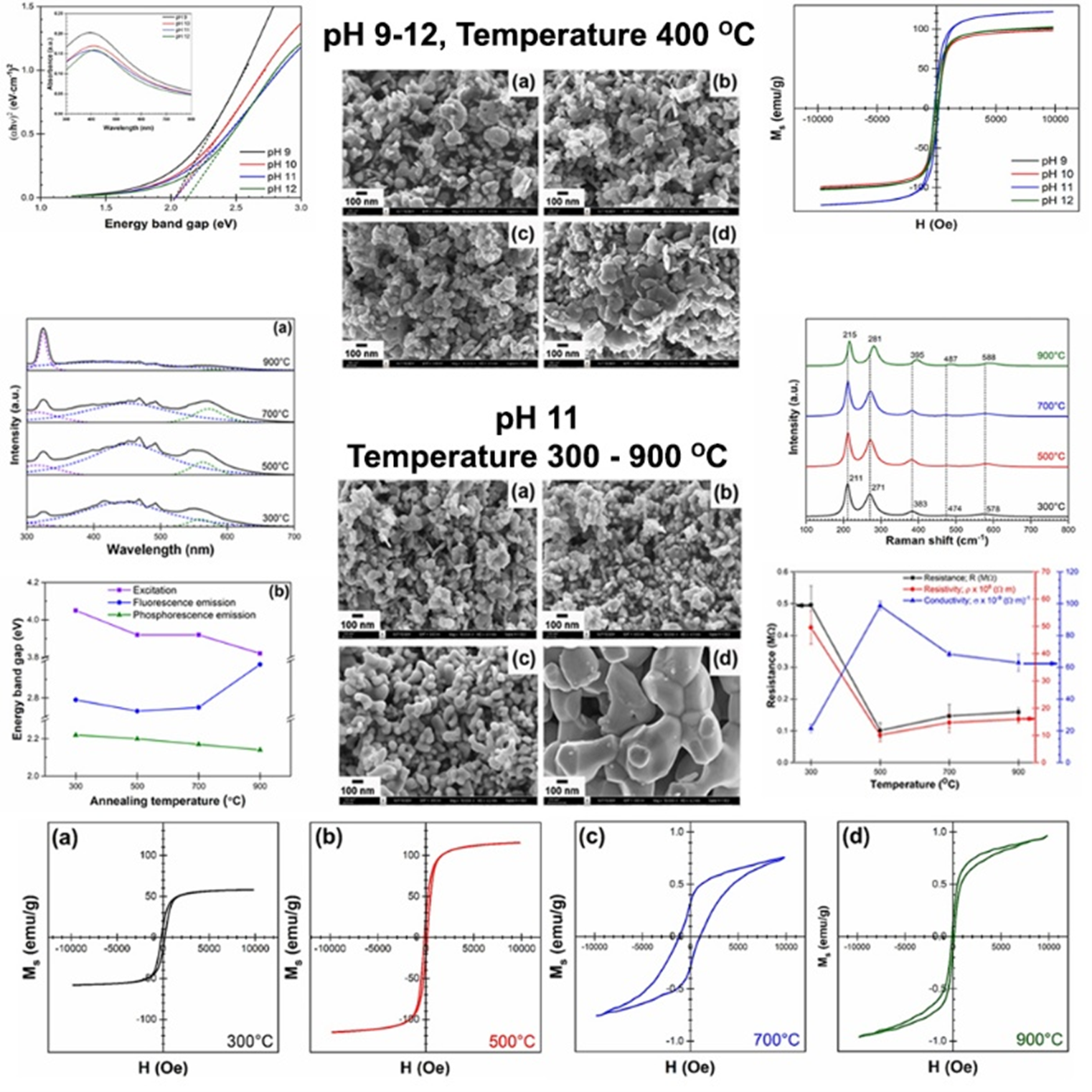
Downloads
References
H Gleiter. Nanostructured materials: Basic concepts and microstructure. Acta Materialia 2000; 4(1), 1-29.
