Bioactive Compounds from Trentepohlia aurea as Potential Antibacterial Agents Targeting DNA Gyrase: An In Vitro and In Silico Approach
DOI:
https://doi.org/10.48048/tis.2026.11324Keywords:
Antibacterial, DNA Gyrase, GC-MS, In Silico, Molecular Docking, Trentepohlia aurea, Antibacterial, DNA gyrase, GC-MS, In silico, Molecular docking, Trentepohlia aureaAbstract
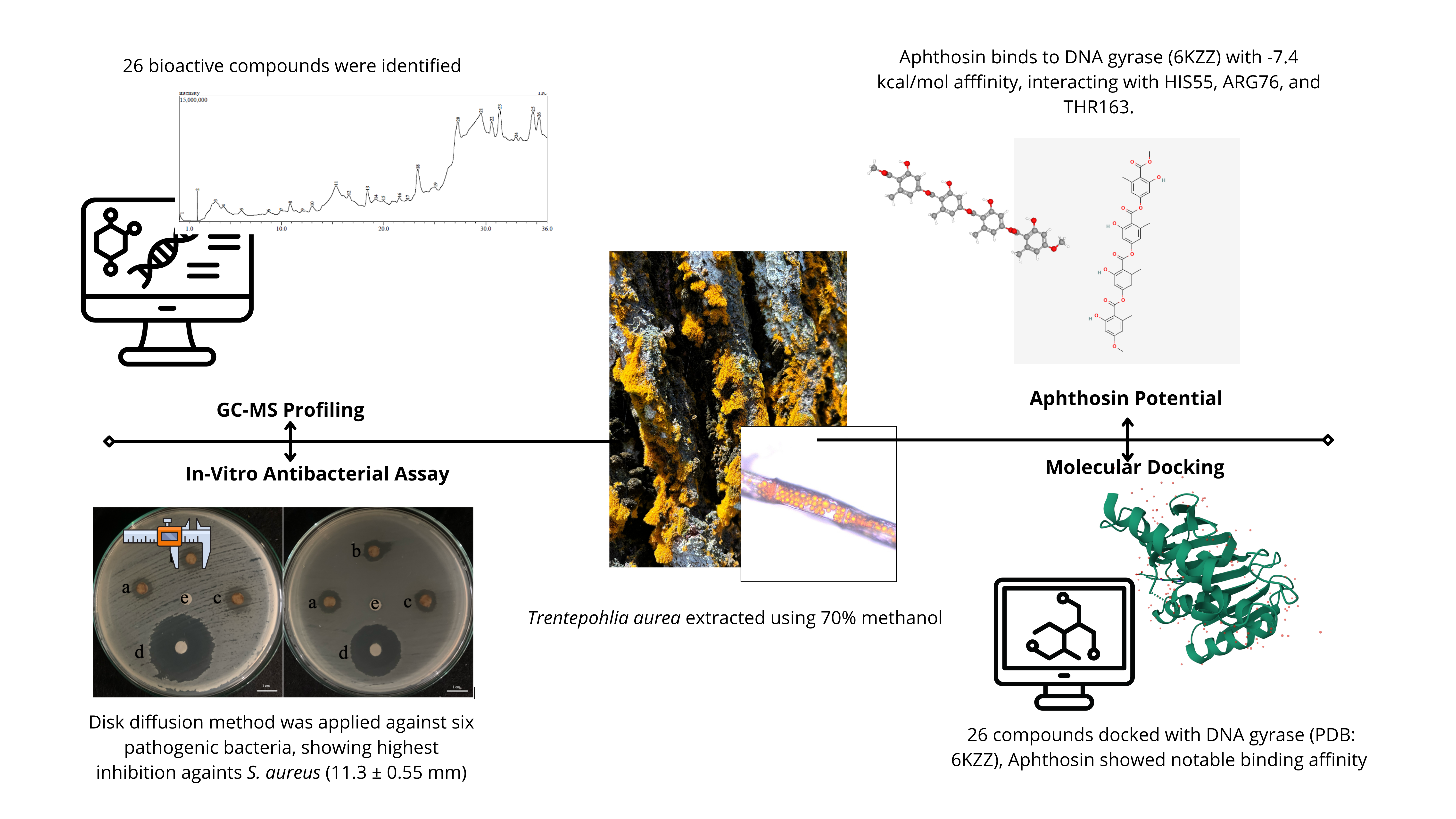
The global rise of antibiotic resistance underscores the urgent need for novel antibacterial agents with new mechanisms of action. Trentepohlia aurea, a carotenoid- and phenolic-rich subaerial green alga, remains largely unexplored for its therapeutic potential. This study evaluated the antibacterial activity of its methanol extract via in vitro disc diffusion assays and investigated the interaction of its bioactive constituents with bacterial DNA gyrase subunit B (PDB ID: 6KZZ) through in silico molecular docking. The extract produced inhibition zones of 11.3 ± 0.55 mm against Staphylococcus aureus, 10.9 ± 0.21 mm against Salmonella Typhii, and 10.8 ± 0.61 mm against Streptococcus mutans, indicating moderate antibacterial activity. GC-MS profiling identified 26 compounds, with Aphthosin exhibiting the strongest predicted gyrase-binding affinity (–7.4 kcal/mol), surpassing a native ligand. ADME-toxicity predictions suggested generally favorable pharmacokinetics with low toxicity, though some compounds displayed organ-specific risks. While MIC/MBC determinations