In-vitro and In-silico Study of Bioactive Compounds from Coccocarpia erythroxyli as Inhibitors Targeting PBP3 (6ILE) for Antibacterial Applications
DOI:
https://doi.org/10.48048/tis.2025.10291Keywords:
Antibacterial, Bioactive compounds, Coccocarpia erythroxyli, Penicillin-binding protein, Antibacterial, Bioactive compounds, Coccocarpia erythroxyli, Penicillin-binding proteinAbstract
Lichens are recognized as sources of diverse bioactive metabolites with potential antibacterial properties. This study evaluated the antibacterial activity of Coccocarpia erythroxyli (Spreng.) Swinscow & Krog and its interaction with Penicillin-Binding Protein 3 (PBP3, PDB ID: 6I1E). Methanol extracts of C. erythroxyli were tested against six bacterial strains using the disc diffusion method, revealing moderate antibacterial activity, with the highest inhibition zone observed against Escherichia coli (18.6 ± 0.44 mm), compared to chloramphenicol (29.13 mm). GC-MS analysis identified 33 bioactive compounds, which were further evaluated through in-silico drug-likeness screening and molecular docking. Among these, Cembrene showed the highest binding affinity (−5.9 kcal/mol), interacting with hydrophobic residues PHE182 and TRP185, while the control compound displayed a slightly stronger affinity (−6.3 kcal/mol). These results highlight the moderate antibacterial potential of C. erythroxyli and suggest that its metabolites warrant further investigation as lead compounds in antibacterial drug development, particularly in the context of antibiotic resistance.
HIGHLIGHTS
The graphical abstract illustrates the workflow of the study on Coccocarpia erythroxyli bioactive compounds as potential antibacterial agents. It begins with the extraction of bioactive compounds from the lichen, followed by in-vitro antibacterial testing. Simultaneously, in-silico molecular docking is conducted to evaluate the interaction of the compounds with PBP3 (6I1E), highlighting key binding sites and interaction energies. The combined results emphasize the potential of these compounds to inhibit bacterial growth and serve as a basis for future drug development targeting resistant bacteria.
