Advances and Challenges in Serum-Free Culture Systems for Mesenchymal Stem Cells: Toward Clinical-Grade Expansion
DOI:
https://doi.org/10.48048/tis.2026.11874Keywords:
Mesenchymal stem cells, Serum-free media, Xeno-free, Clinical-grade MSCs, GMP production, MSC-derived exosomes, Stem cell therapyAbstract
Mesenchymal stem cells (MSCs) are advancing rapidly towards clinical translation based on their immunomodulatory and regenerative potential. However, fetal bovine serum (FBS) dependence is still compromising safety, reproducibility, and regulatory clearance. To overcome this, serum-free and xeno-free platforms such as human platelet lysate, recombinant proteins, and chemically defined media (CDM) are strong contenders as alternatives. These media are a step up in safety, minimizing variability, and in compliance with good manufacturing practice (GMP); there are still some issues though. Costliness of recombinant supplements, donor variation in human-source additives, and lack of standardized potency assays are roadblocks in large-scale adoption. New advancements in bioreactors, omics-directed quality control, and artificial intelligence–regulated optimization hold promise for crossing roadblocks. This review is a compilation of state-of-the-art developments, critical analysis of translational roadblocks ahead, and a blueprint for making MSC expansion in serum-free settings scalable, economical, and clinically compliant.
HIGHLIGHTS
- Provides a comprehensive review of serum-free, xeno-free, and chemically defined media for mesenchymal stem cell expansion, with emphasis on translational and regulatory challenges.
- Summarizes the advantages and limitations of human-derived supplements, recombinant proteins, and synthetic components, highlighting their roles in replacing fetal bovine serum.
- Discusses commercial serum-free/xeno-free products, comparing cost-effectiveness, GMP compliance, and clinical applicability.
GRAPHICAL ABSTRACT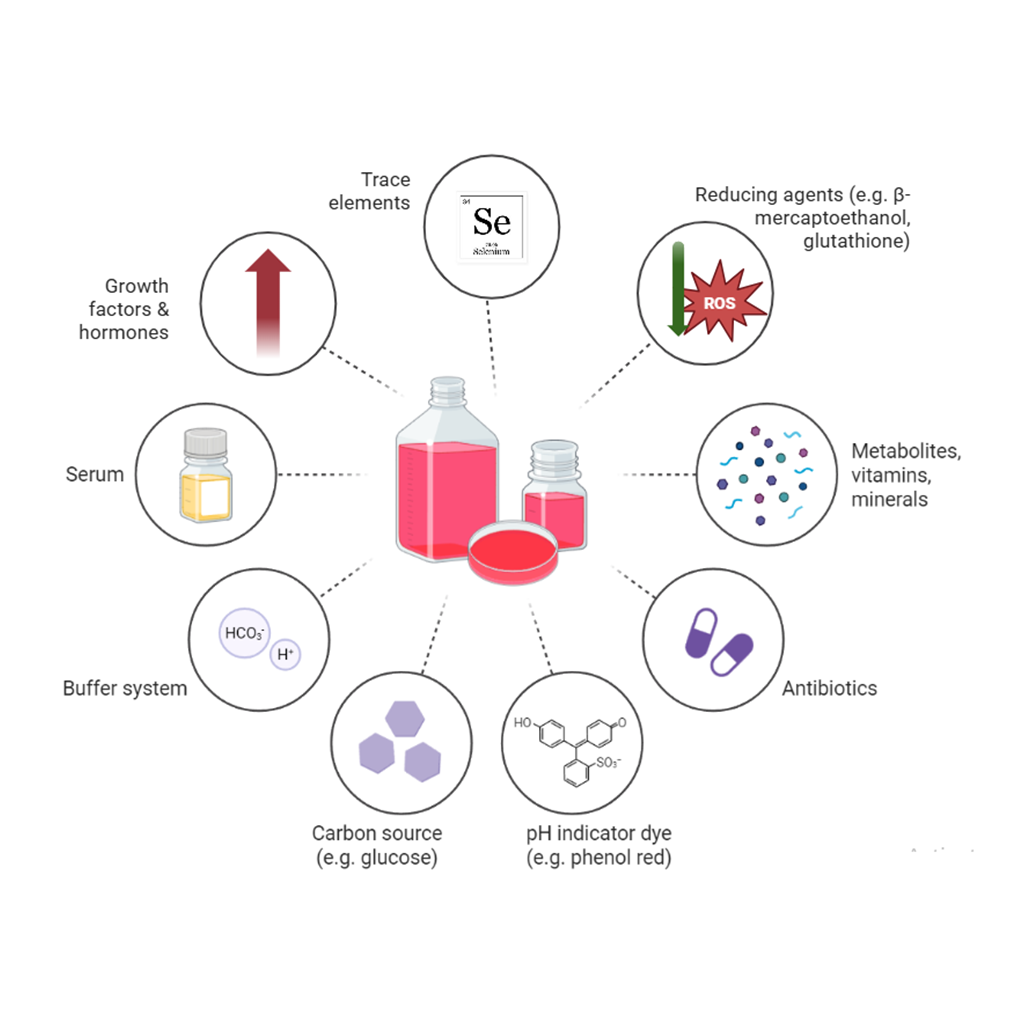
Downloads
References
OK Ma and KH Chan. Immunomodulation by mesenchymal stem cells: Interplay between mesenchymal stem cells and regulatory lymphocytes. World Journal of Stem Cells 2016; 8(9), 268-278.