and direct in vitro testing of individual compounds were not performed, this first report on T. aurea as a potential DNA gyrase inhibitor highlights its promise as a source of structurally diverse natural products for antibacterial drug discovery and warrants further mechanistic validation and potency optimization.
HIGHLIGHTS
This study demonstrates the antibacterial potential of Trentepohlia aurea, with its methanol extract showing notable inhibition against S. aureus and S. typhii. GC-MS analysis identified 26 bioactive compounds, with in silico docking highlighting Aphthosin as the top candidate (–7.4 kcal/mol) for binding to bacterial DNA gyrase (6KZZ). ADME and toxicity predictions indicated favourable pharmacokinetic profiles and generally low toxicity. While the findings support the potential of T. aurea as a natural antibacterial agent targeting DNA gyrase, the study is limited to GC-MS identification without isolation or purification of key compounds such as Aphthosin for further biological validation.
GRAPHICAL ABSTRACT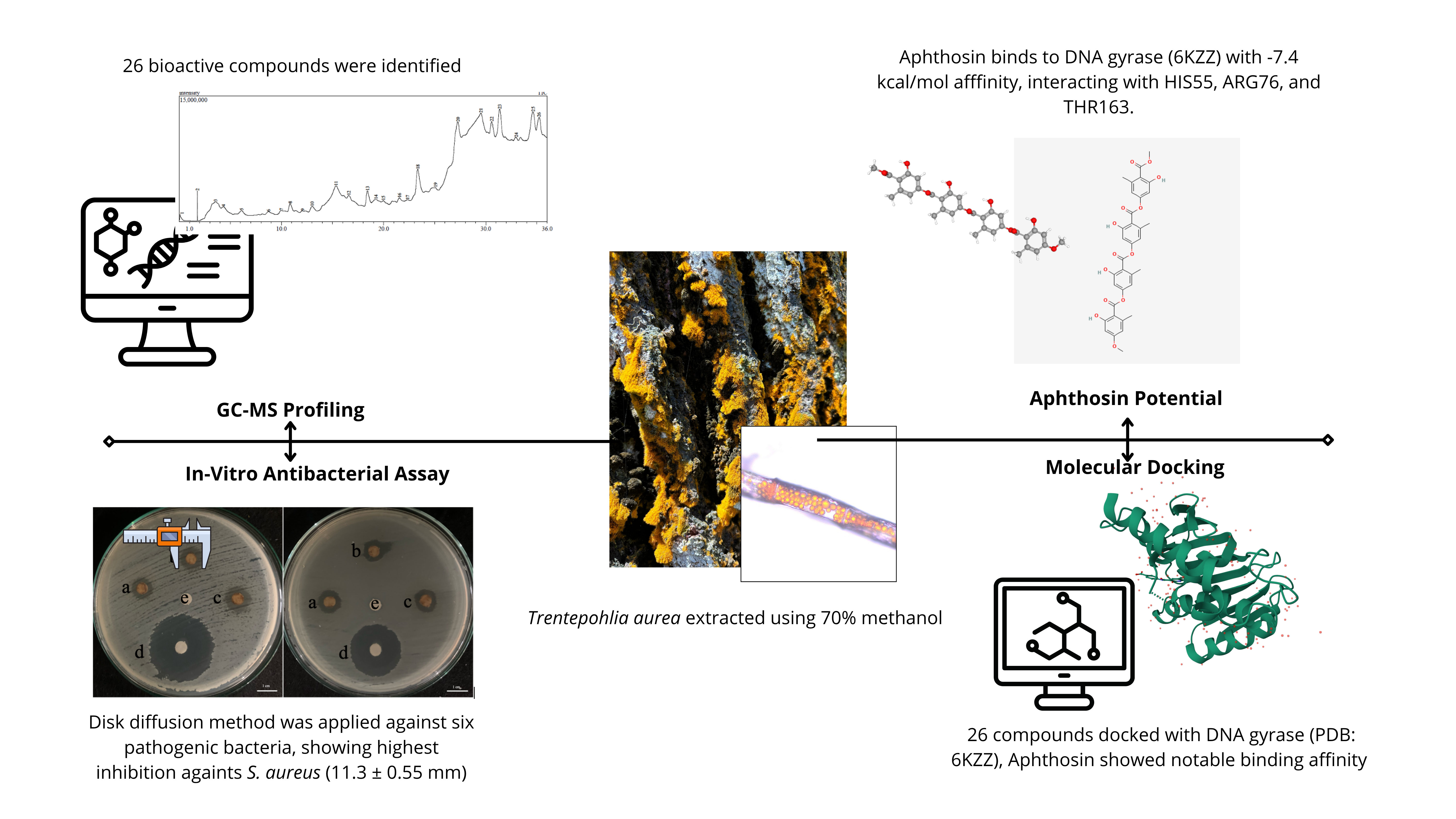
Downloads
References
MM El-Sheekh, N Abdullah and I Ahmad. Handbook of research on algae as a sustainable solution for food, energy, and the environment. IGI Global, Pennsylvania, 2022.
P Mallick and S Chatterjee. Textbook of algae. Techsar, New Delhi, India, 2024.
TNBT Ibrahim, NAS Feisal, NH Kamaludin, WY Cheah, V How, A Bhatnagar and PL Show. Biological active metabolites from microalgae for healthcare and pharmaceutical industries: A comprehensive review. Bioresource Technology 2023; 372, 12866.
