Synthesis of N,N-Dimethylcyclohexylamine-Hyptolide and Activity Assessment Against Breast Cancer Stem Cells (BCSCs) Using In Vitro and In Silico Approaches
DOI:
https://doi.org/10.48048/tis.2026.11659Keywords:
Hyptolide, Diels-Alder synthesis, Breast cancer stem cells, Cell cycle arrest, Molecular dockingAbstract
The overexpression of histone deacetylase (HDAC) proteins in breast cancer stem cells (BCSCs) significantly contributes to tumor progression, chemotherapy resistance, and impaired apoptotic signaling through the silencing of tumor suppressor genes. Targeting HDAC with small-molecule inhibitors is therefore considered a promising therapeutic strategy. In this study, a novel hyptolide-based compound, N,N-dimethylcyclohexylamine-hyptolide (compound 3), was successfully synthesized via a Diels-Alder reaction. The compound was structurally characterized using FTIR, 1H-NMR, 13C-NMR and TLC-MS, confirming the formation of a cyclohexene ring bearing a tertiary amine group. The product exhibited a melting point of 83 - 84 °C and a molecular weight of 595 g/mol. In vitro cytotoxicity assays on BCSCs revealed an IC50 value of 47.42 µg/mL, classifying it as moderately active and indicating its potential as a therapeutic candidate for BCSCs. Moreover, flow cytometry analysis demonstrated that the compound induced cell cycle arrest at both the S phase (DNA synthesis) and the G2/M phase (cell division). Complementary in silico molecular docking simulations showed stable binding interactions between the compound and key active-site residues of HDAC, supporting its potential as an HDAC inhibitor. These findings suggest that N,N-dimethylcyclohexylamine-hyptolide may serve as a promising lead for the development of HDAC-targeted therapies, particularly in the treatment of aggressive and drug-resistant breast cancers.
HIGHLIGHTS
- N,N-dimethylcyclohexylamine-hyptolide, as a derivative compound of hyptolide was successfully synthesized via Diels-Alder reaction.
- N,N-dimethylcyclohexylamine-hyptolide exhibits anticancer activity against breast cancer stem cells (BCSCs) by inducing cell cycle arrest in the S and G2/M phases.
- N,N-dimethylcyclohexylamine-hyptolide has potential activity as an HDAC inhibitor based on in silico molecular docking study.
