Challenges and Strategies in Nanoparticle-Mediated Drug Release: Mechanisms and Future Directions
DOI:
https://doi.org/10.48048/tis.2025.10344Keywords:
Nanoparticle, Drug Release, Nanoparticle Design-Engineering, Stimuli-Responsive Nanoparticle, Hybrid Nanoparticle System, Nanoparticle, Drug release, Nanoparticle design-engineering, Stimuli-responsive nanoparticle, Hybrid nanoparticle systemAbstract
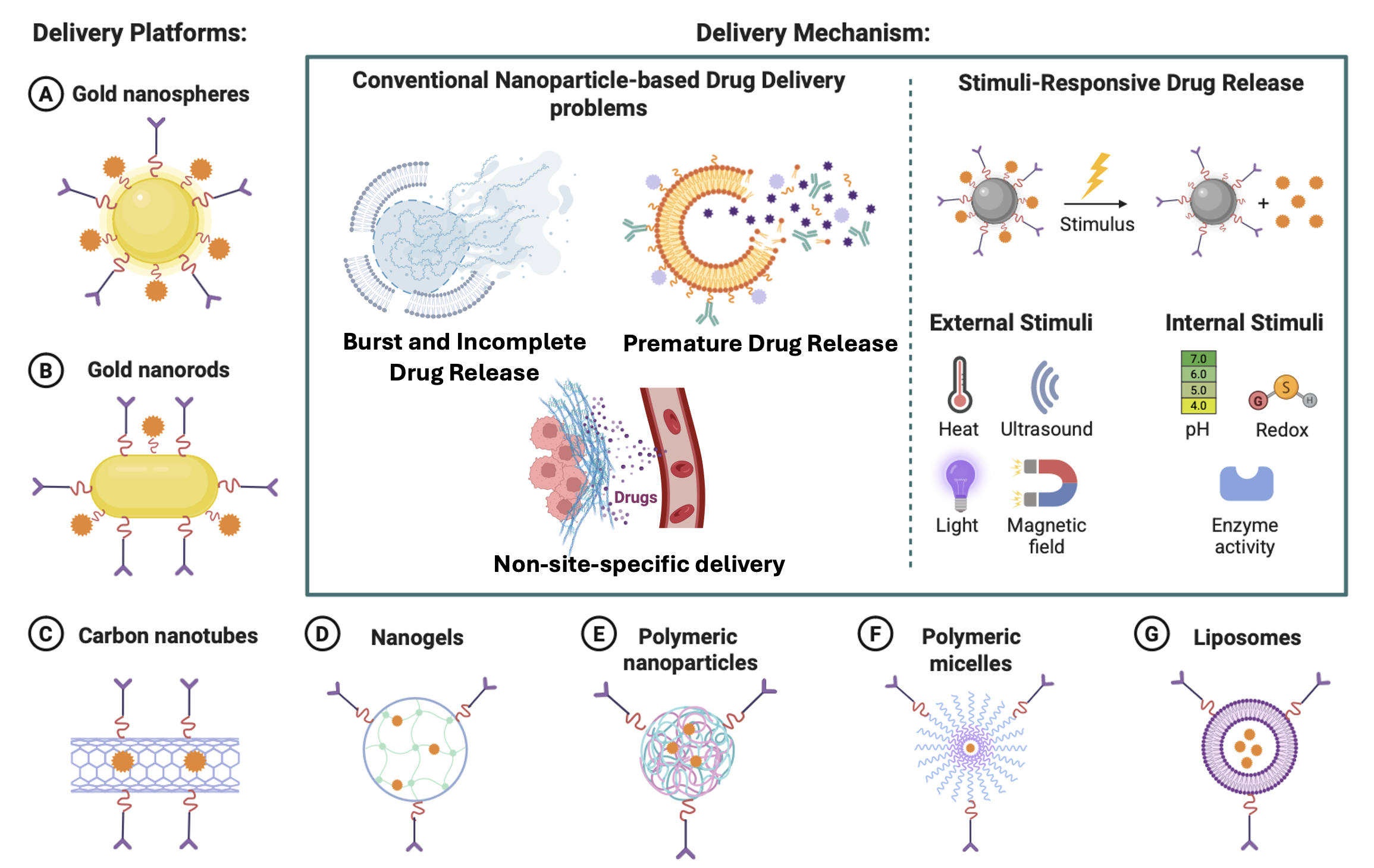
Nanoparticle drug delivery systems (NPDDS) promise to increase the efficacy and safety of therapeutic agents, yet achieving controlled and sustained release of active ingredients from these nanoparticles remains a significant challenge hindering the full realization of this technology’s benefits. This paper aims to uncover the key problems associated with nanoparticle drug release by delving into the fundamental concepts and mechanisms underlying this intricate process. Drug release mechanisms like diffusion, erosion, and stimuli-responsive release are intricately examined, while critically evaluating the obstacles posed by factors such as particle size, surface properties, drug-carrier interactions, and physiological barriers. Some of the effects of not achieving controlled and sustained release are burst release and incomplete release, as well as premature drug release and instability. Future directions and strategies for improved drug release are proposed, including nanoparticle design and engineering, stimuli-responsive nanoparticle, and hybrid nanoparticle systems. By synergistically addressing the key problem of nanoparticle drug release through this comprehensive approach, the full potential of this transformative technology can be unlocked, paving the way for more effective and safer therapeutic interventions, ultimately leading to improved patient outcomes and reduced healthcare costs.