X Chen, MA Armstrong and G Li. Mesenchymal stem cells in immunoregulation. Immunology & Cell Biology 2006; 84(5), 413-21.
W Jiang and J Xu. Immune modulation by mesenchymal stem cells. Cell Proliferation 2020; 53(1), e12712.
Q Zhang, J Yu, Q Chen, H Yan, H Du and W Luo. Regulation of pathophysiological and tissue regenerative functions of MSCs mediated via the WNT signaling pathway (Review). Molecular Medicine Reports 2021; 24(3), 648.
NC Truong, TN Phan, NT Huynh, KD Pham and PV Pham. Interferon-gamma increases the immune modulation of umbilical cord-derived mesenchymal stem cells but decreases their chondrogenic potential. In: PV Pham (Ed.). Advances in mesenchymal stem cells and tissue engineering. ICRRM 2023. Advances in experimental medicine and biology. Springer, Cham, Switzerland, 2023, p. 19-33.
K Nilsson. Mammalian cell culture. Methods in Enzymology 1987; 135, 387-393.
J van der Valk. Fetal bovine serum-a cell culture dilemma. Science 2022; 375(6577), 143-144.
KS Chelladurai, JDS Christyraj, K Rajagopalan, BV Yesudhason, S Venkatachalam, M Mohan, NC Vasantha and JRSS Christyraj. Alternative to FBS in animal cell culture - An overview and future perspective. Heliyon 2021; 7(8), e07686.
T Weber and J Wiest. Case studies exemplifying the transition to animal component-free cell culture. Alternatives to Laboratory Animals 2022; 50(5), 330-338.
M Zhang, X Zhao, Y Li, Q Ye, Y Wu, Q Niu, Y Zhang, G Fan, T Chen, J Xia and Q Wu. Advances in serum-free media for CHO cells: From traditional serum substitutes to microbial-derived substances. Biotechnology Journal 2024; 19(6), e2400251.
K Zhang and K Cheng. Stem cell-derived exosome versus stem cell therapy. Nature Reviews Bioengineering 2023; 1(9), 608-609.
A Nuschke, M Rodrigues, AW Wells, K Sylakowski and A Wells. Mesenchymal stem cells/multipotent stromal cells (MSCs) are glycolytic and thus glucose is a limiting factor of in vitro models of MSC starvation. Stem Cell Research and Therapy 2016; 7(1), 179.
S Tan, W Wu, Y Chen and H Gao. High glucose induces senescence in synovial mesenchymal stem cells through mitochondrial dysfunction. BMC Oral Health 2025; 25(1), 569.
D Zhang, H Lu, Z Chen, Y Wang, J Lin, S Xu, C Zhang, B Wang, Z Yuan, X Feng, X Jiang and J Pan. High glucose induces the aging of mesenchymal stem cells via Akt/mTOR signaling. Molecular Medicine Reports 2017; 16(2), 1685-1690.
T Lo, JH Ho, MH Yang and OK Lee. Glucose reduction prevents replicative senescence and increases mitochondrial respiration in human mesenchymal stem cells. Cell Transplantation 2011; 20(6), 813-825.
GA Higuera, D Schop, TW Spitters, R van Dijkhuizen-Radersma, M Bracke, JD de Bruijn, D Martens, M Karperien, A van Boxtel and CA van Blitterswijk. Patterns of amino acid metabolism by proliferating human mesenchymal stem cells. Tissue Engineering Part A 2012; 18(5-6), 654-664.
T Sartori, ACA Santos, R Oliveira da Silva, G Kodja, MM Rogero, P Borelli and RA Fock. Branched chain amino acids improve mesenchymal stem cell proliferation, reducing nuclear factor kappa B expression and modulating some inflammatory properties. Nutrition 2020; 78, 110935.
M Clémot, RS Demarco and DL Jones. Lipid mediated regulation of adult stem cell behavior. Frontiers in Cell and Developmental Biology 2020; 8, 115.
I Nikolits, S Nebel, D Egger, S Kreß and C Kasper. Towards physiologic culture approaches to improve standard cultivation of mesenchymal stem cells. Cells 2021; 10(4), 886.
J Massagué. TGFβ signalling in context. Nature Reviews Molecular Cell Biology 2012; 13(10), 616-630.
L Yang, Y Pang and HL Moses. TGF-beta and immune cells: an important regulatory axis in the tumor microenvironment and progression. Trends in Immunology 2010; 31(6), 220-227.
F Ng, S Boucher, S Koh, KS Sastry, L Chase, U Lakshmipathy, C Choong, Z Yang, MC Vemuri, MS Rao and V Tanavde. PDGF, TGF-beta, and FGF signaling is important for differentiation and growth of mesenchymal stem cells (MSCs): Transcriptional profiling can identify markers and signaling pathways important in differentiation of MSCs into adipogenic, chondrogenic, and osteogenic lineages. Blood 2008; 112(2), 295-307.