GRAPHICAL ABSTRACT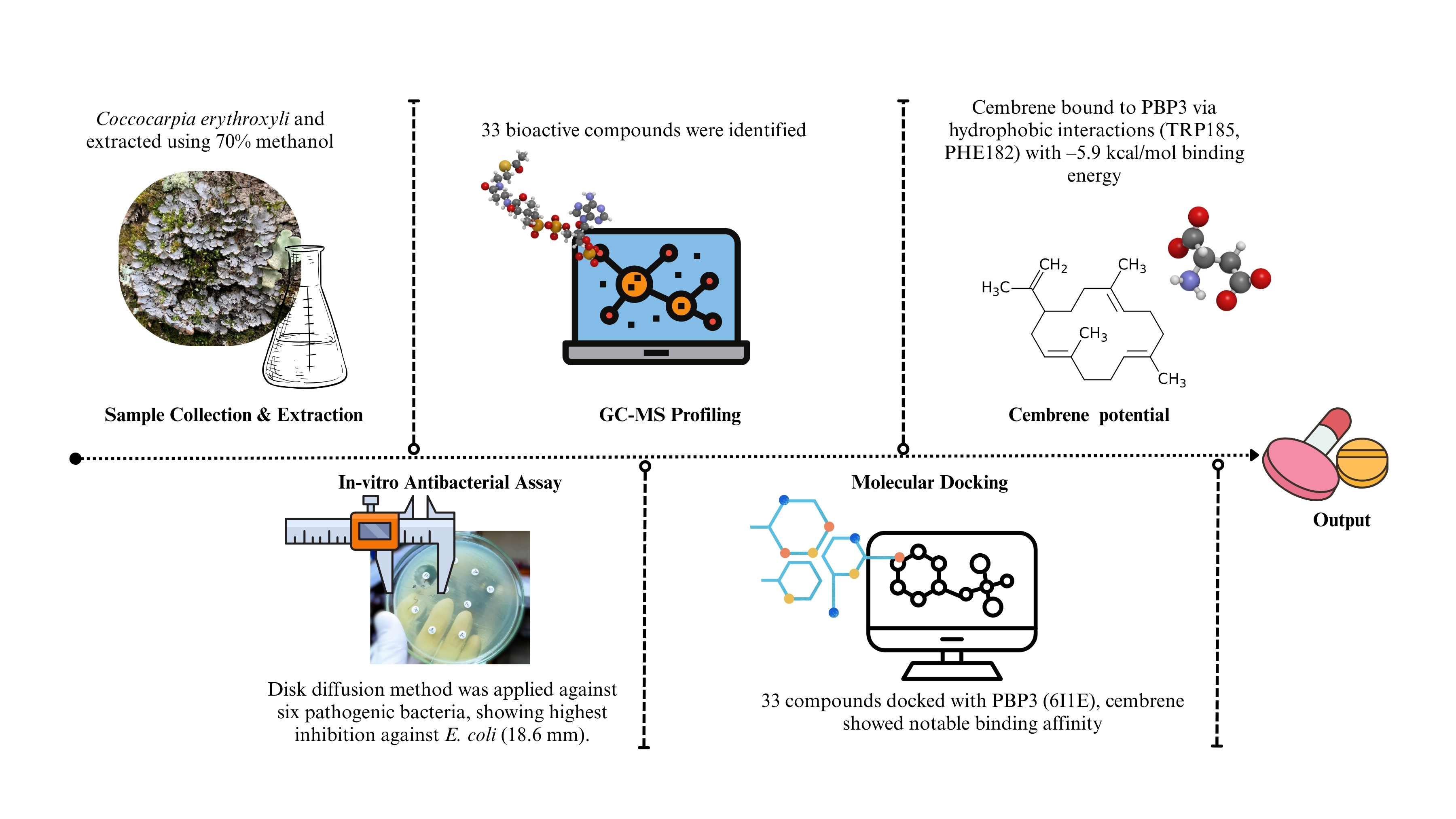
Downloads
References
Devashree, A Pandey and A Dikshit. Lichens: Fungal symbionts and their secondary metabolites. New and Future Developments in Microbial Biotechnology and Bioengineering. Elsevier, Netherlands, 2021.
OT Adenubi, IM Famuyide, LJ McGaw and JN Eloff. Lichens: An update on their ethnopharmacological uses and potential as sources of drug leads. Journal Ethnopharmacol 2022; 298, 115657.
B Ranković and M Kosanić. Lichens as a potential source of bioactive secondary metabolites. Springer Cham, Switzerland, 2019.
FE Dayan and JG Romagni. Lichens as a potential source of pesticides. Pesticide Outlook 2001; 12(6), 229-232.
V Shukla, GP Joshi and MSM Rawat. Lichens as a potential natural source of bioactive compounds: A review. Phytochemistry Reviews 2010; 9(2), 303-314.
LF Coca, HT Lumbsch, JA Mercado-Díaz, TJ Widhelm, B Goffinet, P Kirika and R Lücking. Diversity, phylogeny, and historical biogeography of the genus Coccocarpia (lichenized Ascomycota: Peltigerales) in the tropics. Molecular Phylogenetics and Evolution 2025; 206, 108312.
J Miadlikowska, F Kauff, V Hofstetter, E Fraker, M Grube, J Hafellner and F Lutzoni. New insights into classification and evolution of the Lecanoromycetes (Pezizomycotina, Ascomycota) from phylogenetic analyses of three ribosomal RNA-and two protein-coding genes. Mycologia 2006; 98(6), 1088-1103.
OK Atni, E Munir, ES Siregar and MN Saleh. Antimicrobial and antioxidant properties of the lichens Coccocarpia palmicola, Parmotrema clavuliferum and Parmotrema tinctorum. IOP Conference Series: Earth and Environmental Science 2025; 1445(1), 012024.
KS Vinayaka, KTRP Prashith and KSK Praveen. Antimicrobial activity of coccocarpia erythroxyli (Spreng.) swinc. & krog. Journal of Pharmacognosy and Phytochemistry 2017; 6(6), 2419-2422.
PJ Matteï, D Neves and A Dessen. Bridging cell wall biosynthesis and bacterial morphogenesis. Current Opinion in Structural Biology 2010; 20(6), 749-755.
A Typas, M Banzhaf, CA Gross and W Vollmer. From the regulation of peptidoglycan synthesis to bacterial growth and morphology. Nature Reviews Microbiology 2012; 10(2), 123-136.