HIGHLIGHTS
- Provide a comprehensive understanding of the critical challenge of nanoparticle drug release, encompassing the underlying principles, obstacles, and future prospects.
- Mechanisms of drug release from nanoparticles, including diffusion-controlled, degradation-controlled, and stimuli-responsive release processes.
- Critically analyze the challenges associated with achieving the desired drug release profiles, such as burst release, premature drug release, and uncontrollable release on targeted site.
- Explore innovative strategies and future directions for improving drug release from nanoparticles, including the design and engineering of novel nanoparticle architectures, the development of stimuli-responsive systems, the integration of combination and hybrid approaches, and the application of advanced characterization and modelling techniques.
GRAPHICAL ABSTRACT
Downloads
References
SAA Rizvi and AM Saleh. Applications of nanoparticle systems in drug delivery technology. Saudi Pharmaceutical Journal 2018; 26(1), 64-70.
JK Patra, G Das, LF Fraceto, EVR Campos, MP Rodriguez-Torres, LS Acosta-Torres, LA Diaz-Torres, R Grillo, MK Swamy and S Sharma. Solomon Habtemariam & Han-Seung Shin Nano based drug delivery systems: Recent developments and future prospects. Journal of Nanobiotechnology 2018; 16, 71.
DH Kim and DC Martin. Sustained release of dexamethasone from hydrophilic matrices using PLGA nanoparticles for neural drug delivery. Biomaterials 2006; 27(15), 3031-3037.
A Kumari, SK Yadav and SC Yadav. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids and Surfaces B: Biointerfaces 2010; 75(1), 1-18.
E Roblegg, E Fröhlich, C Meindl, B Teubl, M Zaversky and A Zimmer. Evaluation of a physiological in vitro system to study the transport of nanoparticles through the buccal mucosa. Nanotoxicology 2012; 6(4), 399-413.
CJM Rivas, M Tarhini, W Badri, K Miladi, H Greige-Gerges, QA Nazari, SAG Rodríguez, RÁ Román, H Fessi and A Elaissari. Nanoprecipitation process: From encapsulation to drug delivery. International Journal of Pharmaceutics 2017; 532(1), 66-81.
RA Siegel and MJ Rathbone. Overview of controlled release mechanisms. In: J Siepmann, R Siegel and M Rathbone (Eds.). Fundamentals and applications of controlled release drug delivery. Springer, Boston, United States, p. 19-43.
AK Bajpai, SK Shukla, S Bhanu and S Kankane. Responsive polymers in controlled drug delivery. Progress in Polymer Science 2008; 33(11), 1088-1118.
J Siepmann and F Siepmann. Modeling of diffusion controlled drug delivery. Journal of Controlled Release 2012; 161(2), 351-362.
A Fick. V. on liquid diffusion. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 1855; 10(63), 30-39.
J Crank. The mathematics of diffusion. 2nd ed. Oxford University Press, New York, 1979.
ML Bruschi. Main mechanisms to control the drug release. In: ML Bruschi (Ed.). Strategies to modify the drug release from pharmaceutical systems. Elsevier, Amsterdam, The Netherlands, 2015, p. 37-62.
S Freiberg and XX Zhu. Polymer microspheres for controlled drug release. International Journal of Pharmaceutics 2004; 282(1-2), 1-18.
T Higuchi. Mechanism of sustained‐action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. Journal of Pharmaceutical Sciences 1963; 52(12), 1145-1149.
J Siepmann and NA Peppas. Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Advanced Drug Delivery Reviews 2001; 64, 163-174.
N Kamaly, B Yameen, J Wu and OC Farokhzad. Degradable controlled-release polymers and polymeric nanoparticles: Mechanisms of controlling drug release. Chemical Reviews 2016; 116(4), 2602-2663.
A Sadžak, M Eraković and S Šegota. Kinetics of flavonoid degradation and controlled release from functionalized magnetic nanoparticles. Molecular Pharmaceutics 2023; 20(10), 5148-5159.
JH Lee and Y Yeo. Controlled drug release from pharmaceutical nanocarriers. Chemical Engineering Science 2015; 125, 75-84.