GRAPHICAL ABSTRACT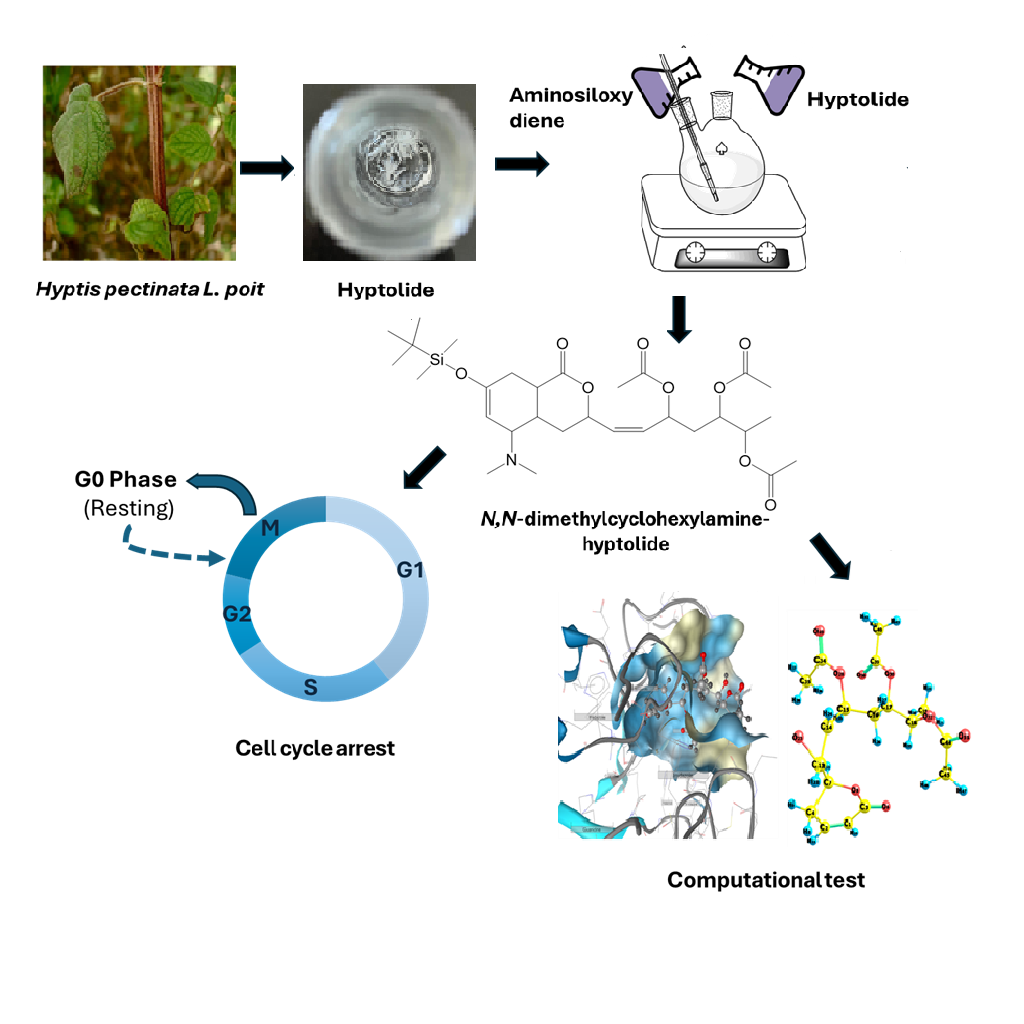
Downloads
References
N Dasari, GS Guntuku and SKSS Pindiprolu Targeting triple negative breast cancer stem cells using nanocarriers. Discover Nano 2024; 19(1), 41.
SKSS Pindiprolu, PT Krishnamurthy, PK Chintamaneni and VVSR Karri. Nanocarrier based approaches for targeting breast cancer stem cells. Artificial Cells, Nanomedicine, and Biotechnology 2017; 46(5), 885-898
RK Khangura, A Bali, AS Jaggi and N Singh. Histone acetylation and histone deacetylation in neuropathic pain: An unresolved puzzle? European Journal of Pharmacology 2017; 795, 36-42.
HR Gatla, N Muniraj, P Thevkar, S Yavvari, S Sukhavasi and MR Makena. Regulation of chemokines and cytokines by histone deacetylases and an update on histone decetylase inhibitors in human diseases. International Journal of Molecular Sciences 2019; 20, 1-27.
RI Jenie, ND Amalina, A Hermawan, M Suzery, A Putra and E Meiyanto. Caesalpinia sappan reduces the stemness of breast cancer stem cells involving the elevation of intracellular reactive oxygen species. Research in Pharmaceutical Sciences 2023; 18, 708-721.
JK Opara, S Sanchez, SM Thomas and S Anant. Natural products targeting cancer stem cells: A promising therapeutic approach. Exploration of Drug Science 2025; 3, 1008106.
M Suzery, B Cahyono and ND Amalina. Antiproliferative and apoptosis effect of hyptolide from hyptis pectinata (l.) poit on human breast cancer cells. Journal of Applied Pharmaceutical Science 2020; 10(2), 001-006.
B Cahyono, M Suzery, ND Amalina and DN Bima. Synthesis and antibacterial activity of epoxide from hyptolide (Hyptis pectinata (L.) Poit) against gram-positive and gram-negative bacteria. Journal of Applied Pharmaceutical Science 2020; 10(12), 013-022.
M Suzery, B Cahyono, ND Amalina, C Budiman, I Bayu and M Asy. Antiplasmodial activity of Hyptis pectinata extract and its analog against 3D7 Plasmodium falciparum through caseinolytic protease proteolytic (ClpP) inhibition The Thai Journal of Pharmaceutical Sciences 2021; 45, 527-531
C V Barbosa, PGV Aquino, KAL Ribeiro-Júnior, FBP Moura, MS Alexandre-Moreira, AEG Sant’Ana, JRO Ferreira, MO Moraes, C Pessoa, JS Aguiar, TG Silva and JX Araújo-Júnior. Cytotoxic and antitumor activities of Hyptis pectinata (Sambacaitá) extract. Pharmacologyonline 2012; 3, 70-74.