BH Shan and FG Wu. Hydrogel-based growth factor delivery platforms: Strategies and recent advances. Advanced Materials 2024; 36(5), e2210707.
DE Discher, DJ Mooney and PW Zandstra. Growth factors, matrices, and forces combine and control stem cells. Science 2009; 324(5935), 1673-1677.
F Gattazzo, A Urciuolo and P Bonaldo. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochimica et Biophysica Acta 2014; 1840(8), 2506-2519.
P Singh and JE Schwarzbauer. Fibronectin and stem cell differentiation - lessons from chondrogenesis. Journal of Cell Science 2012; 125(16), 3703-3712.
J Veevers-Lowe, SG Ball, A Shuttleworth and CM Kielty. Mesenchymal stem cell migration is regulated by fibronectin through α5β1-integrin-mediated activation of PDGFR-β and potentiation of growth factor signals. Journal of Cell Science 2011; 124(8), 1288-1300.
AA El-Rashidy and S El Moshy. Effect of polymeric matrix stiffness on osteogenic differentiation of mesenchymal stem/progenitor cells: Concise review. Polymers 2021; 13(17), 2950.
S Dupont, L Morsut, M Aragona, E Enzo, S Giulitti, M Cordenonsi, F Zanconato, J Le Digabel, M Forcato, S Bicciato, N Elvassore and S Piccolo. Role of YAP/TAZ in mechanotransduction. Nature 2011; 474(7350), 179-183.
A Mohyeldin, T Garzón-Muvdi and A Quiñones-Hinojosa. Oxygen in stem cell biology: A critical component of the stem cell niche. Cell Stem Cell 2010; 7(2), 150-161.
M Schieber and NS Chandel. ROS function in redox signaling and oxidative stress. Current Biology 2014; 24(10), R453-R462.
V Estrella, T Chen, M Lloyd, J Wojtkowiak, HH Cornnell, A Ibrahim-Hashim, K Bailey, Y Balagurunathan, JM Rothberg, BF Sloane, J Johnson, RA Gatenby and RJ Gillies. Acidity generated by the tumor microenvironment drives local invasion. Cancer Reserach 2013; 73(5), 1524-1535.
G Gstraunthaler, T Lindl and Jvd Valk. A plea to reduce or replace fetal bovine serum in cell culture media. Cytotechnology 2013; 65, 791-793.
M Cassotta, JJ Bartnicka, F Pistollato, S Parvatam, T Weber, V D’Alessandro, LF Bastos and S Coecke. A worldwide survey on the use of animal-derived materials and reagents in scientific experimentation. Engineering in Life Sciences 2022; 22(9), 564-583.
G Gstraunthaler. Alternatives to the use of fetal bovine serum: Serum-free cell culture. Altex 2003; 20(4), 275-281.
AJ Stout, AB Mirliani, ML Rittenberg, M Shub, EC White, JSK Yuen and DL Kaplan. Simple and effective serum-free medium for sustained expansion of bovine satellite cells for cell cultured meat. Communications Biology 2022; 5(1), 466.
S Gorfien, R Fike, G Godwin, J Dzimian, DA Epstein D Gruber and P Price. 2014, Serum-free mammalian cell culture medium, and uses thereof, U. S. Patent 8,455,246.
HTH Bui, LT Nguyen and UTT Than. Influences of xeno-free media on mesenchymal stem cell expansion for clinical application. Tissue Engineering and Regenerative Medicine 2021; 18(1), 15-23.
A Trounson and C McDonald. Stem cell therapies in clinical trials: Progress and challenges. Cell Stem Cell 2015; 17(1), 11-22.
D Brunner, J Frank, H Appl, H Schöffl, W Pfaller and G Gstraunthaler. Serum-free cell culture: The serum-free media interactive online database. Altex 2010; 27(1), 53-62.
X Wu, Z Ma, Y Yang, Y Mu and D Wu. Umbilical cord mesenchymal stromal cells in serum-free defined medium display an improved safety profile. Stem Cell Research & Therapy 2023; 14(1), 360.
L da Fonseca, GS Santos, SC Huber, TM Setti, T Setti and JF Lana. Human platelet lysate - A potent (and overlooked) orthobiologic. Journal of Clinical Orthopaedics and Trauma 2021; 21, 101534.
L Delila, YW Wu, O Nebie, R Widyaningrum, ML Chou, D Devos and T Burnouf. Extensive characterization of the composition and functional activities of five preparations of human platelet lysates for dedicated clinical uses. Platelets 2021; 32(2), 259-272.
T Burnouf, D Strunk, MB Koh and K Schallmoser. Human platelet lysate: Replacing fetal bovine serum as a gold standard for human cell propagation? Biomaterials 2016; 76, 371-387.