AI Visan, G Popescu-Pelin and G Socol. Degradation behavior of polymers used as coating materials for drug delivery - a basic review. Polymers 2021; 13(8), 1272.
N Islam, I Dmour and MO Taha. Degradability of chitosan micro/nanoparticles for pulmonary drug delivery. Heliyon 2019; 5(5), e01684.
F Von Burkersroda, L Schedl and A Göpferich. Why degradable polymers undergo surface erosion or bulk erosion. Biomaterials 2002; 23(21), 4221-4231.
JC Middleton and AJ Tipton. Synthetic biodegradable polymers as orthopedic devices. Biomaterials 2000; 21(23), 2335-2346.
WC Lee and IM Chu. Preparation and degradation behavior of polyanhydrides nanoparticles. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2008; 84(1), 138-146.
YX Weng, XL Wang and YZ Wang. Biodegradation behavior of PHAs with different chemical structures under controlled composting conditions. Polymer Testing 2011; 30(4), 372-380.
YZ Zhang, LY Ran, CY Li and XL Chen. Diversity, structures, and collagen-degrading mechanisms of bacterial collagenolytic proteases. Applied and Environmental Microbiology 2015; 81(18), 6098-6107.
J Yan, S Ai, F Yang, K Zhang and Y Huang. Study on mechanism of chitosan degradation with hydrodynamic cavitation. Ultrasonics Sonochemistry 2020; 64, 105046.
D Bendix. Chemical synthesis of polylactide and its copolymers for medical applications. Polymer Degradation and Stability 1998; 59(1-3), 129-135.
S Liu, J Yu, H Li, K Wang, G Wu, B Wang, M Liu, Y Zhang, P Wang, J Zhang, J Wu, Y Jing, F Li and M Zhang. Controllable drug release behavior of polylactic acid (PLA) surgical suture coating with ciprofloxacin (CPFX)-polycaprolactone (PCL)/ polyglycolide (PGA). Polymers 2020; 12(2), 288.
Z Cui, Y Peng, K Li, J Peng, H Zhao, LS Turng and C Shen. The degradation rate of polyanhydride (polysebacic acid), diacetoxy terminated, PSADT). Journal Wuhan University of Technology, Materials Science Edition 2013; 28(4), 793-797.
S Rana, J Singh, A Wadhawan, A Khanna, G Singh and M Chatterjee. Evaluation of in vivo toxicity of novel biosurfactant from Candida parapsilosis loaded in PLA-PEG polymeric nanoparticles. Journal of Pharmaceutical Sciences 2021; 110(4), 1727-1738.
JM Metselaar, P Bruin, LWT de Boer, T de Vringer, C Snel, C Oussoren, MHM Wauben, DJA Crommelin, G Storm and WE Hennink. A novel family of l-amino acid-based biodegradable polymer-lipid conjugates for the development of long-circulating liposomes with effective drug-targeting capacity. Bioconjugate Chemistry 2003; 14(6), 1156-1164.
S Mura, J Nicolas and P Couvreur. Stimuli-responsive nanocarriers for drug delivery. Nature Materials 2013; 12(11), 991-1003.
M Karimi, A Ghasemi, PS Zangabad, R Rahighi, SMM Basri, H Mirshekari, M Amiri, ZS Pishabad, A Aslani, M Bozorgomid, D Ghosh, A Beyzavi, A Vaseghi, AR Aref, L Haghani, S Bahramia and MR Hamblin. Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Royal Society of Chemistry 2016; 45(5), 1457-1501.
R Mo, T Jiang, R Disanto, W Tai and Z Gu. ATP-triggered anticancer drug delivery. Nature Communications 2014; 5(1), 3364.
AG Arranja, V Pathak, T Lammers and Y Shi. Tumor-targeted nanomedicines for cancer theranostics. Pharmacological Research 2017; 115, 87-95.
T Wang, MD Wang, C Di Ding and JJ Fu. Mono-benzimidazole functionalized β-cyclodextrins as supramolecular nanovalves for pH-triggered release of p-coumaric acid. Chemical Communications 2014; 50(83), 12469-12472.
S Wu, X Huang and X Du. pH- and redox-triggered synergistic controlled release of a ZnO-gated hollow mesoporous silica drug delivery system. Journal of Materials Chemistry B 2015; 3(7), 1426-1432.
T Wang, GP Sun, MD Wang, BJ Zhou and JJ Fu. Voltage/pH-driven mechanized silica nanoparticles for the multimodal controlled release of drugs. ACS Applied Materials & Interfaces 2015; 7(38), 21295-21304.
CE Ashley, EC Carnes, KE Epler, DP Padilla, GK Phillips, RE Castillo, DC Wilkinson, BS Wilkinson, CA Burgard, RM Kalinich, JL Townson, B Chackerian, CL Willman, DS Peabod, W Wharton and J Brinker. Delivery of small interfering RNA by peptide-targeted mesoporous silica nanoparticle-supported lipid bilayers. ACS Nano 2012; 6(3), 2174-2188.