JE Ogbezode, US Ezealigo, A Bello, VC Anye and AP Onwualu. A narrative review of the synthesis, characterization, and applications of iron oxide nanoparticles. Discover Nano 2023; 18, 125.
AK Gupta and M Gupta. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005; 26(18), 3995-4021.
DS Chaudhari, RP Upadhyay, GY Shinde, MB Gawande, J Filip, RS Varma and R Zbořil. A review on sustainable iron oxide nanoparticles: Synthesis and application in organic catalysis and environmental remediation. Green Chemistry 2024; 26, 7579-7655.
S Laurent, D Forge, M Port, A Roch, C Robic, LV Elst and RN Muller. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chemical Reviews 2008; 108(6), 2064-2110.
AH Lu, EL Salabas and F Schüth. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angewandte Chemie International Edition 2007; 46(8), 1222-1244.
MA Gabal, A Alshammari, H Alotaibi and S Alharbi. Cytotoxicity and hemostatic 1 step green synthesis of iron nanoparticles coated with green tea for biomedical application. Trends in Sciences 2022; 19(3), 2062.
J Yuennan, N Muensit, N Tohluebaji, W Chailad, L Yang, N Sukhawipat, GA Ashraf and P Channuie. Tailoring dielectric properties and crystallinity in poly(vinylidene fluoride-co-hexafluoropropylene) nanocomposites via iron (III) chloride hexahydrate incorporation. Scientific Reports 2025; 15, 17810.
J Yuennan, S Al-Sulaimi, M Karimov, A Abduvokhidov, M Mirzayev, GA Ashraf, Z Javed, A Ayari-Akkari, RM Ali, A Guesmi and P Channuie. Photocatalytic degradation of tetracycline via peroxymonosulfate activation via W0.5Ag0.5FeO3: A synergistic approach for sustainable water treatment. Vacuum 2025; 241, 114639.
N Lertcumfu, FN Sayed, SN Shirodkar, S Radhakrishnan, A Mishra, G Rujijanagul, AK Singh, BI Yakobson, CS Tiwary and PM Ajayan. Structure-dependent electrical and magnetic properties of iron oxide composites. Physica Status Solidi (A) 2019; 216(16), 1801004.
S Singh and N Goswami. Structural, magnetic and dielectric study of Fe2O3 nanoparticles obtained through exploding wire technique. Current Applied Physics 2021; 20, 20-29.
YP Yew, K Shameli, M Miyake, NBBA Khairudin, SEB Mohamad, T Naiki and KX Lee. Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: A review. Arabian Journal of Chemistry 2020; 13(1), 2287-2308.
K Sivula, FL Formal and M Grätzel. Solar water splitting: Progress using hematite (α-Fe2O3) photoelectrodes. ChemSusChem 2011; 4(4), 432-449.
W Wu, Q He and X Zhong. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Research Letters 2008; 3, 397-415.
S Sun, H Zeng, DB Robinson, S Raoux, PM Rice, SX Wang and G Li. Monodisperse MFe2O4 (M = Fe, Co, Mn) nanoparticles. Journal of the American Chemical Society 2004; 126(1), 273-279.
M Unni, AM Uhl, S Savliwala, BH Savitzky, R Dhavalikar, N Garraud, DP Arnold, LF Kourkoutis, JS Andrew and C Rinaldi. Thermal decomposition synthesis of iron oxide nanoparticles with diminished magnetic dead layer by controlled addition of oxygen. ACS Nano 2017; 11(2), 2284-2303.
MO Besenhard, AP LaGrow, A Hodzic, M Kriechbaum, L Panariello, G Bais, K Loizou, S Damilos, MM Cruz, NTK Thanh and A Gavriilidis, Co-precipitation synthesis of stable iron oxide nanoparticles with NaOH: New insights and continuous production via flow chemistry. Chemical Engineering Journal 2020; 399, 125740.
Y Lu, Y Yin, BT Mayers and Y Xia. Modifying the surface properties of superparamagnetic iron oxide nanoparticles through a sol-gel approach. Nano Letters 2002; 2(3), 183-186.
A Ghosh, V Srinivas and R Sundara. Comprehensive structural and magnetic properties of iron oxide nanoparticles synthesized through chemical routes. Journal of Alloys and Compounds 2020; 818, 152931.
