The Impact of Streptozotocin-Induced Diabetes on Testosterone Hormone and Androgen Receptor Expression and Correlation with Sperm Quality Impairment in Sprague Dawley Rats
DOI:
https://doi.org/10.48048/tis.2025.10409Keywords:
Streptozotocin, Type 2 diabetes mellitus, Testosterone, Testicular histopathology, Spermatozoa, Androgen receptor, Sprague DawleyAbstract
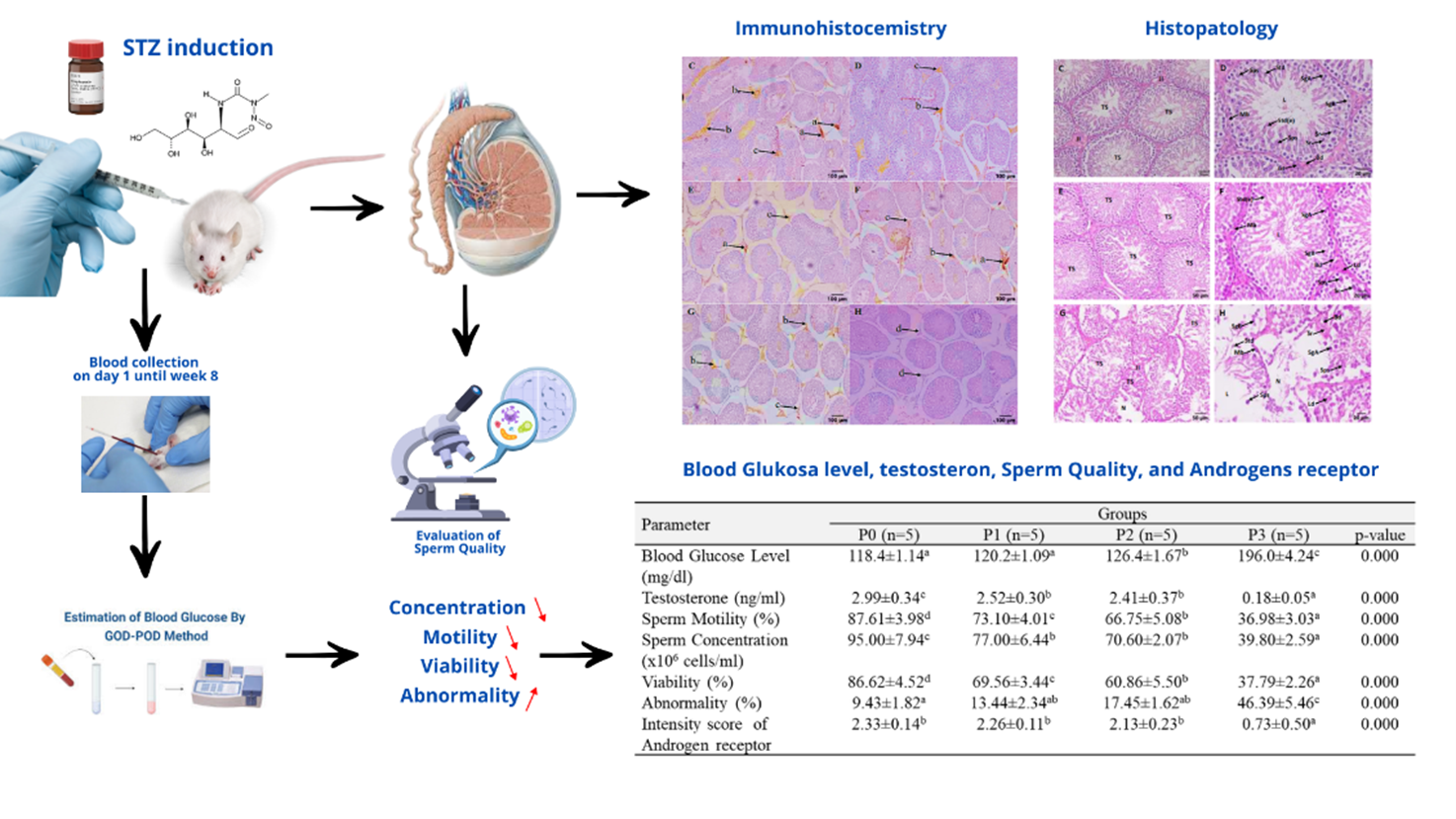
The growing prevalence of type 2 diabetes mellitus (T2DM) has raised significant concerns about its detrimental effects on male reproductive health. Streptozotocin (STZ)-induced diabetic models, particularly when combined with a high-fat diet (HFD), effectively replicate key features of human T2DM, including hyperglycemia and subsequent reproductive dysfunction. This study investigates the impact of STZ with various doses in inducing diabetes on testosterone levels, androgen receptor (AR) expression, and semen quality in male Sprague Dawley rats. Twenty rats, aged approximately 2 months and weighing 200 - 250 g, were divided into 4 groups (n = 5). Each group received a single intraperitoneal injection of STZ at doses of 0 mg/kg BW (P0), 20 mg/kg BW (P1), 30 mg/kg BW (P2), and 40 mg/kg BW (P3), using 0.1 M citrate buffer (pH 4.5) as the solvent. On day 56, blood glucose levels (BGL) were significantly elevated (p < 0.05) in all treated groups, particularly in P3 (average BGL 196.0 ± 4.24 mg/dL), indicating successful DM induction. Testosterone concentration in P3 was significantly lower (0.18 ± 0.05 ng/dL) compared to other groups (p < 0.05). Additionally, sperm concentration, motility, and viability in