Q Sun, X Ma, B Zhang, Z Zhou, E Jin, Y Shen, EA Van Kirk, WJ Murdoch, M Radosz and W Sun. Fabrication of dendrimer-releasing lipidic nanoassembly for cancer drug delivery. Biomaterials Science 2016; 4(6), 958-969.
W Li, L Huang, X Ying, Y Jian, Y Hong, F Hu and Y Du. Antitumor drug delivery modulated by a polymeric micelle with an upper critical solution temperature. Angewandte Chemie - International Edition 2015; 54(10), 3126-3131.
MK Park, S Jun, I Kim, SM Jin, JG Kim, TJ Shin and E Lee. Stepwise drug-release behavior of onion-like vesicles generated from emulsification-induced assembly of semicrystalline polymer amphiphiles. Advanced Functional Materials 2015; 25(29), 4570-4579.
S Karthik, B Saha, SK Ghosh and NDP Singh. Photoresponsive quinoline tethered fluorescent carbon dots for regulated anticancer drug delivery. Chemical Communications 2013; 49(89), 10471-10473.
J Liu, C Wang, X Wang, X Wang, L Cheng, Y Li and Z Liu. Mesoporous silica coated single-walled carbon nanotubes as a multifunctional light-responsive platform for cancer combination therapy. Advanced Functional Materials 2015; 25(3), 384-392.
D Maciel, P Figueira, S Xiao, D Hu, X Shi, J Rodrigues, H Tomás and Y Li. Redox-responsive alginate nanogels with enhanced anticancer cytotoxicity. Biomacromolecules 2013; 14(9), 3140-3146.
AA Noyes, RW Willis and BA Arthcr. The rate of solution of solid substances in their own solutions. Journal of the American Chemical Society 1897; 19(12), 930-934.
W Ostwald. Über die vermeintliche Isomerie des roten und gelben Quecksilberoxyds und die Oberflächenspannung fester Körper (in German). Zeitschrift für physikalische Chemie 1900; 34(1), 495-503.
M Hasan, K Elkhoury, CJF Kahn, E Arab-Tehrany and M Linder. Preparation, characterization, and release kinetics of chitosan-coated nanoliposomes encapsulating curcumin in simulated environments. Molecules 2019; 24(10), 2023.
AL Cartaxo, AR Costa‐Pinto, A Martins, S Faria, VMF Gonçalves, ME Tiritan, H Ferreira and NM Neves. Influence of PDLA nanoparticles size on drug release and interaction with cells. Journal of Biomedical Materials Research Part A 2019; 107(3), 482-493.
MB Sedelnikova, EG Komarova, YP Sharkeev, VV Chebodaeva, TV Tolkacheva, AM Kondranova, AM Zakharenko and OV Bakina. Effect of the porosity, roughness, wettability, and charge of micro-arc coatings on the efficiency of doxorubicin delivery and suppression of cancer cells. Coatings 2020; 10(7), 664.
J Xue, Y Zhu, S Bai, C He, G Du, Y Zhang, Y Zhong, W Chen, H Wang and X Sun. Nanoparticles with rough surface improve the therapeutic effect of photothermal immunotherapy against melanoma. Acta Pharmaceutica Sinica B 2022; 12(6), 2934-2949.
C Piotto, SP Pujari, H Zuilhof and P Bettotti. Surface heterogeneous nucleation-mediated release of beta-carotene from porous silicon. Nanomaterials 2020; 10(9), 1659.
LK Chiu, WJ Chiu and YL Cheng. Effects of polymer degradation on drug release a mechanistic study of morphology and transport properties in 50:50 poly(d/-lactide-co-glycolide). International Journal of Pharmaceutics 1995; 126(1-2), 169-178.
A Albanese and WCW Chan. Effect of gold nanoparticle aggregation on cell uptake and toxicity. ACS Nano 2011; 5(7), 5478-5489.
S Sant, V Nadeau and P Hildgen. Effect of porosity on the release kinetics of propafenone-loaded PEG-g-PLA nanoparticles. Journal of Controlled Release 2005; 107(2), 203-214.
H Xu, X Xu, S Li, WL Song, DG Yu and SW Annie Bligh. The effect of drug heterogeneous distributions within core-sheath nanostructures on its sustained release profiles. Biomolecules 2021; 11(9), 1330.
B Mandal, H Bhattacharjee, N Mittal, H Sah, P Balabathula, LA Thoma and GC Wood. Core-shell-type lipid-polymer hybrid nanoparticles as a drug delivery platform. Nanomedicine: Nanotechnology, Biology and Medicine 2013; 9(4), 474-491.
