Homogenization-Assisted Extraction of Antioxidants from Centella asiatica Using a Natural Deep Eutectic Solvent (NADES): Optimization and Evaluation
DOI:
https://doi.org/10.48048/tis.2026.11303Keywords:
Centella asiatica, Natural deep eutectic solvents (NADES), Homogenization-assisted extraction (HAE), Box-Behnken design, Green extraction, Antioxidant capacityAbstract
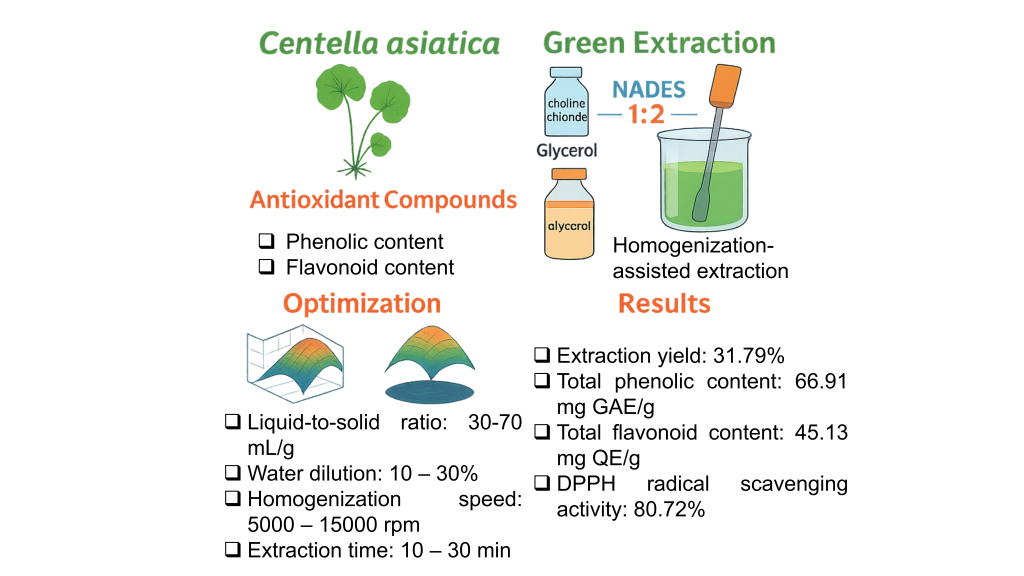
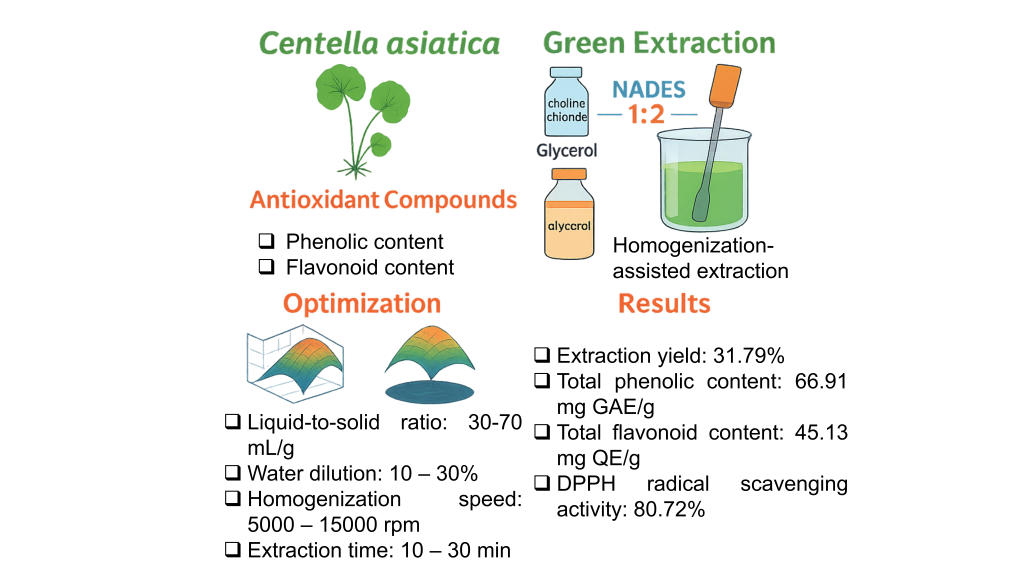
Centella asiatica (L.) Urban is a valued medicinal herb rich in antioxidant compounds, notably phenolics and flavonoids. This study aimed to develop a green and effective method for extracting these bioactives using natural deep eutectic solvents (NADES) coupled with homogenization-assisted extraction (HAE). Twelve NADES systems were screened, and choline chloride:glycerol (1:2) demonstrated the highest efficiency due to its moderate viscosity and strong solvation capacity. A Box-Behnken Design combined with response surface methodology was employed to optimize 4 key factors: Liquid-to-solid ratio (30 - 70 mL/g), solvent concentration (10% - 30%), homogenization speed (5,000 - 15,000 rpm), and extraction time (10 - 30 min). Optimal conditions (50 mL/g, 20% solvent, 10,000 rpm, 20 min) achieved a 31.79% extraction yield, 66.91 mg GAE/g of total phenolic content, 45.13 mg QE/g of total flavonoid content, and 80.72% DPPH radical scavenging activity. Statistical models exhibited strong predictive power (R2 > 0.91), validating the process efficiency. The integration of NADES and HAE provides a scalable, solvent-free alternative to conventional extraction, supporting sustainable production of antioxidant-rich extracts. This method holds promise for applications in functional foods, pharmaceuticals, and cosmetics where clean-label, eco-friendly ingredients are in demand.
HIGHLIGHTS
- A green extraction method was developed using NADES and homogenization for Centella asiatica.
- Box-Behnken Design was used to optimize L/S ratio, water content, speed, and time.
- Optimal conditions yielded high phenolic (66.91 mg GAE/g) and flavonoid content (45.13 mg QE/g).
- The method preserved antioxidant activity with 80.72% DPPH radical scavenging.
- The process is eco-friendly, scalable, and suitable for food, cosmetic, and pharmaceutical industries.
