Cost-Effective Tissue Culture Protocols for Neglected Indigenous Dioscorea Varieties: Optimization of Disinfection, Browning Control, and Growth Enhancement
DOI:
https://doi.org/10.48048/tis.2026.11581Keywords:
Dioscorea spp, Low-cost tissue culture, Bleach disinfection, Browning control, Dioscorea spp., Low-cost tissue culture, Bleach disinfection, Browning controlAbstract
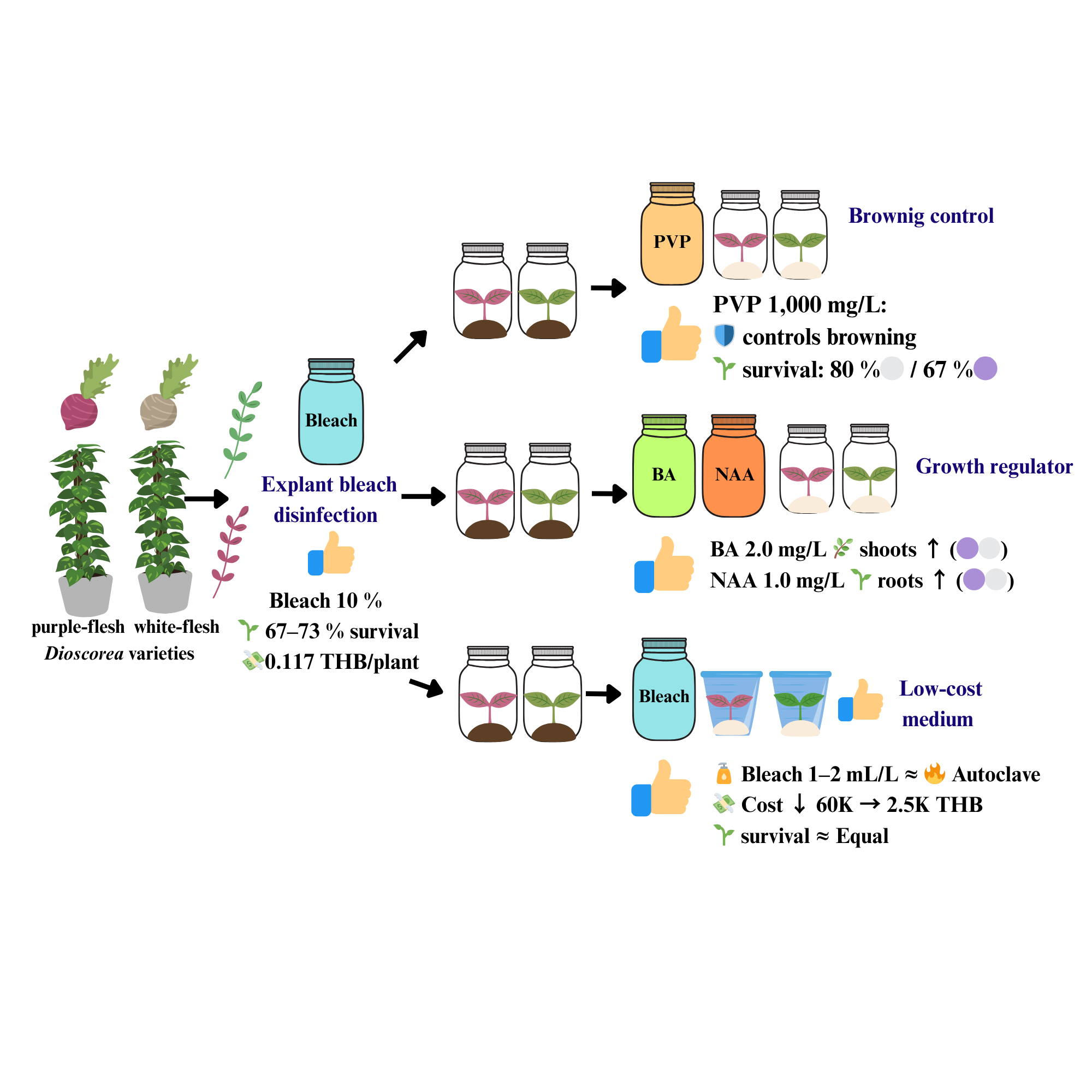
Indigenous Dioscorea species are important neglected and underused species (NUS) that represent rich genetic resources for food security and medicinal applications, offering exceptional climate resilience and nutritional diversity crucial for sustainable agriculture. However, their propagation is hampered by expensive tissue culture techniques and high equipment expenses. The study optimized disinfection techniques, browning control, and growth promotion to develop comprehensive low-cost tissue culture procedures for purple- and white-fleshed indigenous Dioscorea varieties. Nodal explants were examined with commercial bleach (5% - 20%) for surface disinfection, polyvinylpyrrolidone, PVP (0 - 1,500 mg/L) for browning management, hormone combinations (BA and NAA at 0.5 - 2.0 mg/L) for morphogenesis, and alternative medium disinfection methods (bleach 0.5 - 4.0 mL/L versus autoclaving). Optimal explant disinfection was achieved using 10% commercial bleach, resulting in 67% - 73% survival at a cost of 0.117 THB (Thai Baht) per surviving plant. PVP at 1,000 mg/L substantially suppressed browning while increasing survival to 80% in white-fleshed and 67% in purple-fleshed varieties, with white-fleshed varieties regularly outperforming, purple-fleshed variants. BA at 2.0 mg/L increased shoot growth (5.87 shoots per explant), while NAA at 1.0 mg/L increased root formation (5.89 roots