RM Rodrigues, VS Valim, M Berger, APM da Silva, FNS Fachel, Wilke, II, WOB da Silva, L Santi, MAL da Silva, B Amorin, F Sehn, JR Yates, JA Guimarães and L Silla. The proteomic and particle composition of human platelet lysate for cell therapy products. The Journal of Cellular Biochemistry 2022; 123(9), 1495-1505.
N Fekete, M Gadelorge, D Fürst, C Maurer, J Dausend, S Fleury-Cappellesso, V Mailänder, R Lotfi, A Ignatius, L Sensebé, P Bourin, H Schrezenmeier and MT Rojewski. Platelet lysate from whole blood-derived pooled platelet concentrates and apheresis-derived platelet concentrates for the isolation and expansion of human bone marrow mesenchymal stromal cells: production process, content and identification of active components. Cytotherapy 2012; 14(5), 540-554.
S Palombella, C Perucca Orfei, G Castellini, S Gianola, S Lopa, M Mastrogiacomo, M Moretti and L de Girolamo. Systematic review and meta-analysis on the use of human platelet lysate for mesenchymal stem cell cultures: Comparison with fetal bovine serum and considerations on the production protocol. Stem Cell Research & Therapy 2022; 13(1), 142.
AW Flint, ZK McQuilten, G Irwin, K Rushford, HE Haysom and EM Wood. Is platelet expiring out of date? A systematic review. Transfusion Medicine Reviews 2020; 34(1), 42-50.
K Bieback, B Fernandez-Muñoz, S Pati and R Schäfer. Gaps in the knowledge of human platelet lysate as a cell culture supplement for cell therapy: a joint publication from the AABB and the International Society for Cell & Gene Therapy. Cytotherapy 2019; 21(9), 911-924.
NTN Le and CL Han. Proteomics of human platelet lysates and insight from animal studies on platelet protein diffusion to hippocampus upon intranasal administration. APL Bioengineering 2024; 8(2), 026111.
W Delabie, G Boretti, SA Groot, D Ardanary, O Sigurjónsson, TRL Klei, P Vandekerckhove and HB Feys. Human platelet lysate standardization across three independent European blood establishments. Stem Cell Research & Therapy 2025; 16(1), 329.
I Nikolits and S Nebel. Towards physiologic culture approaches to improve standard cultivation of mesenchymal stem cells. Cells 2021; 10(4), 886.
W Delabie, D De Bleser, V Vandewalle, P Vandekerckhove, V Compernolle and HB Feys. Single step method for high yield human platelet lysate production. Transfusion 2023; 63(2), 373-383.
AR Caseiro, G Ivanova, SS Pedrosa and MV Branquinho. Human umbilical cord blood plasma as an alternative to animal sera for mesenchymal stromal cells in vitro expansion - A multicomponent metabolomic analysis. Plos One 2018; 13(10), e0203936.
TH Tong, XH Do, TT Nguyen, BH Pham, QD Le, XH Nguyen, NTM Hoang, TH Nguyen, NH Nguyen and UTT Than. Umbilical cord blood-derived platelet-rich plasma as a coating substrate supporting cell adhesion and biological activities of wound healing. European Journal of Medical Research 2025; 30(1), 145.
Y Ding, H Yang, JB Feng, Y Qiu, DS Li and Y Zeng. Human umbilical cord-derived MSC culture: the replacement of animal sera with human cord blood plasma. In Vitro Cellular & Developmental Biology 2013; 49(10), 771-777.
N Stute, K Holtz, M Bubenheim, C Lange, F Blake and AR Zander. Autologous serum for isolation and expansion of human mesenchymal stem cells for clinical use. Experimental Hematology 2004; 32(12), 1212-1225.
S Thaweesapphithak, C Tantrawatpan, P Kheolamai, D Tantikanlayaporn, S Roytrakul and S Manochantr. Human serum enhances the proliferative capacity and immunomodulatory property of MSCs derived from human placenta and umbilical cord. Stem Cell Research & Therapy 2019; 10(1), 79.
Y Ma, N Kakudo, N Morimoto, F Lai, S Taketani and K Kusumoto. Fibroblast growth factor-2 stimulates proliferation of human adipose-derived stem cells via Src activation. Stem Cell Research & Therapy 2019; 10(1), 350.
J Wang, J You, D Gong, Y Xu, B Yang and C Jiang. PDGF-BB induces conversion, proliferation, migration, and collagen synthesis of oral mucosal fibroblasts through PDGFR-β/PI3K/ AKT signaling pathway. Cancer Biomarkers 2021; 30(4), 407-415.
H Werner. The IGF1 signaling pathway: From basic concepts to therapeutic opportunities. International Journal of Molecular Sciences 2023; 24(19), 14882.
K Lott, P Collier, M Ringor and KM Howard. Administration of Epidermal Growth Factor (EGF) and Basic Fibroblast Growth Factor (bFGF) to induce neural differentiation of Dental Pulp Stem Cells (DPSC) isolates. Biomedicines 2023; 11(2), 255.