GRAPHICAL ABSTRACT
Downloads
References
JT James and IA Dubery. Pentacyclic triterpenoids from the medicinal herb, Centella asiatica (L.) urban. Molecules 2009; 14, 3922-3941.
B Brinkhaus, M Lindner, D Schuppan and EG Hahn. Chemical, pharmacological and clinical profile of the East Asian medical plant Centella aslatica. Phytomedicine 2000; 7(5), 427-448.
NE Gray, AA Magana, P Lak, KM Wright, J Quinn, JF Stevens, CS Maier and A Soumyanath. Centella asiatica: Phytochemistry and mechanisms of neuroprotection and cognitive enhancement. Phytochemistry Reviews 2018; 17(1), 161-194.
S Tripathy, DK Verma, M Thakur, N Chakravorty, S Singh and PP Srivastav. Recent trends in extraction, identification and quantification methods of Centella asiatica phytochemicals with potential applications in food industry and therapeutic relevance: A review. Food Bioscience 2022; 49, 101864.
MK Khan, L Paniwnyk and S Hassan. Polyphenols as natural antioxidants: Sources, extraction and applications in food, cosmetics and drugs. In: Y Li and F Chemat (Eds.). Plant based “green chemistry 2.0”. Green chemistry and sustainable technology. Springer, Singapore, 2019, p. 197-235.
R Kunjumon, AJ Johnson and S Baby. Centella asiatica: Secondary metabolites, biological activities and biomass sources. Phytomedicine Plus 2022; 2(1), 100176.
M Vandorou, C Plakidis, IM Tsompanidou, T Adamantidi, EA Panagopoulou and A Tsoupras. A review on apple pomace bioactives for natural functional food and cosmetic products with therapeutic health-promoting properties. International Journal of Molecular Sciences 2024; 25(19), 10856.
F Chemat, MA Vian and G Cravotto. Green extraction of natural products: Concept and principles. International Journal of Molecular Sciences 2012; 13(7), 8615-8627.
CP Mungwari, CK King’ondu, P Sigauke and BA Obadele. Conventional and modern techniques for bioactive compounds recovery from plants: Review. Scientific African 2025; 27, e02509.
U Roobab, RM Aadil, SS Kurup and S Maqsood. Comparative evaluation of ultrasound-assisted extraction with other green extraction methods for sustainable recycling and processing of date palm bioresources and by-products: A review of recent research. Ultrasonics Sonochemistry 2025; 114, 107252.
