In Silico Evaluation of Novel 3-Piperoylindole Compounds Synthesized from Piperine (Piper Nigrum Linn.) for Potential Anti-Cancer
DOI:
https://doi.org/10.48048/tis.2025.10466Keywords:
3-piperoylindole, EGFR tyrosine kinase, Piper nigrum Linn., Anticancer, In silicoAbstract
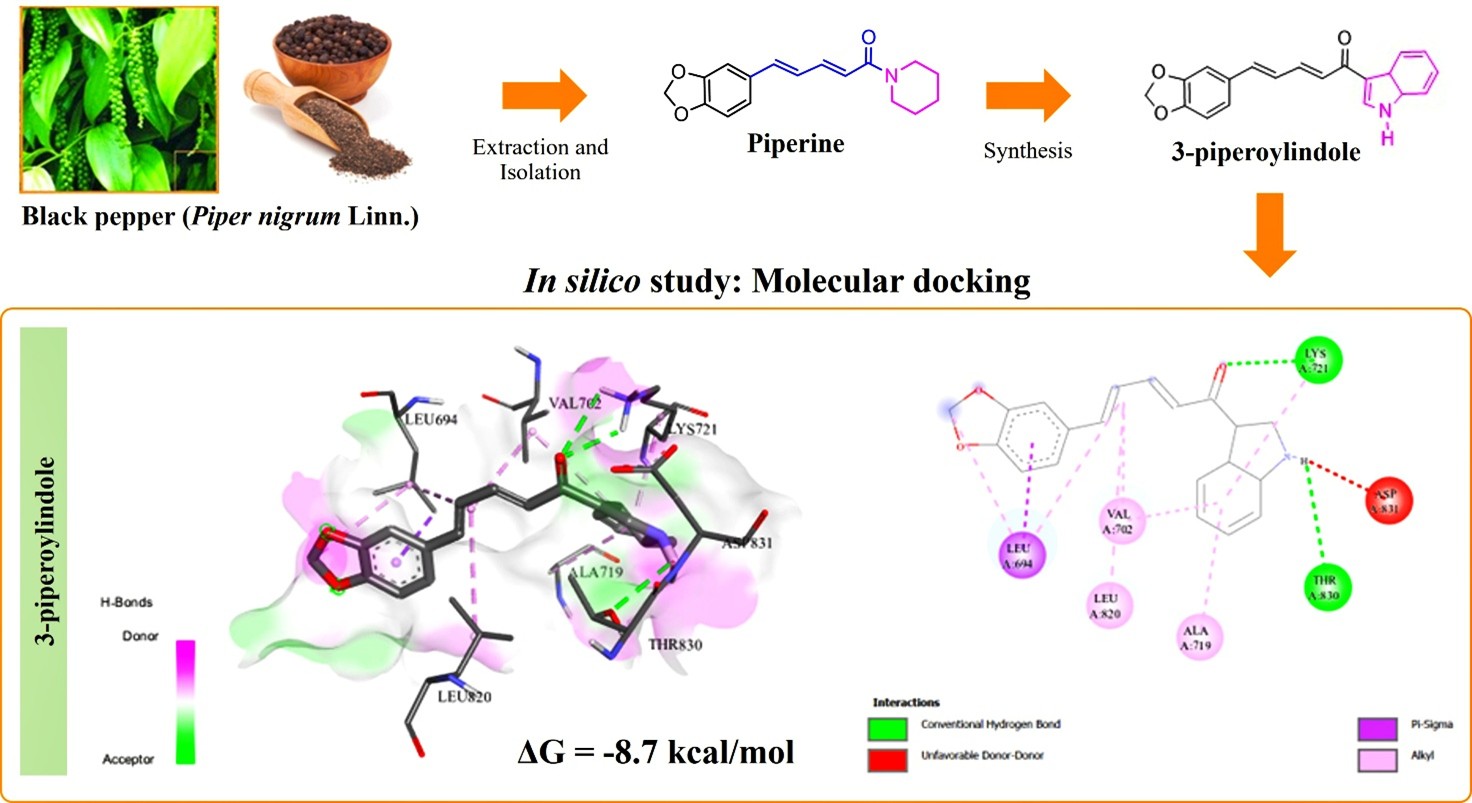
The development of drugs that inhibit EGFR tyrosine kinase (EGFR-TK) is necessary to improve the effectiveness and durability of cancer treatment. In silico study employed a molecular docking approach to simulate the interaction between 3-piperoylindole (3-PI), a novel semi-synthetic derivative of piperine (Piper nigrum Linn.), and EGFR-TK. Redocking between the receptor and the original ligand yielded an RMSD value of 1.382 Å. The yield of the 3-PI compound was 37% w/w from piperoic acid. The docking results revealed that 3-PI (−8.7 kcal/mol) had a higher binding affinity than erlotinib (−7.7 kcal/mol) toward EGFR-TK. The binding affinity of 3-PI toward EGFR-TK was attributed to hydrophobic interactions and hydrogen bonds. These findings highlight the potential of this compound as anti-cancer for further in vitro and in vivo evaluation.