VG Ilves, N Pizúrová, PM Korusenko, SY Sokovnin, ME Balezin, AS Gerasimov, MA Uimin, MG Zuev and AA Vasin. Effect of air annealing on properties of maghemite nanoparticles produced by radiation-chemical method. Ceramics International 2023; 49(15), 25414-25426.
A Lassoued, MS Lassoued, B Dkhil, S Ammar and A Gadri. Synthesis, photoluminescence and magnetic properties of iron oxide (α-Fe2O3) nanoparticles through precipitation or hydrothermal methods. Physica E: Low-dimensional Systems and Nanostructures 2018; 101, 212-219.
Ritik, S Sarkar, Pragti, N Varshney, A Kumar, ML Kuznetsov, HC Jha and S Mukhopadhyay. Nano-Structured Gel Materials for Environmental Remediation and Biomedical Applications. ACS Applied Nano Materials 2024; 7(18), 22292-22303.
W Wu, Z Wu, T Yu, C Jiang and WS Kim. Recent progress on magnetic iron oxide nanoparticles: synthesis, surface functional strategies and biomedical applications. Science and Technology of Advanced Materials 2015; 16, 023501.
P Kingpho and B Toboonsung. Improvement of the electrical properties of ZnO nanomaterials with Fe by co-precipitation method. Current Applied Science and Technology 2025; 25(3), e0263485.
B Toboonsung. Structure, magnetic property and energy band gap of Fe-doped NiO nanoparticles prepared by co-precipitation method. Key Engineering Materials 2017; 751, 379-383.
F Naushin, S Sen, M Kumar, H Bairagi, S Maiti, J Bhattacharya and S Sen. Structural and surface properties of pH-varied Fe2O3 nanoparticles: Correlation with antibacterial properties. ACS Omega 2024; 9(1), 464-473.
FN Sayed and V Polshettiwar. Facile and sustainable synthesis of shaped iron oxide nanoparticles: Effect of iron precursor salts on the shapes of iron oxides. Scientific Reports 2015; 5, 9733.
A Adel, R Locif, BI Yaacoub, H Abdelmadjid, R Selma and B Imed-Eddine. pH-dependent deposition of Cu2O thin films: Tuning defect states and electronic properties for improved energy conversion applications. Applied Physics A 2025; 131(105), 1-21.
B Choudhury, S Bayan, A Choudhury and P Chakraborty. Narrowing of band gap and effective charge carrier separation in oxygen-deficient TiO2 nanotubes with improved visible light photocatalytic activity. Journal of Colloid and Interface Science 2016; 465, 1-10.
N Tohluebaji, R Siri, N Muensit, C Putson, P Channuie,P Porrawatkul and J Yuennan. Hydrophobic and optical properties of p(vdf-hfp)nanofiber filled with nickel (ii) chloride hexahydrate for dye-sensitized solar cells application. Trends in Sciences 2024; 21(9), 8762.
VB Kamble and AM Umarji. Defect induced optical bandgap narrowing in undoped SnO2 nanocrystals. AIP Advances 2013; 3(8), 082120.
JC Mora, YCM Nederstigt, JM Hill and S Ponnurangam. Promoting effect of supports with oxygen vacancies as extrinsic defects on the reduction of iron oxide. The Journal of Physical Chemistry C 2021; 125(26), 14299-14310.
J Wang, Z Wang, B Huang, Y Ma, Y Liu, X Qin, X Zhang and Y. Dai. Oxygen vacancy induced band-gap narrowing and enhanced visible light photocatalytic activity of ZnO. ACS Applied Materials & Interfaces 2012; 4(8), 4024-4030.
P Kushwaha and P Chauhan. Influence of different surfactants on morphological, structural, optical, and magnetic properties of α-Fe2O3 nanoparticles synthesized via co-precipitation method. Applied Physics A 2022; 128(18), 1-14.