per explant). Bleach-based medium disinfection at 1 - 2 mL/L achieved autoclaving performance while substantially lowering equipment investment from 60,000 to 2,500 THB. These integrated protocols establish complete low-cost tissue culture systems utilizing commercial bleach for all disinfection steps, eliminating expensive laboratory equipment requirements and enabling community-based propagation systems for indigenous Dioscorea conservation in resource-constrained environments.
HIGHLIGHTS
- 10% commercial bleach for explant disinfection achieved 67% - 73% survival with minimal contamination.
- PVP at 1,000 mg/L prevented browning with genotype-specific responses.
- BA 2.0 mg/L and NAA 1.0 mg/L optimized shoot and root development.
- Bleach-based medium disinfection reduced equipment costs from 60,000 to 2,500 THB.
- Low-cost protocol enabled Dioscorea tissue culture and conservation.
GRAPHICAL ABSTRACT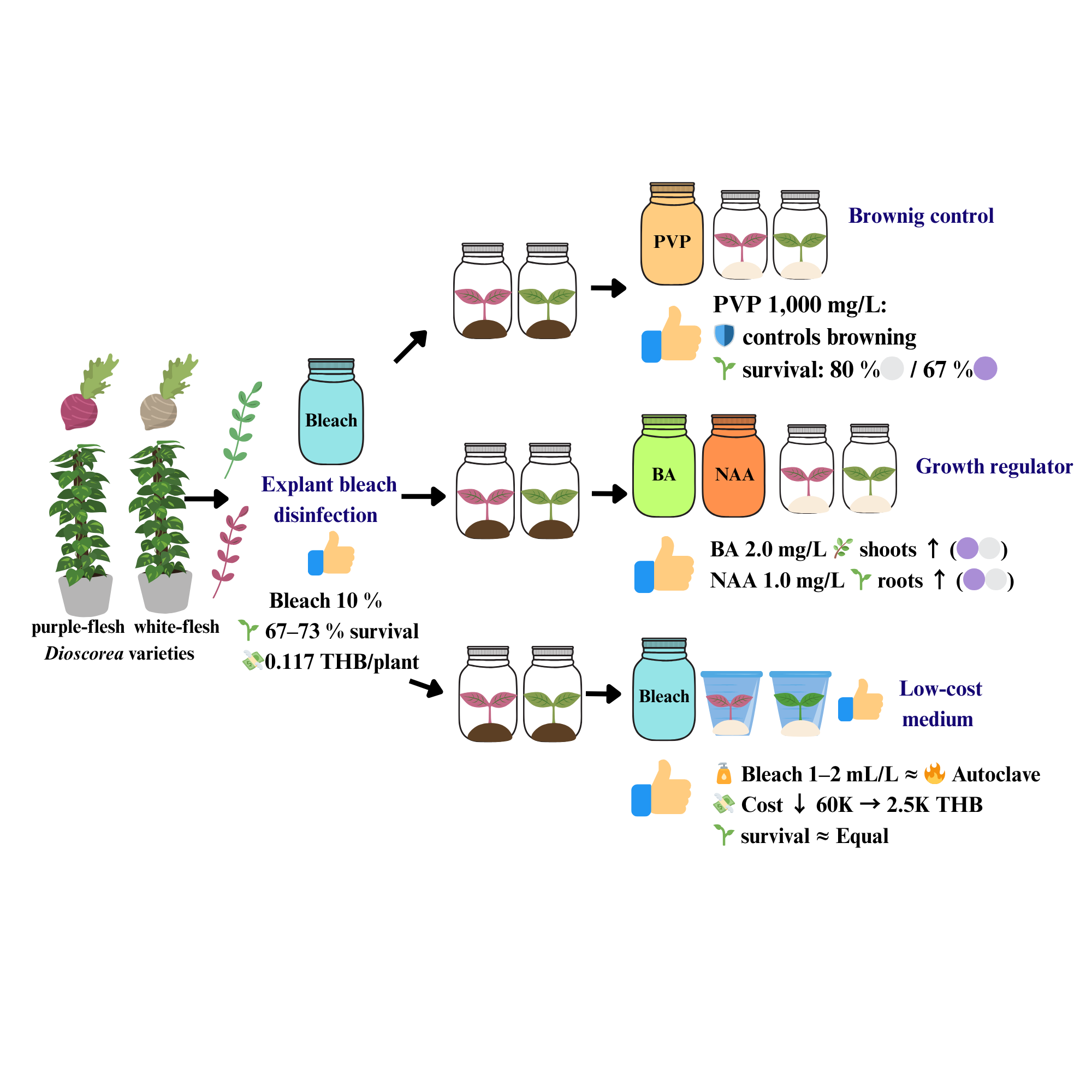
Downloads
References
S Heikonen, M Heino, M Jalava, S Siebert, D Viviroli and M Kummu. Climate change threatens crop diversity at low latitudes. Nature Food 2025; 6(4), 331-342.
AS Toromade, DA Soyombo, E Kupa and TI Ijomah. Reviewing the impact of climate change on global food security: Challenges and solutions. International Journal of Applied Research in Social Sciences 2024; 6(7), 1403-1416.
M Alotaibi. Climate change, its impact on crop production, challenges, and possible solutions. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2023; 51(1), 13020.
TB Mihrete and FB Mihretu. Crop diversification for ensuring sustainable agriculture, risk management and food security. Global Challenges 2025; 9(2), 2400267.
MSA Talucder, UB Ruba and MAS Robi. Potentiality of Neglected and Underutilized Species (NUS) as a future resilient food: A systematic review. Journal of Agriculture and Food Research 2024; 16, 101116.