HIGHLIGHTS
- 3-piperoylindole (3-PI) was synthesized from piperine (Piper nigrum) via a pre-target compound (piperoyl chloride) mechanism.
- 3-PI compound was obtained 37% w/w of yield from piperoic acid.
- The binding energy of the 3-PI compound evaluated using molecular docking approach showed −8.7 kcal/mol, which had a higher binding affinity than erlotinib (−7.7 kcal/mol) toward EGFR-TK.
- The binding affinity of 3-PI toward EGFR-TK was attributed to hydrophobic interactions and hydrogen bonds. These findings highlight the potential of this compound as anti-cancer for further in vitro and in vivo
GRAPHICAL ABSTRACT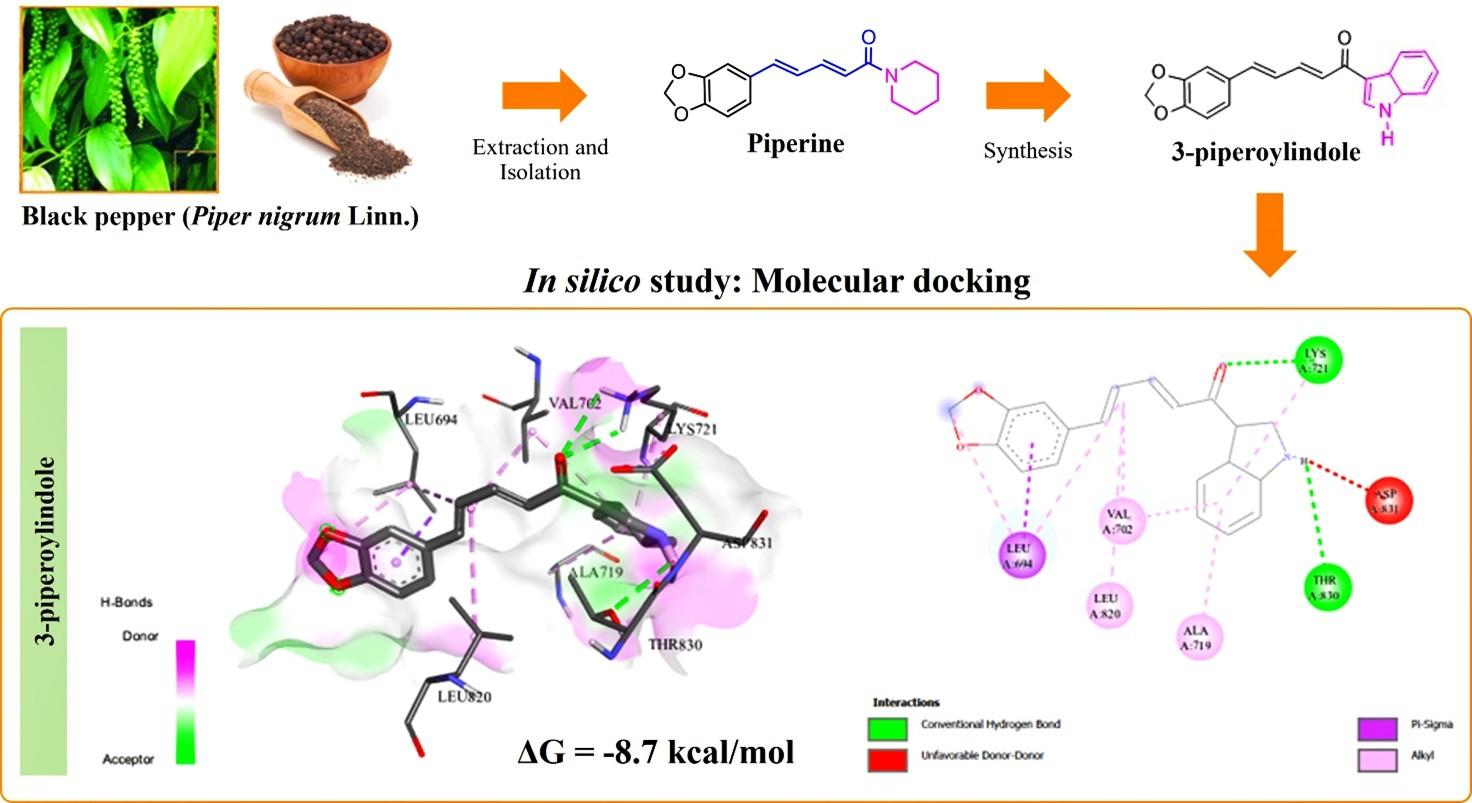
Downloads
References
RL Siegel, TB Kratzer, AN Giaquinto, H Sung and A Jemal. Cancer statistics, 2025. CA: A Cancer Journal for Clinicians 2025; 75(1), 10-45.
A Sharma, S Jasrotia and A Kumar. Effects of chemotherapy on the immune system: Implications for cancer treatment and patient outcomes. Naunyn-Schmiedeberg’s Archives of Pharmacology 2023; 397(5), 2551-2566.
J Rueff and AS Rodrigues. Cancer drug resistance: A brief overview from a genetic viewpoint. In: J Rueff and AS Rodrigues (Eds.). Methods in Molecular Biology. Humana Press, New York, 2016.
M Habban Akhter, N Sateesh Madhav and J Ahmad. Epidermal growth factor receptor based active targeting: A paradigm shift towards advance tumor therapy. Artificial Cells, Nanomedicine, and Biotechnology 2018; 46(2), 1188-1198.