P3 showed a significant decrease (p = 0.00), while sperm abnormalities significantly increased (p = 0.00). AR intensity score (IS) was markedly reduced in P3 compared to other groups (p < 0.01). Histopathological examination revealed necrosis and lysis in seminiferous tubules, interstitial tissue damage, and decreased quantity and quality of spermatogenic cells. These findings confirm that STZ at 40 mg/kg BW effectively induces hyperglycemia and causes significant disruptions in male reproductive parameters, including hormonal profiles, sperm characteristics, and AR expression. This outcome offers a reliable method for studying the reproductive consequences of DM and may serve as a platform for evaluating potential therapeutic strategies to mitigate DM-related infertility.
HIGHLIGHTS
- This study demonstrates that STZ-induced hyperglycemia significantly reduces testosterone levels and androgen receptor expression in male Sprague Dawley rats.
- Elevated BGLs were strongly correlated with decreased sperm concentration, motility, and viability, and increased sperm abnormalities.
- Histopathological analysis revealed extensive testicular damage, including seminiferous tubule disruption and reduced Leydig and Sertoli cell populations.
- IHC findings confirmed downregulation of AR expression in diabetic rats, supporting the role of oxidative stress in reproductive dysfunction.
- The model provides a reliable platform to investigate the pathophysiology of diabetes-induced male infertility and evaluate potential therapeutic interventions.
GRAPHICAL ABSTRACT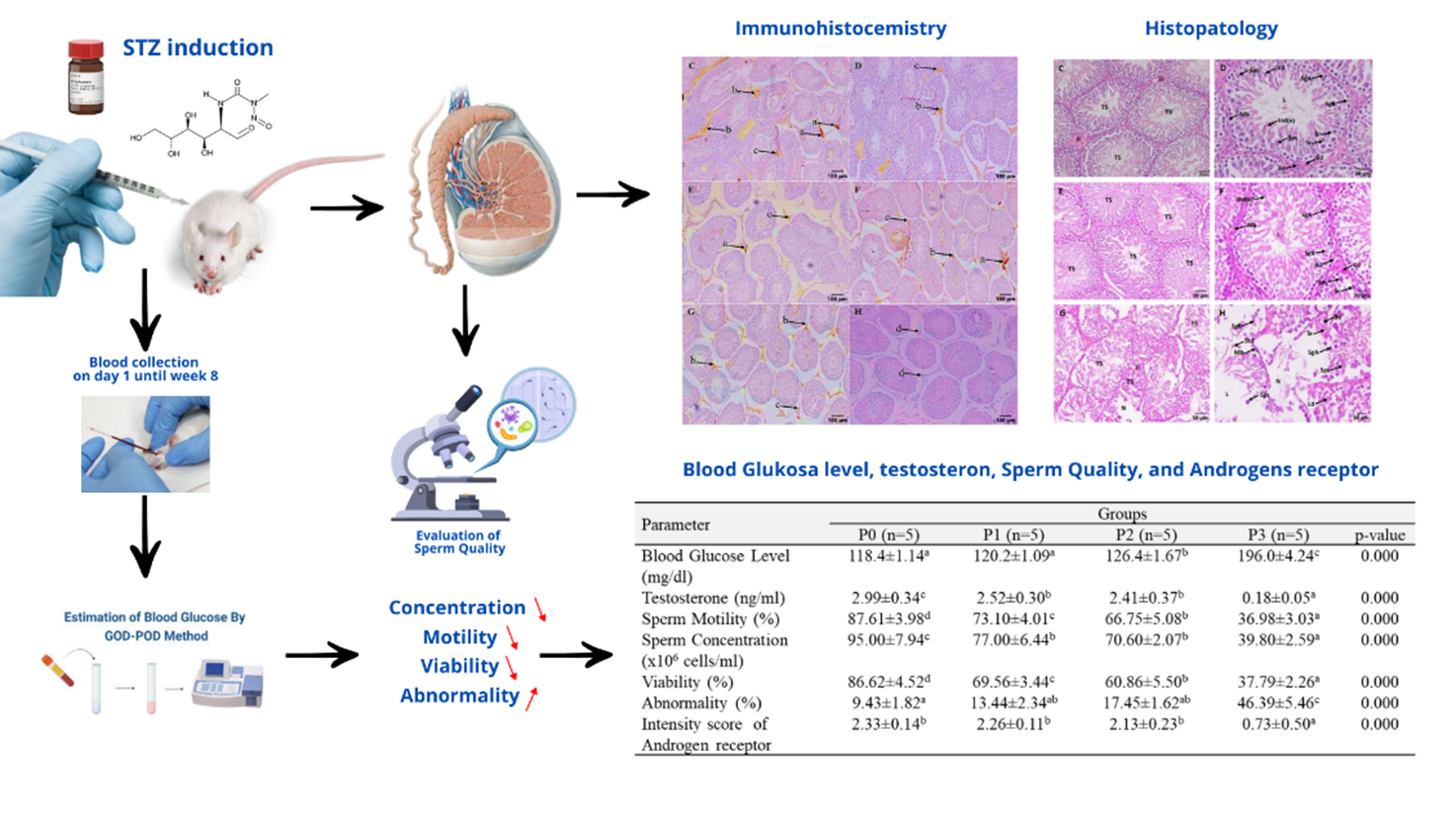
Downloads