S Lee, J Lim, J Lee, H Ju, J Heo, Y Kim, S Kim, HY Yu, CM Ryu, SY Lee, JM Han, YM Oh, H Lee, H Jang, TJ Yoon, HS Ahn, K Kim, HR Kim, J Roe, HM Chung, J Son, JS Kim and DM Shin. Ascorbic acid 2-glucoside stably promotes the primitiveness of embryonic and mesenchymal stem cells through TET- and CREB1-dependent mechanisms. Antioxidants & Redox Signaling 2019; 32(1), 35-59.
D Yan, B Tang, L Yan, L Zhang, M Miao, X Chen, G Sui, Q Zhang, D Liu and H Wang. Sodium selenite improves the therapeutic effect of BMSCs via promoting the proliferation and differentiation, thereby promoting the hematopoietic factors. OncoTargets and Therapy 2019; 12, 9685-9696.
B Işıldar and S Özkan. Preconditioning of human umbilical cord mesenchymal stem cells with a histone deacetylase inhibitor: Valproic acid. Balkan Medical Journal 2024; 41(5), 369-376.
G Jothimani, R Di Liddo, S Pathak, M Piccione, S Sriramulu and A Banerjee. Wnt signaling regulates the proliferation potential and lineage commitment of human umbilical cord derived mesenchymal stem cells. Molecular Biology Reports 2019; 47, 1293-1308.
YY Jia, JY Zhou, Y Chang, F An, XW Li, XY Xu, XL Sun, CY Xiong and JL Wang. Effect of optimized concentrations of basic fibroblast growth factor and epidermal growth factor on proliferation of fibroblasts and expression of collagen: Related to pelvic floor tissue regeneration. Chinese Medical Journal 2018; 131(17), 2089-2096.
PJ Hanley, Z Mei, M da Graca Cabreira-Hansen, M Klis, W Li, Y Zhao, AG Durett, X Zheng, Y Wang, AP Gee and EM Horwitz. Manufacturing mesenchymal stromal cells for phase I clinical trials. Cytotherapy 2013; 15(4), 416-422.
X Liu, T Zhang, R Wang, P Shi, B Pan and X Pang. Insulin-transferrin-selenium as a novel serum-free media supplement for the culture of human amnion mesenchymal stem cells. Annals of Clinical & Laboratory Science 2019; 49(1), 63-71.
L Solmesky, S Lefler, J Jacob-Hirsch, S Bulvik, G Rechavi and M Weil. Serum free cultured bone marrow mesenchymal stem cells as a platform to characterize the effects of specific molecules. Plos One 2010; 5(9), e12689.
S Jung, A Sen, L Rosenberg and LA Behie. Human mesenchymal stem cell culture: Rapid and efficient isolation and expansion in a defined serum-free medium. Journal of Tissue Engineering and Regenerative Medicine 2012; 6(5), 391-403.
S Toyoda, J Shin, A Fukuhara, M Otsuki and I Shimomura. Transforming growth factor β1 signaling links extracellular matrix remodeling to intracellular lipogenesis upon physiological feeding events. The Journal of Biological Chemistry 2022; 298(4), 101748.
X Wang, AM Bove, G Simone and B Ma. Molecular bases of VEGFR-2-mediated physiological function and pathological role. Frontiers in Cell and Developmental Biology 2020; 8, 599281.
Yn Hu, H Chen, M Yang, J Xu, J Liu, Q He, X Xu, Z Ji, Y Yang, M Yan and H Zhang. HGF facilitates the repair of spinal cord injuries by driving the chemotactic migration of MSCs through the β-catenin/TCF4/Nedd9 signaling pathway. Stem Cells 2024; 42(11), 957-975.
A Vilar, M Hodgson-Garms, GD Kusuma, I Donderwinkel, JR Carthew, JL Tan, R Lim and JE Frith. Substrate mechanical properties bias MSC paracrine activity and therapeutic potential. Acta Biomaterialia 2023; 168, 144-158.
L Saldaña, F Bensiamar, G Vallés, FJ Mancebo, E García-Rey and N Vilaboa. Immunoregulatory potential of mesenchymal stem cells following activation by macrophage-derived soluble factors. Stem Cell Research and Therapy 2019; 10(1), 58.
K Hezam, C Wang, E Fu, M Zhou, Y Liu, H Wang, L Zhu, Z Han, ZC Han, Y Chang and Z Li. Superior protective effects of PGE2 priming mesenchymal stem cells against LPS-induced acute lung injury (ALI) through macrophage immunomodulation. Stem Cell Research & Therapy 2023; 14(1), 48.
R Arredondo, F Poggioli, S Martínez-Díaz, M Piera-Trilla, R Torres-Claramunt, L Tío and JC Monllau. Fibronectin-coating enhances attachment and proliferation of mesenchymal stem cells on a polyurethane meniscal scaffold. Regenerative Therapy 2021; 18, 480-486.
Z Tian, CK Wang, Fl Lin, Q Liu, T Wang, TC Sung, AA Alarfaj, AH Hirad, HHC Lee, GJ Wu and A Higuchi. Effect of extracellular matrix proteins on the differentiation of human pluripotent stem cells into mesenchymal stem cells. Journal of Materials Chemistry B 2022; 10(30), 5723-5732.