E Kovacs, JA Zorn, Y Huang, T Barros and J Kuriyan. A structural perspective on the regulation of the epidermal growth factor receptor. Annual Review of Biochemistry 2015; 84(1), 739-764.
JJ Laskin and AB Sandler. Epidermal growth factor receptor: A promising target in solid tumours. Cancer Treatment Reviews 2004; 30(1), 1-17.
RM Sylva and SI Ahmed. Targeted therapies in cancer. Surgery 2024; 42(3), 150-155.
C Schettino, MA Bareschino, P Maione, A Rossi, F Ciardiello and C Gridelli. Small molecule epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors in non-small cell lung cancer treatment. Current Cancer Therapy Reviews 2007; 3(4), 226-235.
XL Huang, MI Khan, J Wang, R Ali, SW Ali, QA Zahra, A Kazmi, A Lolai, YL Huang, A Hussain, M Bilal and F Li. Role of receptor tyrosine kinases mediated signal transduction pathways in tumor growth and angiogenesis - New insight and futuristic vision International Journal of Biological Macromolecules 2021; 180, 739-752.
CJ Tsai and R Nussinov. Emerging allosteric mechanism of EGFR activation in physiological and pathological contexts. Biophysical Journal 2019; 117(1), 5-13.
DA Sabbah, R Hajjo and K Sweidan. Review on epidermal growth factor receptor (EGFR) structure, signaling pathways, interactions, and recent updates of EGFR inhibitors. Current Topics in Medicinal Chemistry 2020; 20(10), 815-834.