AB Wani, Biaza and Irtiza. Neglected and Underutilized Crops-Towards Nutritional Security and Sustainability. Springer Nature, Singapore, 2021, p. 51-70.
L Nhamo, G Paterson, M van der Walt, M Moeletsi, A Modi, R Kunz, V Chimonyo, T Masupha, S Mpandeli, S Liphadzi, J Molwantwa and T Mabhaudhi. Optimal production areas of underutilized indigenous crops and their role under climate change: Focus on Bambara groundnut. Frontiers in Sustainable Food Systems 2022; 6, 990213.
I Proietti, I Jordan and T Borelli. Enhancing nutrition and cost efficiency in Kenyan school meals using neglected and underutilized species and linear programming: A case study from an informal settlement. Sustainability 2025; 17(6), 2436.
HE Bilali, S Rokka, G Calabrese, T Borelli, F Grazioli, SRF Tietiambou, J Nanema, ID Guimbo, L Dambo, B Nouhou, M Gonnella and F Acasto. Conservation and promotion of neglected and underutilized crop species in West Africa: Policy and governance. Sustainability 2024; 16(14), 6194.
A Adomeniene and PR Venskutonis. Dioscorea spp.: Comprehensive review of antioxidant properties and their relation to phytochemicals and health benefits. Molecules 2022; 27(8), 2530.
FN Jahan, MA Rahim, SM Bokhtiar and AK Samanta. Potentiality of underutilized crop Dioscorea spp.: A source of nutraceutical. SAARC Journal of Agriculture 2019; 17(2), 1-13.
B Padhan and D Panda. Potential of neglected and underutilized yams (Dioscorea spp.) for improving nutritional security and health benefits. Frontiers in Pharmacology 2020; 11, 496.
OB Komolafe, OH Oyinloye, ER Ogbimi, KO Hassan and OS Pelemo. Optimizing disinfection protocols for yam explant regeneration in plant tissue culture. International Journal of Horticultural Science 2025; 31, 66-72.