References
ME Jasim and NE Abdul-Razzaq. A comparative study between metformin and insulin on biochemical variables in diabetic patients. Endocrine and Metabolic Science 2024; 16, 100206.
SA Antar, NA Ashour, M Sharaky, M Khattab, NA Ashour, RT Zaid, EJ Roh, A Elkamhawy and AA Al-Karmalawy. Diabetes mellitus: Classification, mediators, and complications; A gate to identify potential targets for the development of new effective treatments. Biomedicine and Pharmacotherapy 2023; 168, 115734.
P Saeedi, I Petersohn, P Salpea, B Malanda, S Karuranga, N Unwin, S Colagiuri, L Guariguata, AA Motala, K Ogurtsova, JE Shaw, D Bright and R Williams. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Research and Clinical Practice 2019; 157, 107843.
J Wang, B Bao, J Feng, Q Zhao, H Dai, F Meng, S Deng, B Wang and H Li. Effects of diabetes mellitus on sperm quality in the Db/Db mouse model and the role of the FoxO1 pathway. Medical Science Monitor 2021; 27, e928232.
A Papadopoulou, P Karkalousos and M Trapali. Effects of diabetes mellitus upon sperm quality insight into molecular level. Journal of Diabetes Mellitus 2022; 12(2), 75-86.
A Matuszewska, K Kowalski, P Jawień, T Tomkalski, D Gaweł-Dąbrowska, A Merwid-Ląd, E Szeląg, K Błaszczak, B Wiatrak, M Danielewski, J Piasny and A Szeląg. The hypothalamic-pituitary-gonadal axis in men with schizophrenia. International Journal of Molecular Sciences 2023; 24(7), 6492.
M Dai, W Guo, S Zhu, G Gong, M Chen, Z Zhong, J Guo and Y Zhang. Type 2 diabetes mellitus and the risk of abnormal spermatozoa: A Mendelian randomization study. Frontiers in Endocrinology 2022; 13, 1035338.