L Chen, L Zhang and T Liu. Concurrent production of carotenoids and lipid by a filamentous microalga Trentepohlia arborum. Bioresource Technology 2016; 214, 567-573.
V Dolganyuk, D Belova, O Babich, A Prosekov, S Ivanova, D Katserov and S Sukhikh. Microalgae: A promising source of valuable bioproducts. Biomolecules 2020; 10(8), 1153.
N Mishra, E Gupta, P Singh and R Prasad. Application of microalgae metabolites in food and pharmaceutical industry. In: C Egbuna, AP Mishra and MR Goyal (Eds.). Preparation of phytopharmaceuticals for the management of disorders. Academic Press, London, 2023, p. 391-408.
H Upadhyay, L Saini and A Chakraborty. Trentepohlia. In: N Amaresan and K Kumar (Eds.). Compendium of phytopathogenic microbes in agro-ecology: Bacteria, protozoa, algae and nematodes. Springer Nature, Cham, Switzerland, 2025, p. 681-701.
S Saraphol, S Vajrodaya, E Kraichak, A Sirikhachornkit and N Sanevas. Environmental factors affecting the diversity and photosynthetic pigments of trentepohlia species in Northern Thailand’s chiang dao wildlife sanctuary. Acta Societatis Botanicorum Poloniae 2020; 89(1), 1-24.
R Kant, K Sarma, N Kumar, M Sharma, NC Halder, A Tyagi, D Gupta and V Malik. Biochemical profiling of chlorophylls, carotenoids, proteins and lipids of Trentepohlia aurea (l) c. Martius, Chlorophyta. Egyptian Journal of Phycology 2024; 25(1), 15-20.