Y Zare. Study of nanoparticles aggregation/agglomeration in polymer particulate nanocomposites by mechanical properties. Composites Part A: Applied Science and Manufacturing 2016; 84, 158-164.
GW Gibbs. Collected works. Thermodynamics. Longmans, London, 1928.
R Toy, PM Peiris, KB Ghaghada and E Karathanasis. Shaping cancer nanomedicine: The effect of particle shape on the in vivo journey of nanoparticles. Nanomedicine 2014; 9(1), 121-134.
M Kaplan, K Öztürk, SC Öztürk, E Tavukçuoğlu, G Esendağlı and S Calis. Effects of particle geometry for PLGA-based nanoparticles: Preparation and in vitro/in vivo evaluation. Pharmaceutics 2023; 15(1), 175.
N Zhang, J Li, W Jiang, C Ren, J Li, J Xin and K Li. Effective protection and controlled release of insulin by cationic β-cyclodextrin polymers from alginate/chitosan nanoparticles. International Journal of Pharmaceutics 2010; 393(1-2), 213-219.
J Qu, S Peng, R Wang, S Tao Yang, Q Han Zhou and J Lin. Stepwise pH-sensitive and biodegradable polypeptide hybrid micelles for enhanced cellular internalization and efficient nuclear drug delivery. Colloids and Surfaces B: Biointerfaces 2019; 181, 315-324.
V Sanna, AM Roggio, S Siliani, M Piccinini, S Marceddu, A Mariani and M Sechi. Development of novel cationic chitosan- and anionic alginate–coated poly(D,L-lactide-co-glycolide) nanoparticles for controlled release and light protection of resveratrol. International Journal of Nanomedicine 2012; 7, 5501-5516.
MM El-Hammadi and JL Arias. Recent advances in the surface functionalization of PLGA-based nanomedicines. Nanomaterials 2022; 12(3), 354.
C Di Natale, V Onesto, E Lagreca, R Vecchione and PA Netti. Tunable release of Curcumin with an in silico-supported approach from mixtures of highly porous PLGA microparticles. Materials 2020; 13(8), 1807.
H Liu and J He. Simultaneous release of hydrophilic and hydrophobic drugs from modified chitosan nanoparticles. Materials Letters 2015; 161, 415-418.
PSR Naidu, M Norret, SA Dunlop, M Fitzgerald, TD Clemons and KS Iyer. Novel hydrophilic copolymer-based nanoparticle enhances the therapeutic efficiency of doxorubicin in cultured MCF-7 cells. ACS Omega 2019; 4(17), 17083-17089.
CE Mora-Huertas, H Fessi and A Elaissari. Polymer-based nanocapsules for drug delivery. International Journal of Pharmaceutics 2010; 385(1-2), 113-142.
A Jain, K Thakur, P Kush and UK Jain. Docetaxel loaded chitosan nanoparticles: Formulation, characterization and cytotoxicity studies. International Journal of Biological Macromolecules 2014; 69, 546-553.
AA D’souza and R Shegokar. Polyethylene glycol (PEG): A versatile polymer for pharmaceutical applications. Expert Opinion on Drug Delivery 2016; 13(9), 1257-1275.
X Huang and CS Brazel. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. Journal of Controlled Release 2001; 73(2-3), 121-136.
JV Natarajan, A Darwitan, VA Barathi, M Ang, HM Htoon, F Boey, KC Tam, TT Wong and SS Venkatraman. Sustained drug release in nanomedicine: A long-acting nanocarrier-based formulation for glaucoma. ACS Nano 2014; 8(1), 419-429.
SK Sahoo, AK Panda and V Labhasetwar. Characterization of porous PLGA/PLA microparticles as a scaffold for 3 dimensional growth of breast cancer cells. Biomacromolecules 2005; 6(2), 1132-1139.
RD Vaishya, V Khurana, S Patel and AK Mitra. Controlled ocular drug delivery with nanomicelles. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2014; 6(5), 422-437.
J Chang, L Mo, J Song, X Wang, H Liu, C Meng and Y Wu. A pH-responsive mesoporous silica nanoparticle-based drug delivery system for targeted breast cancer therapy. Journal of Materials Chemistry B 2022; 10(17), 3375-3385.
R Langer and NA Peppas. Advances in biomaterials, drug delivery, and bionanotechnology. AIChE Journal 2003; 49(12), 2990-3006.
Q Ren, Z Liang, X Jiang, P Gong, L Zhou, Z Sun, J Xiang, Z Xu, X Peng, S Li, W Li, L Cai and J Tang. Enzyme and pH dual-responsive hyaluronic acid nanoparticles mediated combination of photodynamic therapy and chemotherapy. International Journal of Biological Macromolecules 2019; 130, 845-852.
Y Tahara and K Akiyoshi. Current advances in self-assembled nanogel delivery systems for immunotherapy. Advanced Drug Delivery Reviews 2015; 95, 65-76.