C Bitwell, SS Indra, C Luke and MK Kakoma. A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants. Scientific African 2023; 19, e01585.
A Paiva, R Craveiro, I Aroso, M Martins, RL Reis and ARC Duarte. Natural deep eutectic solvents - solvents for the 21st century. ACS Sustainable Chemistry & Engineering 2014; 2(5), 1063-1071.
JJ Queffelec, W Beraud, MD Torres and H Domínguez. Advances in obtaining ready to use extracts with natural solvents. Sustainable Chemistry and Pharmacy 2024; 38, 101478.
FS Bragagnolo, MM Strieder, RS Pizani, LM de Souza Mesquita, M González-Miquel and MA Rostagno. Revisiting natural deep eutectic solvents (NADES) as extraction media and ready-to-use purposes. TrAC Trends in Analytical Chemistry 2024; 175, 117726.
DT da Silva, R Pauletto, S da Silva Cavalheiro, VC Bochi, E Rodrigues, J Weber, CB da Silva, FDP Morisso, MT Barcia and T Emanuelli. Natural deep eutectic solvents as a biocompatible tool for the extraction of blueberry anthocyanins. Journal of Food Composition and Analysis 2020; 89, 103470.
FM Fuad, MM Nadzir and A Harun. Hydrophilic natural deep eutectic solvent: A review on physicochemical properties and extractability of bioactive compounds. Journal of Molecular Liquids 2021; 339, 116923.
B Socas-Rodríguez, MV Torres-Cornejo, G Álvarez-Rivera and JA Mendiola. Deep eutectic solvents for the extraction of bioactive compounds from natural sources and agricultural by-products. Applied Sciences 2021; 11, 4897.
MH Zainal-Abidin, M Hayyan, A Hayyan and NS Jayakumar. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Analytica Chimica Acta 2017; 979, 1-23.
Ü Erdoğan, TH Öztürk, S Önder and M Tonguç. Eco-friendly extraction of 6 different rosemary (Rosmarinus officinalis L.) genotypes-using natural deep eutectic solvents: Optimization and modeling via response surface methodology (RSM). Journal of Molecular Liquids 2024; 407, 125167.
Z Wei, W Zhang, M Du, H Zhong and X Fang. Widely targeted metabolomic and KEGG analyses of natural deep eutectic solvent-based saponins extraction from Camellia oleifera Abel.: Effects on composition. Food Chemistry 2024; 450, 139333.
KJ Lanjekar and VK Rathod. Green extraction of Glycyrrhizic acid from Glycyrrhiza glabra using choline chloride based natural deep eutectic solvents (NADESs). Process Biochemistry 2021; 102, 22-32.
S Carpentieri, F Soltanipour, G Ferrari, G Pataro and F Donsì. Emerging green techniques for the extraction of antioxidants from agri-food by-products as promising ingredients for the food industry. Antioxidants 2021; 10(9), 1417.
C Cannavacciuolo, S Pagliari, R Celano, L Campone and L Rastrelli. Critical analysis of green extraction techniques used for botanicals: Trends, priorities, and optimization strategies - a review. TrAC Trends in Analytical Chemistry 2024; 173, 117627.
P Kumari, P Kaur, V Kumar, B Pandey, R Nazir, K Katoch, P Dwivedi, A Dey and DK Pandey. Response surface methodology and artificial neural network modeling for optimization of ultrasound-assisted extraction and rapid HPTLC analysis of asiaticoside from Centella asiatica. Industrial Crops and Products 2022; 176, 114320.
FM Fuad and MM Nadzir. Ultrasound-assisted extraction of asiaticoside from Centella asiatica using betaine-based natural deep eutectic solvent. Industrial Crops and Products 2023; 192, 116069.
P Mohapatra, A Ray, S Jena, S Nayak and S Mohanty. Influence of extraction methods and solvent system on the chemical composition and antioxidant activity of Centella asiatica L. leaves. Biocatalysis and Agricultural Biotechnology 2021; 33, 101971.
N Ji, C Yu, X Han, X He, S Kang, T Bai, H Liu, G Chen, M Cao and G Liu. Effects of 3 processing technologies on the structure and immunoreactivity of α-tropomyosin from Haliotis discus hannai. Food Chemistry 2023; 405, 134947.