V Hüfner, R Lotfi, S Körper, H Schrezenmeier and MT Rojewski. Coating strategies for expansion of mesenchymal stromal cells in a hollow-fibre bioreactor. Cytotherapy 2020; 22(5), S102-S103.
S Mehrzad, S Hosseini, M Momeni-Moghaddam, M Farshchian, H Hassanzadeh, M Mirahmadi, F Sadeghifar and HR Bidkhori. Vitamin E pretreatment of mesenchymal stem cells: The Interplay of oxidative stress and inflammation. Journal of Cell and Molecular Research. 2020; 11, 99-107.
ALC Ong, SH Lee, S-W Aung, SL Khaing and TS Ramasamy. 5-Azacytidine pretreatment confers transient upregulation of proliferation and stemness in human mesenchymal stem cells. Cells & Development 2021; 165, 203659.
AJ Sheppard, K Delgado, AM Barfield, Q Xu, PA Massey and Y Dong. Rapamycin Inhibits senescence and improves immunomodulatory function of mesenchymal stem cells through IL-8 and TGF-β signaling. Stem Cell Reviews and Reports 2024; 20(3), 816-826.
T Yu, Y Cui, S Xin, Y Fu, Y Ding, L Hao and H Nie. Mesenchymal stem cell conditioned medium alleviates acute lung injury through KGF-mediated regulation of epithelial sodium channels. Biomedicine & Pharmacotherapy Biomedecine & Pharmacotherapie 2023; 169, 115896.
H Huang, W Zhang, J Su, B Zhou and Q Han. Spermidine retarded the senescence of multipotent mesenchymal stromal cells in vitro and in vivo through SIRT3-mediated antioxidation. Stem Cells International 2023; 2023, 9672658.
P Wang, P Zhu, C Yu and J Wu. The proliferation and stemness of peripheral blood-derived mesenchymal stromal cells were enhanced by hypoxia. Front Endocrinol 2022; 13, 873662.
E Kim, BD Riehl, T Bouzid, R Yang, B Duan, HJ Donahue and JY Lim. YAP mechanotransduction under cyclic mechanical stretch loading for mesenchymal stem cell osteogenesis is regulated by ROCK. Frontiers in Bioengineering and Biotechnology 2023; 11, 1306002.
L Leppik, MB Bhavsar, KMC Oliveira, MJ Eischen-Loges, S Mobini and JH Barker. Construction and use of an electrical stimulation chamber for enhancing osteogenic differentiation in mesenchymal stem/stromal cells in vitro. Journal of Visualized Experiments 2019; 143, e59127.
X Li, M Zhang, L Bai, W Bai, W Xu and H Zhu. Electromagnetic Biology and Medicine. 2012; 31356 - 364.
JG Carmelo, A Fernandes-Platzgummer, MM Diogo, CL da Silva and JMS Cabral. A xeno-free microcarrier-based stirred culture system for the scalable expansion of human mesenchymal stem/stromal cells isolated from bone marrow and adipose tissue. Biotechnology Journal 2015; 10(8), 1235-1247.
Thermo Fisher Scientific. Retrieved august 3. Thermo Fisher Scientific, Waltham, 2025.
LT Nguyen, NT Tran, UTT Than, MQ Nguyen, AM Tran, PTX Do, TT Chu, TD Nguyen, AV Bui, TA Ngo, VT Hoang and NTM Hoang. Optimization of human umbilical cord blood-derived mesenchymal stem cell isolation and culture methods in serum- and xeno-free conditions. Stem Cell Research & Therapy 2022; 13(1), 15.
G Chen, A Yue, Z Ruan, Y Yin, R Wang, Y Ren and L Zhu. Human umbilical cord-derived mesenchymal stem cells do not undergo malignant transformation during long-term culturing in serum-free medium. Plos One 2014; 9(6), e98565.
M Mehrian, T Lambrechts, M Marechal, FP Luyten, I Papantoniou and L Geris. Predicting in vitro human mesenchymal stromal cell expansion based on individual donor characteristics using machine learning. Cytotherapy 2020; 22(2), 82-90.
T Yang, G Chen, S Xue, M Qiao, Hw Liu, H Tian, S Qiao, F Chen, Z Chen, AN Sun and DP Wu. Comparison of the biological characteristics of serum-free and fetal bovine serum-contained medium cultured umbilical cord-derived mesenchymal stem cells. Zhonghua Xueyexue Zazhi 2012; 33(9), 715-719.
LG Chase, U Lakshmipathy, LA Solchaga, MS Rao and MC Vemuri. A novel serum-free medium for the expansion of human mesenchymal stem cells. Stem Cell Research & Therapy 2010; 1(1), 8.
C Aussel, E Busson, H Vantomme, J Peltzer and C Martinaud. Quality assessment of a serum and xenofree medium for the expansion of human GMP-grade mesenchymal stromal cells. PeerJ 2022; 10, e13391.
A Walewska, A Janucik, M Tynecka, M Moniuszko and A Eljaszewicz. Mesenchymal stem cells under epigenetic control - the role of epigenetic machinery in fate decision and functional properties. Cell Death & Disease 2023; 14(11), 720.