S Hajebi, A Abdollahi, H Roghani-Mamaqani and M Salami-Kalajahi. Temperature-Responsive Poly(N-Isopropylacrylamide) Nanogels: The role of hollow cavities and different shell cross-linking densities on doxorubicin loading and release. Langmuir 2020; 36(10), 2683-2694.
J Pardeike, A Hommoss and RH Müller. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. International Journal of Pharmaceutics 2009; 366(1-2), 170-184.
RH Müller, M Radtke and SA Wissing. Nanostructured lipid matrices for improved microencapsulation of drugs. International Journal of Pharmaceutics 2002; 242(1), 121-128.
G Fytianos, A Rahdar and GZ Kyzas. Nanomaterials in cosmetics: Recent updates. Nanomaterials 2020; 10(5), 979.
C Oliveira, C Coelho, JA Teixeira, P Ferreira-Santos and CM Botelho. Nanocarriers as active ingredients enhancers in the cosmetic industry - the european and north america regulation challenges. Molecules 2022; 27(5), 1669.
P Dong, FF Sahle, SB Lohan, S Saeidpour, S Albrecht, C Teutloff, R Bodmeier, M Unbehauen, C Wolff, R Haag, J Lademann, A Patzelt, M Schäfer-Korting and MC Meinke. pH-sensitive Eudragit® L 100 nanoparticles promote cutaneous penetration and drug release on the skin. Journal of Controlled Release 2019; 295, 214-222.
S Arora, JM Rajwade and KM Paknikar. Nanotoxicology and in vitro studies: The need of the hour. Toxicology and Applied Pharmacology 2012; 258(2), 151-165.
A Ladaycia, C Passirani and E Lepeltier. Microbiota and nanoparticles: Description and interactions. European Journal of Pharmaceutics and Biopharmaceutics 2021; 169, 220-240.
K AL-Smadi, VR Leite-Silva, NA Filho, PS Lopes and Y Mohammed. Innovative approaches for maintaining and enhancing skin health and managing skin diseases through microbiome-targeted strategies. Multidisciplinary Digital Publishing Institute 2023; 12(12), 1698.
MS Muthu, SA Kulkarni, J Xiong and SS Feng. Vitamin E TPGS coated liposomes enhanced cellular uptake and cytotoxicity of docetaxel in brain cancer cells. International Journal of Pharmaceutics 2011; 421(2), 332-340.
S Thakral, NK Thakral and DK Majumdar. Eudragit®: A technology evaluation. Expert Opinion on Drug Delivery 2013; 10(1), 131-149.
Ç Yücel, GŞ Karatoprak and Y Aktaş. Nanoliposomal resveratrol as a novel approach to treatment of diabetes mellitus. Journal of Nanoscience and Nanotechnology 2017; 18(6), 3856-3864.
C Chittasupho, T Thongnopkoon and P Kewsuwan. Surface modification of poly(D,L-lactic-co-glycolic acid) nanoparticles using sodium carboxymethyl cellulose as colloidal stabilizer. Current Drug Delivery 2016; 13(1), 95-104.
S Mitragotri, PA Burke and R Langer. Overcoming the challenges in administering biopharmaceuticals: Formulation and delivery strategies. Nature Reviews Drug Discovery 2014; 13(9), 655-672.
N Joy, D Venugopal and S Samavedi. Robust strategies to reduce burst and achieve tunable control over extended drug release from uniaxially electrospun composites. European Polymer Journal 2022; 168, 111102.
C Zheng and W Liang. A one-step modified method to reduce the burst initial release from PLGA microspheres. Drug Delivery 2010; 17(2), 77-82.
M Julinová, L Vaňharová and M Jurča. Water-soluble polymeric xenobiotics - Polyvinyl alcohol and polyvinylpyrrolidon - and potential solutions to environmental issues: A brief review. Journal of Environmental Management 2018; 228, 213-222.
S Bajaj, N Sakhuja and D Singla. Stability testing of pharmaceutical products. Journal of Applied Pharmaceutical Science 2012; 03, 129-138.
X Zhang, T Liang and Q Ma. Layer-by-layer assembled nano-drug delivery systems for cancer treatment. Drug Delivery 2021; 2021(1), 655-669.
VNSK Varma, HG Shivakumar, V Balamuralidhara, M Navya and U Hani. Development of pH sensitive nanoparticles for intestinal drug delivery using chemically modified guar gum co-polymer. Iranian Journal of Pharmaceutical Research 2016; 15(1), 83.
L Palanikumar, S Al-Hosani, M Kalmouni, VP Nguyen, L Ali, R Pasricha, FN Barrera and M Magzoub. pH-responsive high stability polymeric nanoparticles for targeted delivery of anticancer therapeutics. Communications Biology 2020; 3(1), 95.