A Zapun, C Contreras-Martel and T Vernet. Penicillin-binding proteins and β-lactam resistance. FEMS Microbiology Reviews 2008; 32(2), 361-385.
E Sauvage, K Kerff Bush and PA Bradford. β-Lactams and β-lactamase inhibitors: An overview. Cold Spring Harbor Perspectives in Medicine 2016; 6(8), 025247.
E Sauvage, F Kerff, M Terrak, JA Ayala and P Charlier. The penicillin-binding proteins: Structure and role in peptidoglycan biosynthesis. FEMS Microbiology Reviews 2008; 32(2), 234-258.
AF Bertonha, CC Silva, KT Shirakawa, DM Trindade and A Dessen. Penicillin-binding protein (PBP) inhibitor development: A 10-year chemical perspective. Experimental Biology and Medicine 2023; 248(19), 1657-1670.
N Pasaribu, OK Atni and JP Siregar. Diversity and species composition of lichens across altitudinal range in the Batang Toru Forest North Sumatra Indonesia. Biodiversitas: Journal of Biological Diversity 2023; 24(4), 2171-2178.
OK Atni, E Munir, ES Siregar and M Saleh. Lichen diversity and taxonomy in Bukit Barisan Grand Forest Park North Sumatra Indonesia. Biodiversitas: Journal of Biological Diversity 2024; 25(4), 1623-1630.
A Russell and J Furr. Susceptibility of porin- and lipopolysaccharide- deficient strains of Escherichia coli to some antiseptics and disinfectants. Journal of Hospital Infection 1986; 8, 47-56.
M Yoneyama, A Fujii and S Maeda. Wavelength-doubled spiral fragments in photosensitive monolayers. Journal of the American Chemical Society 1995; 117(31), 8188-8191.
PA James and DS Reeves. Bacterial resistance to cephalosporins as a function of outer membrane permeability and access to their target. Journal of Chemotherapy 1996; 8, 37-47.
J Jayakumar, VA Kumar, L Biswas, R Biswas. Therapeutic applications of lysostaphin against Staphylococcus aureus. Journal of Applied Microbiology. 2021; 131(3), 1072-1082.
MDCDF Bastos, BG Coutinho, MLV Coelho. Lysostaphin: A staphylococcal bacteriolysin with potential clinical applications. Pharmaceuticals. 2010; 3(4), 1139-1161.
CA Lipinski, F Lombardo, BW Dominy and PJ Feeney. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews 1997; 23, 3-25.
CA Lipinski, F Lombardo, BW Dominy and PJ Feeney. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews 2001; 46, 3-26.
MG Drew, DH Templeton and A Zalkin. The crystal and molecular structure of cembrene. Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials 1969; 25(2), 261-267.
M Zubair, W Alarif, K Al-Footy, M Ph, M Ali, S Basaif, S Al-Lihaibi and SE Ayyad. New antimicrobial biscembrane hydrocarbon and cembranoid diterpenes from the soft coral Sarcophyton trocheliophorum. Turkish Journal of Chemistry 2016; 40(3), 385-392.
LW Chen, HL Chung, CC Wang, JH Su, YJ Chen and CJ Lee. Anti-acne effects of cembrene diterpenoids from the cultured soft coral Sinularia flexibilis. Marine Drugs 2020; 18(10), 487.
D Bellini, L Koekemoer, H Newman and CG Dowson. Novel and improved crystal structures of H. influenzae, E. coli and P. aeruginosa penicillin-binding protein 3 (PBP3) and N. gonorrhoeae PBP2: Toward a better understanding of β-lactam target-mediated resistance. Journal of Molecular Biology 2019; 431(18), 3501-3519.
OK Atni, E Munir and N Pasaribu. Diversity of bioactive compounds from Parmotrema xanthinum as antimicrobial potential through in-vitro and in-silico assessment. Biodiversitas: Journal of Biological Diversity 2024; 25(11), 4438-4449.
M Dabhi, R Patel, V Shah, R Soni, M Saraf, R Rawal and D Goswami. Penicillin-binding proteins: The master builders and breakers of bacterial cell walls and its interaction with β-lactam antibiotics. Journal of Proteins and Proteomics. 2024; 15(2), 215-232.
LM Jacobs, P Consol and Y Chen. Drug discovery in the field of β-Lactams: An academic perspective. Antibiotics 2024; 13(1), 59.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