C Neji, A Muthu, D Ungai, E Seres, É Domokos-Szabolcsy, J Prokisch, E Máthé and P Sipos. Assessing deep eutectic solvent efficiency by optimizing soybean protein extraction. Food and Bioprocess Technology 2025. https://doi.org/10.1007/s11947-025-03973-9
S Srivastava, VK Pandey, A Fatima, S Pandey, R Singh, AH Dar and B Dhillon. A literature review on process intensification: An innovative and sustainable food processing method. Applied Food Research 2024; 4(1), 100363.
J Ponphaiboon, W Krongrawa, WW Aung, N Chinatangkul, S Limmatvapirat and C Limmatvapirat. Advances in natural product extraction techniques, electrospun fiber fabrication, and the integration of experimental design: A comprehensive review. Molecules 2023; 28, 5163.
C Wang, Y You, W Huang and J Zhan. Efficient recovery of procyanidins from grape residue by UAE-NADES and macroporous resin. Ultrasonics Sonochemistry 2025; 119, 107400.
Q Zhang, KDO Vigier, S Royer and F Jérôme. Deep eutectic solvents: syntheses, properties and applications. Chemical Society Reviews 2012; 41(21), 7108-7146.
Y Dai, J Van Spronsen, GJ Witkamp, R Verpoorte and YH Choi. Natural deep eutectic solvents as new potential media for green technology. Analytica Chimica Acta 2013; 766, 61-68.
ND de Lima, BRSM Wanderley, ALA Ferreira, M Pereira-Coelho, IC da Silva Haas, L Vitali, L.A. dos Santos Madureira, JM Müller, CB Fritzen-Freire and RDMC Amboni. Green extraction of phenolic compounds from the by-product of purple araçá (Psidium myrtoides) with natural deep eutectic solvents assisted by ultrasound: Optimization, comparison, and bioactivity. Food Research International 2024; 191, 114731.
IL Oliveira, G Domínguez-Rodríguez, L Montero, J Viganó, A Cifuentes, MA Rostagno and E Ibáñez. Advanced extraction techniques combined with natural deep eutectic solvents for extracting phenolic compounds from pomegranate (Punica granatum L.) peels. International Journal of Molecular Sciences 2024; 25(18), 9992.
L Hu, Y Luo, J Yang and C Cheng. Botanical flavonoids: Efficacy, absorption, metabolism and advanced pharmaceutical technology for improving bioavailability. Molecules 2025; 30, 1184.
LA Jiménez-Ortega, J Kumar-Patra, RG Kerry, G Das, JD Mota-Morales and JB Heredia. Synergistic antioxidant activity in deep eutectic solvents: Extracting and enhancing natural products. ACS Food Science & Technology 2024; 4(12), 2776-2798.
TP Vo, TV Pham, K Weina, TNH Tran, LTV Vo, PT Nguyen, TLH Bui, TH Phan and DQ Nguyen. Green extraction of phenolics and flavonoids from black mulberry fruit using natural deep eutectic solvents: Optimization and surface morphology. BMC Chemistry 2023; 17(1), 119.
LS Chua, HY Thong and J Soo. Effect of pH on the extraction and stability of anthocyanins from jaboticaba berries. Food Chemistry Advances 2024; 5, 100835.
V Taco, D Almachi, P Bonilla, I Gijón-Arreortúa, S Benali, JM Raquez, P Duez and A Nachtergael. Natural Deep Eutectic Solvents (NADESs) for the extraction of bioactive compounds from quinoa (Chenopodium quinoa Willd.) leaves: A semi-quantitative analysis using high performance thin-layer chromatography. Molecules 2025; 30(12), 2620.
H Kivelä, M Salomäki, P Vainikka, E Mäkilä, F Poletti, S Ruggeri, F Terzi and J Lukkari. Effect of water on a hydrophobic deep eutectic solvent. The Journal of Physical Chemistry B 2022; 126, 513-527.
FN Idris and M Mohd Nadzir. Comparative studies on different extraction methods of Centella asiatica and extracts bioactive compounds effects on antimicrobial activities. Antibiotics 2021; 10(4), 457.