AK Kashyap and SK Dubey. Molecular mechanisms in cancer development. In: B Jain and S Pandey (Eds.). Understanding cancer. Academic Press, New York, 2022, p. 79-90.
R Damare, K Engle and G Kumar. Targeting epidermal growth factor receptor and its downstream signaling pathways by natural products: A mechanistic insight. Phytotherapy Research 2024; 38(5), 2406-2447.
H Takooree, MZ Aumeeruddy, KRR Rengasamy, KN Venugopala, R Jeewon, G Zengin and MF Mahomoodally. A systematic review on black pepper (Piper nigrum L.): From folk uses to pharmacological applications. Critical Reviews in Food Science and Nutrition 2019; 59(S1), S210-S243.
PV Dludla, I Cirilli, F Marcheggiani, S Silvestri, P Orlando, N Muvhulawa, MT Moetlediwa, BB Nkambule, SE Mazibuko-Mbeje, N Hlengwa, S Hanser, D Ndwandwe, JL Marnewick, AK Basson and L Tiano. Bioactive properties, bioavailability profiles, and clinical evidence of the potential benefits of black pepper (Piper nigrum) and red pepper (Capsicum annum) against diverse metabolic complications. Molecules 2023; 28(18), 6569.
DR Anandh and D Priya. Review on the pharmacological activities of black pepper. Journal of Natural Remedies 2024; 24(3), 441-451.
Y Liu, VR Yadev, BB Aggarwal and MG Nair. Inhibitory effects of black pepper (Piper Nigrum) extracts and compounds on human tumor cell proliferation, cyclooxygenase enzymes, lipid peroxidation and nuclear transcription factor-kappa-B. Natural Product Communications 2010; 5(8), 1253-1257.
PM Paarakh, DC Sreeram and SPS Ganapathy. In vitro cytotoxic and in silico activity of piperine isolated from Piper nigrum fruits Linn. In Silico Pharmacology 2015; 3(1), 9.
S Bahri, Y Ambarwati, M Iqbal and AA Baihaqy. Synthesis 4-piperoilmorpholine from piperine. Journal of Physics: Conference Series 2019; 1338(1), 012010.
NK Singh, P Kumar, DK Gupta, S Singh, and VK Singh. UV-spectrophotometric method development for estimation of piperine in Chitrakadi Vati. Der Pharmacia Lettre 2011; 3(3), 178-182.
L Gorgani, M Mohammadi, GD Najafpour and M Nikzad. Sequential microwave-ultrasound-assisted extraction for isolation of piperine from black pepper (Piper Nigrum L.). Food and Bioprocess Technology 2017; 10(12), 2199-2207.
A Yasir, S Ishtiaq, M Jahangir, M Ajaib, U Salar and KM Khan. Biology-Oriented Synthesis (BIOS) of piperine derivatives and their comparative analgesic and antiinflammatory activities. Medicinal Chemistry 2018; 14(3), 269-280.
P Choochana, J Moungjaroen, N Jongkon, W Gritsanapan and P Tangyuenyongwatana. Development of piperic acid derivatives from Piper nigrum as UV protection agents. Pharmaceutical Biology 2014; 53(4), 477-482.
Z Zarai, E Boujelbene, NB Salem, Y Gargouri and A Sayari. Antioxidant and antimicrobial activities of various solvent extracts, piperine and piperic acid from Piper nigrum. LWT - Food Science and Technology 2013; 50(2), 634-641.
DM Aziz, JR Hama and SM Alam. Synthesising a novel derivative of piperine from black pepper (Piper Nigrum L.). Journal of Food Measurement and Characterization 2015; 9(3), 324-331.
I Kufareva and R Abagyan. Methods of protein structure comparison. In: A Orry and R Abagyan (Eds.). Methods in molecular biology. Humana Press, New Jersey, United States, 2011, p. 231-257.
M Sandor, R Kiss and GM Keserű. Virtual fragment docking by glide: A validation study on 190 protein-fragment complexes. Journal of Chemical Information and Modeling 2010; 50(6), 1165-1172.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