S Dang, R Gao, Y Zhang and Y Feng. In vitro regeneration and its histological characteristics of Dioscorea nipponica Makino. Scientific Reports 2022; 12(1), 18436.
M Niazian. Application of genetics and biotechnology for improving medicinal plants. Planta 2019; 249(4), 953-973.
PA Ebile, J Opata and S Hegele. Evaluating suitable low-cost agar substitutes, clarity, stability, and toxicity for resource-poor countriesʼ tissue culture media. In Vitro Cellular and Developmental Biology - Plant 2022; 58, 989-1001.
SAM Rodriguez, MB Garcia, EE Abeal and IP Rodriguez. Uso de distintos tratamientos de desinfección en el cultivo in vitro de Dioscorea alata L. clon caraqueño. Revista Colombiana de Biotecnología 2009; 11(2), 127-135.
JE Pardaz, S Ojagh and HD Kazemnia. Effect of polyvinylpyrrolidone (PVP) on meristem establishment and in-vitro organogenesis of Iranian pear (Pyrus glabra). International Journal of Agriculture and Biosciences 2015; 4(5), 206-208.
D dos Santos Vila Verde, MIDS Mendes, A da Silva Souza, CR Pinto, LVC Nobre, JE dos Santos Melo and CA da Silva Ledo. Ácido ascórbico e polivinilpirrolidona no cultivo in vitro de Dioscorea spp. Research, Society and Development 2021; 10(9), 10510917812.
N Permadi, SI Akbari, D Prismantoro, NN Indriyani, M Nurzaman, AN Alhasnawi, F Doni and E Julaeha. Traditional and next-generation methods for browning control in plant tissue culture: Current insights and future directions. Current Plant Biology 2024; 38, 100339.
C Chen. Cost analysis of plant micropropagation of Phalaenopsis. Plant Cell, Tissue and Organ Culture (PCTOC) 2016; 126(1), 167-175.
A Kaur and JS Sandhu. High throughput in vitro micropropagation of sugarcane (Saccharum officinarum L.) from spindle leaf roll segments: Cost analysis for agri-business industry. Plant Cell, Tissue and Organ Culture (PCTOC) 2015; 120, 339-350.
NM Gitonga, O Ombori, KSD Murithi and M Ngugi. Low technology tissue culture materials for initiation and multiplication of banana plants. African Crop Science Journal 2010; 18(4), 243-251.
AMH Ramirez, DHP Hajduk and AIU Trujillo. Cost analysis of cacao (Theobroma cacao L.) plant propagation through the somatic embryogenesis method. Revista Bionatura 2022; 7(2), 1-13.
L Chongloi, R Gunnaiah, K Hipparagi, P Guranna, DP Prakasha, R Chittapur, A Kamble and S Vishweshwar. Economic analysis of micropropagation of dragon fruit (Hylocereus undatus (Haw.) Britton and Rose). International Journal of Plant and Soil Science 2022; 34(22), 1267-1275.
SE Peiris, EDUD De Silva, M Edussuriya, AMURK Attanayake and BCN Peiris. CSUP technique: A low cost sterilization method using sodium hypochlorite to replace the use of expensive equipment in micropropagation. Journal of the National Science Foundation of Sri Lanka 2012; 40(1), 49-54.
S Sookruksawong. Sterilization and low-cost tissue culture techniques of Dioscorea bulbifera. Science, Technology and Social Sciences Procedia 2022; 2022(4), 014.
K Amare and G Dugassa. Plant tissue culture challenges in Ethiopia and alternative options for low-cost media. F1000Research 2022; 11, 828.
HD Mignouna, MM Abang, R Asiedu and R Geeta. Yam (Dioscorea) husbandry: Cultivating yams in the field or greenhouse. Cold Spring Harbor Protocols 2009; 2009(11), 5324.
Y Yoshida and K Kanahama. Effects of photoperiod and temperature on the development of spikes and new tubers in Chinese yam (Dioscorea opposita Thunb. cv. Ichoimo). Journal of the Japanese Society for Horticultural Science 1999; 68(1), 124-129.
R Homhual, M Wongmaneeroj, S Jamjumrus, S Promdany, W Tongdonae, P Donsomprai, R Agarum and S Chanprame. Propagation of Dioscorea spp. for alternative food resources. Journal of Science and Technology 2017; 6(1), 1-13.
T Murashige and F Skoog. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiologia Plantarum 1962; 15(3), 473-497.