D Zhang, M Wan, EA del Rio-Chanona, J Huang, W Wang and Y Li. Vassiliadis vs. dynamic modelling of Haematococcus pluvialis photoinduction for astaxanthin production in both attached and suspended photobioreactors. Algal Research 2016; 13, 69-78.
D Kharkongor and P Ramanujam. Antioxidant activities of 4 dominant species of Trentepohlia (Trentepohliales, Chlorophyta). International Journal of Complementary and Alternative Medicine 2017; 8(5), 00270.
E Sanniyasi and PP Raj. Accumulation of antioxidant pigment canthaxanthin (Β, Β-carotene-4, 4’-dione) from an aero-terrestrial, filamentous green microalga Trentepohlia Sp. Bioresource Technology Reports 2024; 25, 101803.
F Ushiyama, H Amada, T Takeuchi, N Tanaka-Yamamoto, H Kanazawa, K Nakano and N Ohtake. Lead identification of 8-(methylamino)-2-oxo-1, 2-dihydroquinoline derivatives as DNA gyrase inhibitors: Hit-to-lead generation involving thermodynamic evaluation. ACS Omega 2020; 5(17), 10145-10159.
S Ruan, T Chih-Han and CR Bourne. Friend or foe: Protein inhibitors of DNA gyrase. Biology 2024; 13(2), 84.
M Salman, P Sharma, M Kumar, AS Ethayathulla and P Kaur. Targeting novel sites in DNA gyrase for development of anti-microbials. Briefings in Functional Genomics 2023; 22(2), 180-194.
GS Bisacchi and JI Manchester. A new-class antibacterial almost. Lessons in drug discovery and development: A critical analysis of more than 50 years of effort toward ATPase inhibitors of DNA gyrase and topoisomerase IV. ACS Infectious Diseases 2025; 1(1), 4-41.
J Hudzicki. Kirby-Bauer disk diffusion susceptibility test protocol. American Society for Microbiology 2019; 15(1), 1-23.
OK Atni, E Munir and N Pasaribu. Diversity of bioactive compounds from Parmotrema xanthinum as antimicrobial potential through in-vitro and in-silico assessment. Biodiversitas Journal of Biological Diversity 2024; 25(11), 4438-4449.
OK Atni, E Munir and N Pasaribu. In-vitro and in-silico study of bioactive compounds from Coccocarpia erythroxyli as inhibitors targeting PBP3 (6ILE) for antibacterial applications. Trends in Sciences 2025; 22(8), 10291.
S Simic, M Kosanic and B Rankovic. Evaluation of in vitro antioxidant and antimicrobial activities of green microalgae Trentepohlia umbrina. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2012; 40(2), 86-91.
RM Epand, C Walker, RF Epand and NA Magarvey. Molecular mechanisms of membrane targeting antibiotics. Biochimica et Biophysica Acta (BBA)-Biomembranes 2016; 1858(5), 980-987.
MP Mingeot-Leclercq and JL Décout. Bacterial lipid membranes as promising targets to fight antimicrobial resistance, molecular foundations and illustration through the renewal of aminoglycoside antibiotics and emergence of amphiphilic aminoglycosides. MedChemComm 2016; 7(4), 586-611.
K Chekanov. Diversity and distribution of carotenogenic algae in Europe: A review. Marine Drugs 2023; 21(2), 108.
BA Jaber, KR Majeed and AG Al Hashimi. Antioxidant and antibacterial activity of β-carotene pigment extracted from Paracoccus homiensis strain bka7 isolated from Air of Basra, Iraq. Annals of the Romanian Society for Cell Biology 2021; 25(4), 14006-14027.
CA Lipinski, F Lombardo, BW Dominy and PJ Feeney. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews1997; 23(1-3), 3-25.
G Petit, C Kornreich, X Noel, P Verbanck and S Campanella. Alcohol-related context modulates performance of social drinkers in a visual Go/No-Go task: A preliminary assessment of event-related potentials. Plos One 2012; 7(5), e37466.
K Rajakumari, K Aravind, M Balamugundhan, M Jagadeesan, A Somasundaram, P Devi and P Ramasamy. Comprehensive review of DNA gyrase as enzymatic target for drug discovery and development. European Journal of Medicinal Chemistry Reports 2024; 12, 100233.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