H Husnurrizal, S Wahyuni, Hafizuddin, CN Thasmi, MR Ramadhan, MM Ahsan and TN Siregar. Testosterone concentration, libido and spermatozoa quality in gembrong goats after in vivo and in vitro PGF2α administration. American Journal of Animal and Veterinary Sciences 2024; 19(2), 142-149.
F Lotti and M Maggi. Effects of diabetes mellitus on sperm quality and fertility outcomes: Clinical evidence. Andrology 2023; 11(2), 399-416.
NY Edelsztein and RA Rey. Importance of the androgen receptor signaling in gene transactivation and transrepression for pubertal maturation of the testis. Cells 2019; 8(8), 861.
Y Li, B Yao, J Men, Y Pang, J Gao, Y Bai, H Wang, J Zhang, L Zhao, X Xu, J Dong, C Li and R Peng. Oxidative stress and energy metabolism in male reproductive damage from single and combined high-power microwave exposure at 1.5 and 4.3GHz. Reproductive Toxicology 2025; 132, 108759.
A Fernández-Guasti, BG Quintanar, R Reyes, A Hernández, R Chavira and CE Roselli. Androgen receptors immunoreactivity in the rat brain of males with same-sex preference. Hormones and Behavior 2022; 146, 105279.
BL Furman. Streptozotocin-induced diabetic models in mice and rats. Current Protocols in Pharmacology 2021; 1(4), e78.
A Shokri, B Pourheydar, G Hossein Farjah, M Krimipour and M Pourheydar. Effects of glibenclamide and troxerutin on the sperm parameters and histopathological changes of testis in streptozotocin-induced diabetic male rats: An experimental study. International Journal of Reproductive Biomedicine 2023; 21(2), 123-138.
B Hitchen, K Norwood, VA Gault and JC Leslie. Behavioural evaluation of mouse models of type 2 diabetes. Learning and Motivation 2021; 74, 101730.
F Husna, FD Suyatna, W Arozal and EH Purwaningsih. Animal models in diabetes research (in Indonesian). Pharmaceutical Sciences and Research 2019; 6(3), 131-141.
Baharuddin and R Ikawaty. Bibliometric analysis: The use of Streptozotocin (STZ) In diabetic animal model. Journal of Documentation and Information Science 2023; 7(2), 83-91.
Herlina, A Amriani, I Solihah, P Damayanti and SW Rais. Effectiveness of ethanolic extract ketepeng cina leaves (Senna alata L.) as antidiabetic activity test in male wistar rats induced by alloxan. In: Proceedings of the Sriwijaya International Conference on Basic and Applied Science, Palembang, Indonesia. 2019, p. 12085.
SF Pasaribu, B Wiboworini and LR Kartikasari. Effect of germinated black rice krisna extract on fasting blood glucose and body weight in diabetes mellitus rats. International Journal of Nutrition Sciences 2021; 6(4), 194-200.
G Gholib, S Wahyuni, OH Kadar, M Adam, TM Lubis, A Azhar, M Akmal, TN Siregar, T Armansyah and TP Nugraha. Measurement of serum testosterone in kacang goat byusing enzyme-linked immunosorbent assay (ELISA) technique: The importance of kit validation (in Indonesian). Jurnal Kedokteran Hewan 2016; 10(1), 32-36.
C Dunn, D Brettle, M Cockroft, E Keating, C Revie and D Treanor. Quantitative assessment of H&E staining for pathology: Development and clinical evaluation of a novel system. Diagnostic Pathology 2024; 19(1), 42.
LKB Mudduwa. Quick score of hormone receptor status of breast carcinoma: Correlation with the other clinicopathological prognostic parameters. Indian Journal of Pathology and Microbioligy 2009; 52(2), 159-163.
A Ågmo. Androgen receptors and sociosexual behaviors in mammals: The limits of generalization. Neuroscience and Biobehavioral Reviews 2024; 157, 105530.
H Rehman, K Ullah, A Rasool, R Manzoor, Y Yuan, AM Tareen, I Kaleem, N Riaz, S Hameed and S Bashir. Comparative impact of streptozotocin on altering normal glucose homeostasis in diabetic rats compared to normoglycemic rats. Scientific Reports 2023; 13(1), 7921.
A Ghasemi and S Jeddi. Streptozotocin as a tool for induction of rat models of diabetes: A practical guide. EXCLI Journal 2023; 22, 274-294.