D Nadhiya, A Kala, P Sasikumar, MKA Mohammed, P Thirunavukkarasu, M Prabhaharan, C Karnan, S Albukhaty, MS Jabir, A Syed, AM Elgorban and NSS Zaghloul. Influence of Cu2+ substitution on the structural, optical, magnetic, and antibacterial behaviour of zinc ferrite nanoparticles. Journal of Saudi Chemical Society 2023; 27(5), 101696.
MM Arman and SI El-Dek. Structural, surface, magnetic study and application of nanoparticles CoFe2O4, ZnO and its nanocomposite. Journal of Superconductivity and Novel Magnetism 2023; 36, 1913-1925.
M Filippousi, M Angelakeris, M Katsikini, E Paloura, I Efthimiopoulos, Y Wang, D Zamboulis and G Van Tendeloo. Surfactant effects on the structural and magnetic properties of iron oxide nanoparticles. The Journal of Physical Chemistry C 2014; 118(29), 16209-16217.
X Batlle, C Moya, M Escoda-Torroella, Ò Iglesias, AF Rodríguez and A Labarta. Magnetic nanoparticles: From the nanostructure to the physical properties. Journal of Magnetism and Magnetic Materials 2022; 543, 168594.
A Lassoued, MS Lassoued, B Dkhil, S Ammar and A Gadri. Synthesis, structural, morphological, optical and magnetic characterization of iron oxide (α-Fe2O3) nanoparticles by precipitation method: Effect of varying the nature of precursor. Physica E: Low-dimensional Systems and Nanostructures 2018; 97, 328-334.
ACB Jesus, JR Jesus, RJS Lima, KO Moura, JMA Almeida, JGS Duque and CT Meneses. Synthesis and magnetic interaction on concentrated Fe3O4 nanoparticles obtained by the co-precipitation and hydrothermal chemical methods. Ceramics International 2020; 46(8), 11149-11153.
APA Faiyas, EM Vinod, J Joseph, R Ganesan and RK Pandey. Dependence of pH and surfactant effect in the synthesis of magnetite (Fe3O4) nanoparticles and its properties. Journal of Magnetism and Magnetic Materials 2010; 322(4), 400-404.
J Lei, Z Luo, S Qing, X Huang and F Li. Effect of surfactants on the stability, rheological properties, and thermal conductivity of Fe3O4 nanofluids. Powder Technology 2022; 399, 117197.
AR Chakraborty, FTZ Toma, K Alam, SB Yousuf and KS Hossain. Influence of annealing temperature on Fe2O3 nanoparticles: Synthesis optimization and structural, optical, morphological, and magnetic properties characterization for advanced technological applications. Heliyon 2024; 10(21), e40000.
P Kushwaha and P Chauhan. Influence of annealing temperature on microstructural and magnetic properties of Fe2O3 nanoparticles synthesized via sol-gel method. Inorganic and Nano-Metal Chemistry 2022; 52(7), 937-950.
DK Bora, A Braun, S Erat, O Safonova, T Graule and EC Constable. Evolution of structural properties of iron oxide nanoparticles during temperature treatment from 250 °C - 900 °C: X-ray diffraction and Fe K-shell pre-edge X-ray absorption study. Current Applied Physics 2012; 12(3), 817-825.
E Darezereshki, F Bakhtiari, M Alizadeh, AB Vakylabad and M Ranjbar. Direct thermal decomposition synthesis and characterization of hematite (α-Fe2O3) nanoparticles. Materials Science in Semiconductor Processing 2012; 15(1), 91-97.
A Lassoued, MS Lassoued, B Dkhil, A Gadri and S Ammar. Synthesis, structural, optical and morphological characterization of hematite through the precipitation method: Effect of varying the nature of the base. Journal of Molecular Structure 2017; 1141(5), 99-106.
M Cho, EQ Contreras, SS Lee, CJ Jones, W Jang and VL Colvin. Characterization and optimization of the fluorescence of nanoscale iron oxide/quantum dot complexes. The Journal of Physical Chemistry C 2014; 118(26), 14606-14616.
S Sudewi, CH Li, S Dayalan, M Zulfajri, PV Sai Sashankh and GG Huang. Enhanced fluorescent iron oxide quantum dots for rapid and interference free recognizing lysine in dairy products. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2022; 279(15), 21453.