FR Santana, L Luna-Dulcey, VU Antunes, CF Tormena, MR Cominetti, MC Duarte and JA da Silva. Evaluation of the cytotoxicity on breast cancer cell of extracts and compounds isolated from Hyptis Pectinata (L.) poit. Natural Product Research 2020; 34, 102-109.
SA Kozmin, MT Green and VH Rawal. On the reactivity of 1-amino-3-siloxy-1 , 3-dienes: Kinetics investigation and theoretical interpretation at providing fundamental quantitative information re- garding the diels - alder kinetics of amino siloxy dienes larly interested in determining if t. The Journal of Organic Chemistry 2002; 64(21), 8045-8047.
EE Kwan and SG Huang. Structural elucidation with NMR spectroscopy: Practical strategies for organic chemists. European Journal of Organic Chemistry 2008; 2008(16), 2671-2688.
M Suzery, B Cahyono and ND Amalina. Unveiling the potential of hyptis capitata and hyptis brevipes compounds in overcoming apoptosis resistance and inducing targeted cell cycle arrest in breast cancer stem cells. Trends in Sciences 2024; 21(12), 8404.
D Hermansyah, D Munir, A Lelo, A Putra, ND Amalina and I Alif. The synergistic antitumor effects of curcuma longa and phyllanthus niruri extracts on promoting apoptotic pathways in breast cancer stem cells. The Thai Journal of Pharmaceutical Sciences 2022; 46(5), 541-550.
M Ikawati, RI Jenie, RY Utomo, ND Amalina, GPN Ilmawati, M Kawaichi and E Meiyanto. Genistein enhances cytotoxic and antimigratory activities of doxorubicin on 4T1 breast cancer cells through cell cycle arrest and ROS generation. Journal of Applied Pharmaceutical Science 2020;10(10), 95-104.
MV Sefton, H Uludag, J Babensee, T Roberts, V Horvath and U De Boni. Microencapsulation of cells in thermoplastic copolymer (hydroxyethyl methacrylate-methyl methacrylate). Methods in Neurosciences 1994; 21, 371-386.
G Bora-Tatar, D Dayangaç-Erden, AS Demir, S Dalkara, K Yelekçi and H Erdem-Yurter. Molecular modifications on carboxylic acid derivatives as potent histone deacetylase inhibitors: Activity and docking studies. Bioorganic & Medicinal Chemistry 2009; 17(14), 5219-5228.
SA Kozmin and VH Rawal. Preparation and Diels−Alder Reactivity of 1-Amino-3-siloxy-1,3-butadienes. The Journal of Organic Chemistry 1997; 62(16), 5252-5253.
SF Pedersen and AM Myers. Understanding the principle of organic chemistry a laboratory course. 10th ed. Mary Finch Acquisitions, Virginia, United States, 2011.
SA Kozmin, JM Janey and VH Rawal. 1-amino-3-siloxy-1,3-butadienes: Highly reactive dienes for the diels- alder reaction. The Journal of Organic Chemistry 1999; 64, 3039-3052.
S Harada and A Nishida. Catalytic and enantioselective diels-alder reaction of siloxydienes. Asian Journal of Organic Chemistry 2019; 8(6), 732-745.
CJ Huang and EY Li. Molecular design principles towards exo-exclusive Diels-Alder reactions. RSC Advances 2019; 9(13), 7246-7250.
A Oluwasanmi and C Hoskins. Potential use of the Diels-Alder reaction in biomedical and nanomedicine applications. International Journal of Pharmaceutics 2021; 604, 120727.
J Coates. Interpretation of infrared spectra, a practical approach. Encyclopedia of Analytical Chemistry 2000; 12, 10815-10837.
C Hao, N Sousou, D Eikel and J Henion. Thin-layer chromatography/mass spectrometry analysis of sample mixtures using a compact mass spectrometer. American Laboratory 2015; 47(4), 24-27.
A Jaggupilli and E Elkord. Significance of CD44 and CD24 as cancer stem cell markers: An enduring ambiguity. Journal of Immunology Research 2012; 2012(1), 708036.
NH Zakaria, N Saad, CA Che Abdullah and N Mohd. Esa. The antiproliferative effect of chloroform fraction of Eleutherine bulbosa (Mill.) Urb. on 2D- and 3D-human lung cancer cells (A549) model. Pharmaceuticals 2023; 16(7), 936.