GN Poornima and VRR Ravishankar. In vitro propagation of wild yams, Dioscorea oppositifolia (Linn) and Dioscorea pentaphylla (Linn). African Journal of Biotechnology 2007; 6(20), 2348-2352.
CR Deb and T Langhu. Efficient in vitro propagation protocol of a medicinally important plant, Dioscorea villosa L. via organogenesis from nodal segments. Plant Cell Biotechnology and Molecular Biology 2017; 18(7-8), 559-569.
S Fukuzaki. Mechanisms of actions of sodium hypochlorite in cleaning and disinfection processes. Biocontrol Science 2006; 11(4), 147-157.
GI Eliwa, EF El-Dengawy, MS Gawish and MM Yamany. Comprehensive study on in vitro propagation of some imported peach rootstocks b. in vitro explant surface sterilization and bud proliferation. Scientific Reports 2025; 15, 17905.
S Sookruksawong. Explants sterilization techniques of Dioscorea spp. for tissue culture. In: Proceedings of the 14th engineering, science, technology and architecture conference 2023. Kalasin University, Thailand, 2023, p. 680-688.
HJ Shah and SS Lele. In vitro propagation of Dioscorea alata var. purpurae. Applied Biochemistry and Biotechnology 2012; 167(6), 1811-1817.
S Shukla and SK Shukla. In vitro regeneration of Dioscorea hispida through nodal explants – a rich source of starch. GSTF Journal of BioSciences 2014; 3(1), 34-39.
O Omidiji, JE Okpuzor and O Otubu. The contribution of an ionic peroxidase isozyme to enzyme-mediated browning in Dioscorea esculenta L. tubers. Pakistan Journal of Nutrition 2006; 5(5), 478-480.
S Graham-Acquaah, GS Ayernor, B Bediako-Amoa, FK Saalia and EO Afoakwa. Spatial distribution of total phenolic content, enzymatic activities and browning in white yam (Dioscorea rotundata) tubers. Journal of Food Science and Technology 2014; 51(10), 2833-2838.
D dos Santos Vila Verde, MI de Souza Mendes, A da Silva Souza, C Rodrigues Pinto, LV Costa Nobre, KC Fialho dos Santos and CA da Silva Ledo. Culture media in the in vitro cultivation of Dioscorea spp. Concilium 2023; 23(9), 459-480.
V Amankwaah, N Ntorinkansah, G Osei, R Prempeh, H Zakpaa, D Appiah-Kubi, E Marceline and M Quain. Evaluation of media protocols for in vitro propagation of an improved variety and two landraces of Dioscorea rotundata. In vitro Cellular and Developmental Biology - Plant 2025; 61, 646-657.
MT Islam, ERJ Keller and DP Dembele. Effects of growth regulators on in vitro propagation and tuberization of four Dioscorea Species. Plant Tissue Culture and Biotechnology 2008; 18(1), 25-35.
GC Sanchez-Lopez, D Carranza-Ojeda, L Perez-Picaso, R Martinez-Pascual, O Vinas-Bravo, A Lopez-Torres, E Perez Molphe-Bach, E Garcia-Rios, JA Morales-Serna and E Villalobos-Amador. Establishment of in vitro root cultures and hairy roots of Dioscorea composita for diosgenin production. Plant Cell, Tissue and Organ Culture (PCTOC) 2025; 161, 24.
J Deubel. 2023, Optimizing shoot propagation methods in tropical yam - Dioscorea alata. Master Thesis, University of Hawaii at Mānoa, Honolulu, Hawaii.
BN Weber, RA Witherell and AO Charkowski. Low-cost potato tissue culture with microwave and bleach media preparation and sterilization. American Journal of Potato Research 2015; 92, 128-137.
NH Gavilan, FC Furlan, AZ Zorz, LS de Oliveira, WF Campos and GE Brondani. Chemical sterilization of culture medium for in vitro multiplication of Cochlospermum regium. Ciência Rural 2018; 48(9), 20170581.
SK Datta, D Chakraborty and T Janakiram. Low cost tissue culture: An overview. Journal of Plant Science Research 2017; 33(2), 181-199.
S Sookruksawong. Micropropagation of Kratom (Mitragyna speciosa (Korth.) Havil.): Disinfectants, growth regulators, and low-cost chemical disinfection for in vitro establishment. Asian Health, Science and Technology Reports 2024; 32(3), 29-44.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