S Pujar and GK Rao. Annealing induced strong NBE emission of SILAR deposited ZnO thin films. Materials Today: Proceedings 2022; 55, 56-61.
S Arsalani, J Oliveira, EJ Guidelli, JFDF Araujo, F Wiekhorst and O Baffa. Synthesis of radioluminescent iron oxide nanoparticles functionalized by anthracene for biomedical applications. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2020; 602, 125105.
AL Flores, N Medina-Berríos, W Pantoja-Romero, DB Plaza, K Kisslinger, J Beltran-Huarac, G Morell and BR Weiner. Geometry and surface area optimization in iron oxide nanoparticles for enhanced magnetic properties. ACS Omega 2024; 9(30), 32980-32990.
A Tufani and A Qureshi. Iron oxide nanoparticles based magnetic luminescent quantum dots (MQDs) synthesis and biomedical/biological applications: A review. Materials Science and Engineering: C 2021, 118, 111545.
N Chekina, D Horák, P Jendelová, M Trchová, MJ Beneš, M Hrubý, V Herynek, K Turnovcová and E Syková. Fluorescent magnetic nanoparticles for biomedical applications. Journal of Materials Chemistry 2011; 21, 7630-7639.
X Sun, A Tayal, A Ullrich, O Petracic and S Haas. Phase composition of iron oxide nanoparticles studied using hard X-ray absorption spectroscopy. The Journal of Physical Chemistry C 2023; 127(25), 12077-12083.
H Donya, TA Taha, A Alruwaili, IBI Tomsah and M Ibrahim. Micro-structure and optical spectroscopy of PVA/iron oxide polymer nanocomposites. Journal of Materials Research and Technology 2020; 9(4), 9189-9194.
MY Liao, CH Wu, PS Lai, J Yu, HP Lin, TM Liu and CC Huang. Surface state mediated NIR 2-photon fluorescence of iron oxides for nonlinear optical microscopy. AIP Advances 2012; 23(16), 2044-2051.
I Castellanos-Rubio, M Insausti, E Garaio, IG Muro, F Plazaola, T Rojo and L Lezama. Fe3O4 nanoparticles prepared by the seeded-growth route for hyperthermia: electron magnetic resonance as a key tool to evaluate size distribution in magnetic nanoparticles. Nanoscale 2014; 6, 7542-7552.
SW Ge, XY Wang, T Chang, B Chen, P Hu, FF Yang, Q Cao, F Yang, L Kang and KS Wang. Fe3O4 nanoparticles synthesized by 1-step reduction with nanoscale size-dependent magnetic properties. Journal of Sol-Gel Science and Technology 2023; 105, 98-105.
SA Kulkarni, PS Sawadh, PK Palei and KK Kokate. Effect of synthesis route on the structural, optical and magnetic properties of Fe3O4 nanoparticles. Ceramics International 2014; 40(1), 1945-1949.
P Hu, S Zhang, H Wang, D Pan, J Tian, Z Tang and AA Volinsky. Heat treatment effects on Fe3O4 nanoparticles structure and magnetic properties prepared by carbothermal reduction. Journal of Alloys and Compounds 2011; 509(5), 2316-2319.
AH Rezayan, M Mousavi, S Kheirjou, G Amoabediny, MS Ardestani and J Mohammadnejad. Monodisperse magnetite (Fe3O4) nanoparticles modified with water soluble polymers for the diagnosis of breast cancer by MRI method. Journal of Magnetism and Magnetic Materials 2016; 420, 210-217.
S Wu, A Sun, FQ Zhai, J Wang, WH Xu, Q Zhang and AA Volinsky. Fe3O4 magnetic nanoparticles synthesis from tailings by ultrasonic chemical co-precipitation. Materials Letters 2011; 65(12), 1882-1884.
H Zhang and GQ Zhu. One-step hydrothermal synthesis of magnetic Fe3O4 nanoparticles immobilized on polyamide fabric. Applied Surface Science 2012; 258(11), 4952-4959.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