AK Mohan, M Minsa, TR Santhosh Kumar and GS Vinod Kumar. Multi-layered PLGA-PEI nanoparticles functionalized with TKD peptide for targeted delivery of Pep5 to breast tumor cells and spheroids. International Journal of Nanomedicine 2022; 17, 5581-5600.
SW Morton, MJ Lee, ZJ Deng, EC Dreaden, E Siouve, KE Shopsowitz, NJ Shah, MB Yaffe and PT Hammond. A nanoparticle-based combination chemotherapy delivery system for enhanced tumor killing by dynamic rewiring of signaling pathways. Science Signaling 2014; 7(325), ra44.
H Yu, Z Yang, F Li, L Xu and Y Sun. Cell-mediated targeting drugs delivery systems. Drug Delivery 2020; 27(1), 1425-1437.
M Toledano, M Toledano-Osorio, MD Navarro-Hortal, A Varela-López, R Osorio and JL Quiles. Novel polymeric nanocarriers reduced zinc and doxycycline toxicity in the nematode Caenorhabditis elegans. Antioxidants 2019; 8(11), 550.
WM Pardridge. Drug transport across the blood-brain barrier. Journal of Cerebral Blood Flow & Metabolism 2012; 32(11), 1959-1972.
LM Ensign, R Cone and J Hanes. Oral drug delivery with polymeric nanoparticles: The gastrointestinal mucus barriers. Advanced Drug Delivery Reviews 2012; 64(6), 557-570.
RK Jain and T Stylianopoulos. Delivering nanomedicine to solid tumors. Nature Reviews Clinical Oncology 2010; 7(11), 653-664.
H Maeda. Toward a full understanding of the EPR effect in primary and metastatic tumors as well as issues related to its heterogeneity. Advanced Drug Delivery Reviews 2015; 91, 3-6.
N Bertrand, J Wu, X Xu, N Kamaly and OC Farokhzad. Cancer nanotechnology: The impact of passive and active targeting in the era of modern cancer biology. Advanced Drug Delivery Reviews 2014; 66, 2-25.
E Blanco, H Shen and M Ferrari. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nature Biotechnology 2015; 33(9), 941-951.
D Singh, Y Sharma, D Dheer and R Shankar. Stimuli responsiveness of recent biomacromolecular systems (concept to market): A review. International Journal of Biological Macromolecules 2024; 261, 129901.
D Pei and M Buyanova. Overcoming endosomal entrapment in drug delivery. Bioconjugate Chemistry 2019; 30(2), 273-283.
T Stylianopoulos and RK Jain. Combining 2 strategies to improve perfusion and drug delivery in solid tumors. Proceedings of the National Academy of Sciences 2013; 110(46), 18632-18637.
VC Niculescu. Mesoporous silica nanoparticles for bio-applications. Frontiers in Materials 2020; 7, 36.
H Meng, M Xue, T Xia, YL Zhao, F Tamanoi, JF Stoddart, JI Zink and AE Nel. Autonomous in vitro anticancer drug release from mesoporous silica nanoparticles by pH-sensitive nanovalves. Journal of the American Chemical Society 2010; 132(36), 12690-12697.
MN Koopaei, MR Khoshayand, SH Mostafavi, M Amini, MR Khorramizadeh, MJ Tehrani, F Atyabi and R Dinarvand. Docetaxel loaded PEG-PLGA nanoparticles: Optimized drug loading, in-vitro cytotoxicity and in-vivo antitumor effect. 2014; 13(3), 819-833.
JJ Richardson, M Björnmalm and F Caruso. Technology-driven layer-by-layer assembly of nanofilms. Science 2015; 348(6233), aaa2491.
SS Mandal, D Jose and AJ Bhattacharyya. Role of surface chemistry in modulating drug release kinetics in titania nanotubes. Materials Chemistry and Physics 2014; 147(1-2), 247-253.
S La Manna, C Di Natale, V Onesto and D Marasco. Self-assembling peptides: From design to biomedical applications. International Journal of Molecular Sciences 2021; 22(23), 12662.
J Li, C Fan, H Pei, J Shi and Q Huang. Smart drug delivery nanocarriers with self-assembled DNA nanostructures. Advanced Materials 2013; 25(32), 4386-4396.
P Horcajada, R Gref, T Baati, PK Allan, G Maurin, P Couvreur, G Férey, RE Morris and C Serre. Metal-organic frameworks in biomedicine. Chemical Reviews 2012; 112(2), 1232-1268.
JS Suk, Q Xu, N Kim, J Hanes and LM Ensign. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Advanced Drug Delivery Reviews 2016; 99, 28-51.
PC Ke, S Lin, WJ Parak, TP Davis and F Caruso. A decade of the protein corona. ACS Nano 2017; 11(12), 11773-11776.