C Liu, H Xing, C Guo, Z Yang, Y Wang and Y Wang. MiR-124 reversed the doxorubicin resistance of breast cancer stem cells through STAT3/HIF-1 signaling pathways. Cell Cycle 2019; 18(18), 2215-2227.
MC Bajgelman. Principles and applications of flow cytometry. In: G Misra (Ed.). Data processing handbook for complex biological data sources. Elsevier, Amsterdam, Netherlands, 2019.
LC Crowley, AP Scott, BJ Marfell, JA Boughaba, G Chojnowski and NJ Waterhouse. Measuring cell death by propidium iodide uptake and flow cytometry. Cold Spring Harbor Protocols 2016; 2016(7), 647-651.
C Park, HJ Cha, H Lee, H Hwang-Bo, SY Ji, MY Kim, SH Hong, JW Jeong, MH Han, SH Choi, CY Jin, GY Kim and YH Choi. Induction of G2/M cell cycle arrest and apoptosis by genistein in human bladder cancer T24 cells through inhibition of the ROS-dependent PI3k/Akt signal transduction pathway. Antioxidants 2019; 8(9), 327.
Z Kinart, M Hoelm and M Imińska. Evaluating theoretical solvent models for thermodynamic and structural descriptions of dacarbazine - cyclodextrin complexes. The theoretical and conductometric study. Molecules 2025; 30(11), 2309.
X Du, Y Li, YL Xia, SM Ai, J Liang, P Sang, XL Ji and SQ Liu. Insights into protein-ligand interactions: Mechanisms, models, and methods. International Journal of Molecular Sciences 2016; 17(2), 144.
D Hudiyanti, R Khairiah, P Siahaan, FAA Majid, E Fachriyah and NH Zakaria. Enhanced stability and binding efficiency of liposomal andrographolide complexes targeting human papillomavirus for cervical cancer therapy: Molecular docking and molecular dynamics simulations. Results in Chemistry 2025; 16, 102342.
VP Reddy. Fluorinated compounds in enzyme-catalyzed reactions. Organofluorine Compounds in Biology and Medicine 2015; 5, 29-57.
S Ramachandran, P Kota, F Ding and NV Dokholyan. Automated minimization of steric clashes in protein structures. Proteins: Structure, Function, and Bioinformatics 2011; 79(1), 261-270.
TJ Gardella, H Jüppner and JT Potts. Receptors for parathyroid hormone and parathyroid hormone-related protein. In: JP Bilezikian, TJ Martin, TL Clemens and CJ Rosen (Eds.). Principles of bone biology. Academic Press, New York, 2019, p. 691-712.
R Kim and J Skolnick. Assessment of programs for ligand binding affinity prediction. Journal of Computational Chemistry 2008; 29(8), 1316-1331.
A Grassadonia, P Cioffi, F Simiele, L Iezzi, M Zilli and C Natoli. Role of hydroxamate-based histone deacetylase inhibitors (Hb-HDACIs) in the treatment of solid malignancies. Cancers 2013; 5(3), 919-942.
F Leoni, A Zaliani, G Bertolini, G Porro, P Pagani, P Pozzi, G Donà, G Fossati, S Sozzani, T Azam, P Bufler, G Fantuzzi, I Goncharov, SH Kim, BJ Pomerantz, LL Reznikov, B Siegmund, CA Dinarello and P Mascagni. The antitumor histone deacetylase inhibitor suberoylanilide hydroxamic acid exhibits antiinflammatory properties via suppression of cytokines. Proceedings of the National Academy of Sciences 2002; 99(5), 2995-3000.
PM Lombardi, KE Cole, DP Dowling and DW Christianson. Structure, mechanism, and inhibition of histone deacetylases and related metalloenzymes. Current Opinion in Structural Biology 2011; 21(6), 735-743.
A Cortés, M Cascante, ML Cárdenas and A Cornish-Bowden. Relationships between inhibition constants, inhibitor concentrations for 50% inhibition and types of inhibition: New ways of analysing data. Biochemical Journal 2001; 357(1), 263-268.
EC Lima, A Hosseini-Bandegharaei, JC Moreno-Piraján and I Anastopoulos. A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption. Journal of Molecular Liquids 2019; 273, 425-434.
DJ Maxwell, BC Hicks, S Parsons and SE Sakiyama-Elbert. Development of rationally designed affinity-based drug delivery systems. Acta Biomaterialia 2005; 1(1), 101-113.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