A Mizukami, A Fernandes-Platzgummer, JG Carmelo, K Swiech, DT Covas, JM Cabral and CL da Silva. Stirred tank bioreactor culture combined with serum-/xenogeneic-free culture medium enables an efficient expansion of umbilical cord-derived mesenchymal stem/stromal cells. The Biotechnology Journal 2016; 11(8), 1048-1059.
Q Zou, M Wu, L Zhong, Z Fan, B Zhang, Q Chen and F Ma. Development of a Xeno-free feeder-layer system from human umbilical cord mesenchymal stem cells for prolonged expansion of human induced pluripotent stem cells in culture. Plos One 2016; 11(2), e0149023.
PJ Dolley-Sonneville, LE Romeo and ZK Melkoumian. Synthetic surface for expansion of human mesenchymal stem cells in xeno-free, chemically defined culture conditions. Plos One 2013; 8(8), e70263.
SH Al-Saqi, M Saliem, S Asikainen, HC Quezada, A Ekblad, O Hovatta, K Le Blanc, AF Jonasson and C Götherström. Defined serum-free media for in vitro expansion of adipose-derived mesenchymal stem cells. Cytotherapy 2014; 16(7), 915-926.
B Cunha, T Aguiar, SB Carvalho, MM Silva, RA Gomes, MJT Carrondo, P Gomes-Alves, C Peixoto, M Serra and PM Alves. Bioprocess integration for human mesenchymal stem cells: From up to downstream processing scale-up to cell proteome characterization. Journal of Biotechnology 2017; 248, 87-98.
S Bhat, P Viswanathan, S Chandanala, SJ Prasanna and RN Seetharam. Expansion and characterization of bone marrow derived human mesenchymal stromal cells in serum-free conditions. Scientific Reports 2021; 11(1), 3403.
A Bakopoulou, D Apatzidou, E Aggelidou, E Gousopoulou, G Leyhausen, J Volk, A Kritis, P Koidis and W Geurtsen. Isolation and prolonged expansion of oral mesenchymal stem cells under clinical-grade, GMP-compliant conditions differentially affects “stemness” properties. Stem Cell Research & Therapy 2017; 8(1), 247.
VT Hoang, QM Trinh, DTM Phuong, HTH Bui, LM Hang, NTH Ngan, NTT Anh, PY Nhi, TTH Nhung, HT Lien, TD Nguyen, LN Thanh and DM Hoang. Standardized xeno- and serum-free culture platform enables large-scale expansion of high-quality mesenchymal stem/stromal cells from perinatal and adult tissue sources. Cytotherapy 2021; 23(1), 88-99.
PTM Dam, VT Hoang, HTH Bui, LM Hang, DM Hoang, HP Nguyen, HT Lien, HTT Tran, XH Nguyen and LN Thanh. Human adipose-derived mesenchymal stromal cells exhibit high HLA-DR levels and altered cellular characteristics under a xeno-free and serum-free condition. Stem Cell Reviews and Reports 2021; 17(6), 2291-2303.
Ajinomoto. StemFit for mesenchymal stem cell. Ajinomoto Co., Inc., Tokyo, Japan, 2025.
P Mark, M Kleinsorge, R Gaebel, CA Lux, A Toelk, E Pittermann, R David, G Steinhoff and N Ma. Human mesenchymal stem cells display reduced expression of CD105 after culture in serum-free medium. Stem Cells International 2013; 2013, 698076.
Y Wang, H Wu, Z Yang, Y Chi, L Meng, A Mao, S Yan, S Hu, J Zhang, Y Zhang, W Yu, Y Ma, T Li, Y Cheng, Y Wang, S Wang, J Liu, J Han, C Li, L Liu, J Xu, ZB Han and ZC Han. Human mesenchymal stem cells possess different biological characteristics but do not change their therapeutic potential when cultured in serum free medium. Stem Cell Research & Therapy 2014; 5(6), 132.
RoosterBio Inc. RoosterNourishTM-MSC. RoosterBio, Inc, Frederick, 2022.
BA Christy, MC Herzig, IE Abaasah, TC Heard, AP Cap and JA Bynum. Refrigerated human mesenchymal stromal cells as an alternative to cryostorage for use in clinical investigation. Transfusion 2023; 63(7), 1366-1375.
JG Hodge, JL Robinson and AJ Mellott. Mesenchymal stem cell extracellular vesicles from tissue-mimetic system enhance epidermal regeneration via formation of migratory cell sheets. Tissue Engineering and Regenerative Medicine 2023; 20(6), 993-1013.
D Dah-Ching, S Woei-Cherng and L Shinn-Zong. Mesenchymal Stem Cell. Cell Transplant 2011; 20(1), 5-14.
S Bobis-Wozowicz, K Kmiotek, K Kania, E Karnas, A Labedz-Maslowska, M Sekula, S Kedracka-Krok, J Kolcz, D Boruczkowski, Z Madeja and EK Zuba-Surma. Diverse impact of xeno-free conditions on biological and regenerative properties of hUC-MSCs and their extracellular vesicles. Journal of Molecular Medicine 2017; 95(2), 205-220.