B Ghaemi and MJ Hajipour. Tumor acidic environment directs nanoparticle impacts on cancer cells. Journal of Colloid and Interface Science 2023; 634, 684-692.
Y Kato, S Ozawa, C Miyamoto, Y Maehata, A Suzuki, T Maeda and Y Baba. Acidic extracellular microenvironment and cancer. Cancer Cell International 2013; 13, 1-8.
S Ghaeini-Hesaroeiye, HR Bagtash, S Boddohi, E Vasheghani-Farahani and E Jabbari. Thermoresponsive nanogels based on different polymeric moieties for biomedical applications. Gels 2020; 6(3), 20.
X Mou, Z Ali, S Li and N He. Applications of magnetic nanoparticles in targeted drug delivery system. Journal of Nanoscience and Nanotechnology 2015; 15(1), 54-62.
P Mi. Stimuli-responsive nanocarriers for drug delivery, tumor imaging, therapy and theranostics. Theranostics 2020; 10(10), 4557.
W Wang, F Zhong, D Wang, Y Zhao, D Peng, S Li, Q Ning, S Tang, CY Yu and H Wei. Dual gatekeepers-modified mesoporous organic silica nanoparticles for synergistic photothermal-chemotherapy of breast cancer. Journal of Colloid and Interface Science 2023; 646, 118-128.
A Babu, A Munshi and R Ramesh. Combinatorial therapeutic approaches with RNAi and anticancer drugs using nanodrug delivery systems. Drug Development and Industrial Pharmacy 2017; 43(9), 1391-1401.
Y Jiang, N Krishnan, J Heo, RH Fang and L Zhang. Nanoparticle-hydrogel superstructures for biomedical applications. Journal of Controlled Release 2020; 324, 505-521.
M Wacker, K Chen, A Preuss, K Possemeyer, B Roeder and K Langer. Photosensitizer loaded HSA nanoparticles. I: Preparation and photophysical properties. International Journal of Pharmaceutics 2010; 393(1-2), 254-263.
JF Liu, B Jang, D Issadore and A Tsourkas. Use of magnetic fields and nanoparticles to trigger drug release and improve tumor targeting. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2019; 11(6), e1571.
D Bobo, KJ Robinson, J Islam, KJ Thurecht and SR Corrie. Nanoparticle-based medicines: A review of FDA-approved materials and clinical trials to date. Pharmaceutical Research 2016; 33, 2373-2387.
CL Ventola. Progress in nanomedicine: Approved and investigational nanodrugs. Pharmacy and Therapeutics 2017; 42(12), 742-755.
AAH Abdellatif and AF Alsowinea. Alsowinea and F Abdullah. Approved and marketed nanoparticles for disease targeting and applications in COVID-19. Nanotechnology Reviews 2021; 10(1), 1941-1977.
Y Liu, G Yang, Y Hui, S Ranaweera and CX Zhao. Microfluidic nanoparticles for drug delivery. Small 2022; 18, 2106580.
BG Helena, S Cláudia, PS Sérgio and SR Marco. Quality by design in pharmaceutical manufacturing: A systematic review of current status, challenges and future perspectives. European Journal of Pharmaceutics and Biopharmaceutics 2020; 147, 19-37.
HS Leong, KS Butler, CJ Brinker, M Azzawi, S Conlan, C Dufés, A Owen, S Rannard, C Scott, C Chen, MA Dobrovolskaia, SV Kozlov, A Prina-Mello, R Schmid, P Wick, F Caputo, P Boisseau, RM Crist, SE McNeil, …, C Pastore. On the issue of transparency and reproducibility in nanomedicine. Nature Nanotechnology 2019; 4(7), 629-635.
H Lu, S Zhang, J Wang and QA Chen. A review on polymer and lipid-based nanocarriers and its application to nano-pharmaceutical and food-based systems. Frontiers in Nutrition 2021; 1(8), 783831.
Nanotechnology Characterization Laboratory. Assay cascade protocols for nanomedicine characterization. Nanotechnology Characterization Laboratory, Frederick, United States, 2023.
CC Hu, X He, H Gao and J Zhang. DELIVER: The core principles for the clinic translation of nanomedicines. Acta Pharmaceutica Sinica B 2025; 15(2), 1196-1198.
B Sandoval. Perspectives on FDA’s regulation of nanotechnology: Emerging challenges and potential solutions. Comprehensive Reviews in Food Science and Food Safety 2009; 8, 375-393.
V Fanny and V Christine. Practical guidelines for the characterization and quality control of nanoparticles in the pharmaceutical industry. In: JK Patel and YV Pathak (Eds). Emerging technologies for nanoparticle manufacturing. Springer, Cham, Switzerland, 2021, p. 487-508.
ICH Expert Working Group. CH Q13: Continuous manufacturing of drug substances and drug products. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 2023; 54(3), 244-247.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