S Phaisan, F Makkliang, W Putalun, S Sakamoto and G Yusakul. Development of a colorless Centella asiatica (L.) Urb. extract using a natural deep eutectic solvent (NADES) and microwave-assisted extraction (MAE) optimized by response surface methodology. RSC Advances 2021; 11(15), 8741-8750.
J Chen, C Jiang, Z Liu, P Wang, Q Ma and N Zhou. Study on optimization of extraction and purification processes for total flavonoids from Lycopi herba roots and their anti-proliferative effects on fibrous synoviocytes in human rheumatoid arthritis. Ultrasonics Sonochemistry 2025; 112, 107164.
MJ Tan, Y Li, SQ Zhao, FH Yue, DJ Cai, JT Wu, XA Zeng, J Li and Z Han. Synergistic ultrasound pulsed electric field extraction of litchi peel polyphenols and determination of their properties. International Journal of Biological Macromolecules 2024; 260, 129613.
YR Yi and JR Koseff. Underlying physics of mixing efficiency for shear-forced, stratified turbulence. Physical Review Fluids 2023; 8(8), 084803.
A Anvar, MH Azizi and HA Gavlighi. Exploring the effect of natural deep eutectic solvents on zein: Structural and functional properties. Current Research in Food Science 2025; 10, 100965.
MA Alam, G Muhammad, MN Khan, M Mofijur, Y Lv, W Xiong and J Xu. Choline chloride-based deep eutectic solvents as green extractants for the isolation of phenolic compounds from biomass. Journal of Cleaner Production 2021; 309, 127445.
C Spaggiari, L Carbonell-Rozas, H Zuilhof, G Costantino and L Righetti. Structural elucidation and long-term stability of synthesized NADES: A detailed physicochemical analysis. Journal of Molecular Liquids 2025; 424, 127105.
J Yang, N Li, C Wang, T Chang and H Jiang. Ultrasound-homogenization-assisted extraction of polyphenols from coconut mesocarp: Optimization study. Ultrasonics Sonochemistry 2021; 78, 105739.
O Soufi, L Medouni-Haroune, M Bachirbey, S Medouni-Adrar, F Idir, T Heddad, L Ouldsaadi, C Romero, K Madani and L Makhlouf-Boulekbache. Statistical optimization of ultrasound-assisted extraction of polyphenols from olive pomace. Sustainable Chemistry and Pharmacy 2023; 36, 101260.
M Coscarella, M Nardi, K Alipieva, S Bonacci, M Popova, A Procopio, R Scarpelli and S Simeonov. Alternative assisted extraction methods of phenolic compounds using NaDESs. Antioxidants 2023; 13(1), 62.
A Antony and M Farid. Effect of temperatures on polyphenols during extraction. Applied Sciences 2022; 12, 2107.
W Wang, Y Pan, J Zhao, Y Wang, Q Yao and S Li. Development and optimization of green extraction of polyphenols in Michelia alba using natural deep eutectic solvents (NADES) and evaluation of bioactivity. Sustainable Chemistry and Pharmacy 2024; 37, 101425.
RC Lima, APA de Carvalho, BD da Silva, LT Neto, MRS Figueiredo, PHT Chaves, AECC de Almeida and CA Conte-Junior. Green ultrasound-assisted extraction of bioactive compounds of babassu (Attalea speciosa) mesocarp: Effects of solid-liquid ratio extraction, antioxidant capacity, and antimicrobial activity. Applied Food Research 2023; 3(2), 100331.
E Gil-Martín, T Forbes-Hernández, A Romero, D Cianciosi, F Giampieri and M Battino. Influence of the extraction method on the recovery of bioactive phenolic compounds from food industry by-products. Food Chemistry 2022; 378, 131918.
SJ Fan, XY Zhang, Y Cheng, YX Qiu, YY Hu, T Yu, WZ Qian, DJ Zhang and S Gao. Extraction optimization of phenolic compounds from Triadica sebifera leaves: Identification, characterization and antioxidant activity. Molecules 2024; 29(14), 3266.
MAR Himel, T Ahmed, MA Hossain and MS Moazzem. Response surface optimization to extract antioxidants from freeze-dried seeds and peel of pomegranate (Punica granatum L.). Biomass Conversion and Biorefinery 2024; 14(8), 9707-9722.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