I Fajarwati, DD Solihin, T Wresdiyati and I Batubara. Self-recovery in diabetic Sprague Dawley rats induced by intraperitoneal alloxan and streptozotocin. Heliyon 2023; 9(5), e15533.
DAD Magalhães, WT Kume, FS Correia, TS Queiroz, EW Allebrandt, MPD Santos, NH Kawashita and SAD França. High-fat diet and streptozotocin in the induction of type 2 diabetes mellitus: A new proposal. Anais da Academia Brasileira de Ciências 2019; 91(1), e20180314.
TD Alabi, CD Villiers, SSD Plessis, TK Monsees, NL Brooks and OO Oguntibeju. The beneficial role of Anchomanes difformis in STZ-induced reproductive dysfunction in male Wistar rats. Diabetes, Metabolic Syndrome and Obesity 2020; 13, 4543-4560.
KC Racine, L Iglesias-Carres, JA Herring, KL Wieland, PN Ellsworth, JS Tessem, MG Ferruzzi, CD Kay and AP Neilson. The high-fat diet and low-dose streptozotocin type-2 diabetes model induces hyperinsulinemia and insulin resistance in male but not female C57BL/6J mice. Nutrition Research 2024; 131, 135-146.
GA Kanakis, CP Tsametis and DG Goulis. Measuring testosterone in women and men. Maturitas 2019; 125, 41-44.
N Kumari, A Khan, U Shaikh, K Lobes, D Kumar, F Suman, NS Bhutto, F Anees, S Shahid and A Rizwan. Comparison of testosterone levels in patients with and without type 2 diabetes. Cureus 2021; 13(7), e16288.
SR Brodjonegoro, T Ghinorawa, NKR Wonatorey, A Zulfiqqar and DS Heriyanto. Effect of hyperglycemia on fertility in streptozotocin-induced diabetic male Wistar rats: Focus on glucose transporters and oxidative stress. Medical Journal of Indonesia 2021; 30(3), 177-181.
S Roychoudhury, S Chakraborty, AP Choudhury, A Das, NK Jha, P Slama, M Nath, P Massanyi, J Ruokolainen and KK Kesari. Environmental factors-induced oxidative stress: Hormonal and molecular pathway disruptions in hypogonadism and erectile dysfunction. Antioxidants 2021; 10(6), 837.
A Caturano, M D’Angelo, A Mormone, V Russo, MP Mollica, T Salvatore, R Galiero, L Rinaldi, E Vetrano, R Marfella, M Monda, A Giordano and FC Sasso. Oxidative stress in type 2 diabetes: Impacts from pathogenesis to lifestyle modifications. Current Issues in Molecular Biology 2023; 45(8), 6651-6666.
M Naghibi, H Tayefi Nasrabadi, J Soleimani Rad, A Garjani, MSG Farashah and D Mohammadnejad. Forskolin improves male reproductive complications caused by hyperglycemia in type 2 diabetic rats. International Journal of Fertility and Sterility 2023; 17(4), 268-275.
TS Omolaoye and SSD Plessis. The effect of streptozotocin induced diabetes on sperm function: A closer look at AGEs, RAGEs, MAPKs and activation of the apoptotic pathway. Toxicological Research 2021; 37(1), 35-46.
K Fu, C Gao, X Li, H Zhang and B Xue. 3-chloro-1,2-propanediol induces oxidative stress and promotes testicular damage and infertility in rats through CYP2C9. Reproductive Toxicology 2024; 128, 108633.
A Sampannang, S Arun, J Burawat, W Sukhorum and S Iamsaard. Comparison of male reproductive parameters in mice with type 1 and type 2 diabetes. Clinical and Experimental Reproductive Medicine 2020; 47(1), 20-33.
D Julia, S Salni and S Nita. Pengaruh ekstrak bunga kembang sepatu (Hibiscus rosa-sinensis Linn.) terhadap jumlah, motilitas, morfologi, vabilitas spermatozoa tikus jantan (Rattus norvegicus) (in Indonesian). Biomedical Journal of Indonesia: Jurnal Biomedik Fakultas Kedokteran Universitas Sriwijaya 2019; 5(1), 34-42.
TS Omolaoye and SSD Plessis. Male infertility: A proximate look at the advanced glycation end products. Reproductive Toxicology 2020; 93, 169-177.