PT Kiet, NTH Trang, NT Phat, VB Ngoc and P Van Phuc. CellTravel: An injectable, defined medium for cool or ambient temperature transport and short-term storage of human mesenchymal stem cells. Biomedical Research and Therapy 2024; 11(7), 6633-6641.
K Achilleos, C Petrou, V Nicolaidou and Y Sarigiannis. Beyond efficacy: Ensuring safety in peptide therapeutics through immunogenicity assessment. Journal of Peptide Science 2025; 31(6), e70016.
AL McCarthy, YC Callaghan and NM Brien. Protein hydrolysates from agricultural crops - bioactivity and potential for functional food development. Agriculture 2013; 3(1), 112-130.
YY Ho, HK Lu, ZFS Lim, HW Lim, YS Ho and SK Ng. Applications and analysis of hydrolysates in animal cell culture. Bioresour Bioprocess 2021; 8(1), 93.
P Huynh, DH Vo, KV Nguyen and N Vu. Next-generation approaches for mesenchymal stem cell characterization and therapeutic application. Journal of Advanced Biotechnology and Experimental Therapeutics 2025; 8(3), 433.
ME Williams, F Banche Niclot, S Rota, J Lim, J Machado, R de Azevedo, K Castillo, S Adebiyi, R Sreenivasan, D Kota, PC McCulloch and F Taraballi. Optimizing mesenchymal stem cell therapy: From isolation to GMP-compliant expansion for clinical application. BMC Molecular and Cell Biology 2025; 26(1), 15.
JA Dahl, S Duggal, N Coulston, D Millar, J Melki, A Shahdadfar, JE Brinchmann and P Collas. Genetic and epigenetic instability of human bone marrow mesenchymal stem cells expanded in autologous serum or fetal bovine serum. The International Journal of Developmental Biology 2008; 52(8), 1033-1042.
Y Zhu, X Song, J Wang, Y Li, Y Yang, T Yang, H Ma, L Wang, G Zhang, WC Cho, X Liu and J Wei. Placental mesenchymal stem cells of fetal origin deposit epigenetic alterations during long-term culture under serum-free condition. Expert Opinion on Biological Therapy 2015; 15(2), 163-180.
I Mortada and R Mortada. Epigenetic changes in mesenchymal stem cells differentiation. European Journal of Medical Genetics 2018; 61(2), 114-118.
R Chinnadurai. Advanced technologies for potency assay measurement. Advances in Experimental Medicine and Biology 2023; 1420, 81-95.
SK Niazi. A critical analysis of the FDA’s omics-driven pharmacodynamic biomarkers to establish biosimilarity. Pharmaceuticals 2023; 16(11), 1556.
P Wuchter, M Vetter, R Saffrich, A Diehlmann, K Bieback, AD Ho and P Horn. Evaluation of GMP-compliant culture media for in vitro expansion of human bone marrow mesenchymal stromal cells. Experimental Hematology 2016; 44(6), 508-518.
S Jung, KM Panchalingam, RD Wuerth, L Rosenberg and LA Behie. Large‐scale production of human mesenchymal stem cells for clinical applications. Biotechnology and Applied Biochemistry 2012; 59(2), 106-120.
D Salzig, J Barekzai, F Petry, J Zitzmann and P Czermak. Bioprocess development for human mesenchymal stem cell therapy products. In: RM Martínez-Espinosa (Ed.). New advances on fermentation processes. IntechOpen, London, 2019.
D de Sousa Pinto, C Bandeiras, M de Almeida Fuzeta, CAV Rodrigues, S Jung, Y Hashimura, RJ Tseng, W Milligan, B Lee, FC Ferreira, C Lobato da Silva and JMS Cabral. Scalable manufacturing of human mesenchymal stromal cells in the vertical-wheel bioreactor system: An experimental and economic approach. Biotechnology Journal 2019; 14(8), e1800716.
AC Tsai and CA Pacak. Bioprocessing of human mesenchymal stem cells: From planar culture to microcarrier-based bioreactors. Bioengineering 2021; 8(7), 96.
K Zhu, Y Ding, Y Chen, K Su, J Zheng, Y Zhang, Y Hu, J Wei and Z Wang. Advancing regenerative medicine: The Aceman system’s pioneering automation and machine learning in mesenchymal stem cell biofabrication. Biofabrication 2025; 17(2), 025021.
IA Udugama, S Badr, K Hirono, BX Scholz, Y Hayashi, M Kino‐oka and H Sugiyama. The role of process systems engineering in applying quality by design (QbD) in mesenchymal stem cell production. Computers & Chemical Engineering 2023; 172, 108144.
V Bunpetch, H Wu, S Zhang and H Ouyang. From “bench to bedside”: Current advancement on large-scale production of mesenchymal stem cells. Stem Cells and Development 2017; 26(22), 1662-1673.
MJ Sebastião, M Serra, P Gomes-Alves and PM Alves. Stem cells characterization: OMICS reinforcing analytics. Current Opinion in Biotechnology 2021; 71, 175-181.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