F Benko, M Chomová, O Uličná and E Tvrdá. ZDF rats: A suitable model to study male reproductive dysfunction in diabetes mellitus type 2 patients. In: Animal models in medicine and biology. IntechOpen, London, 2020.
SO Olojede, SK Lawal, OS Aladeyelu, KS Olaniyi, R Moodley, CO Rennie, EC Naidu and OO Azu. Studies on testicular ultrastructural and hormonal changes in type-2 diabetic rats treated with highly active antiretroviral therapy conjugated silver nanoparticles. Life Sciences 2022; 298, 120498.
OO Ojo and OO Olorunsogo. Quercetin and vitamin E attenuate diabetes-induced testicular anomaly in Wistar rats via the mitochondrial-mediated apoptotic pathway. Andrologia 2021; 53(10), e14185.
L O’Donnell, LB Smith and D Rebourcet. Sertoli cells as key drivers of testis function. Seminars in Cell and Developmental Biology 2022; 121, 2-9.
D Luo, X Qi, X Xu, L Yang, C Yu and Q Guan. Involvement of p38 MAPK in Leydig cell aging and age-related decline in testosterone. Frontiers in Endocrinology 2023; 14, 1088249.
FD Ni, SL Hao and WX Yang. Multiple signaling pathways in Sertoli cells: Recent findings in spermatogenesis. Cell Death and Disease 2019; 10, 541.
A Nisa and W Abdullah. Impact of glycation and diabetes on sperm DNA integrity that leads to infertility in male. International Journal of Novel Research and Development 2023; 8(4), a185-a193.
OC Badejogbin, OE Chijioke-Agu, MV Olubiyi and MO Agunloye. Pathogenesis of testicular dysfunction in diabetes: Exploring the mechanism and therapeutic interventions. Journal of Assisted Reproduction and Genetics 2025; 42(2), 367-379.
Z He, G Yin, QQ Li, Q Zeng and J Duan. Diabetes mellitus causes male reproductive dysfunction: A review of the evidence and mechanisms. In Vivo 2021; 35(5), 2503-2511.
GA Soliman, AS Saeedan, RF Abdel-Rahman, HA Ogaly, RM Abd-Elsalam and MS Abdel-Kader. Olive leaves extract attenuates type II diabetes mellitus-induced testicular damage in rats: Molecular and biochemical study. Saudi Pharmaceutical Journal 2019; 27(3), 326-340.
SB Meroni, MN Galardo, G Rindone, A Gorga, MF Riera and SB Cigorraga. Molecular mechanisms and signaling pathways involved in sertoli cell proliferation. Frontiers in Endocrinology 2019; 10, 224.
L Moutard, C Goudin, C Jaeger, C Duparc, E Louiset, T Pereira, F Fraissinet, M Delessard, J Saulnier, A Rives-Feraille, C Delalande, H Lefebvre, N Rives, L Dumont and C Rondanino. Steroidogenesis and androgen/estrogen signaling pathways are altered in in vitro matured testicular tissues of prepubertal mice. eLife 2023; 12, RP85562.
W Shah, R Khan, B Shah, A Khan, S Dil, W Liu, J Wen and X Jiang. The molecular mechanism of sex hormones on sertoli cell development and proliferation. Frontiers in Endocrinology 2021; 12, 648141.
JZ AlTamimi, NA AlFaris, GM Alshammari, RI Alagal, DH Aljabryn and MA Yahya. Esculeoside A alleviates reproductive toxicity in streptozotocin-diabetic rats’ s model by activating Nrf2 signaling. Saudi Journal of Biological Sciences 2023; 30(9), 103780.
SJ Gilani, MN Bin-Jumah, FA Al-Abbasi, MS Nadeem, M Afzal, N Sayyed and I Kazmi. Fustin ameliorates hyperglycemia in streptozotocin induced type-2 diabetes via modulating glutathione/Superoxide dismutase/Catalase expressions, suppress lipid peroxidation and regulates histopathological changes. Saudi Journal of Biological Sciences 2021; 28(12), 6963-6971.
MR Al‐Shathly, SS Ali and NN Ayuob. Zingiber officinale preserves testicular structure and the expression of androgen receptors and proliferating cell nuclear antigen in diabetic rats. Andrologia 2020; 52(3), e13528.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






