An Evaluation of the Alteration in the Number of Neuron and Expression of S100B, MMP-9, and GFAP in Ischemic Rat Brain Tissue Induced by the Left Unilateral Common Carotid Artery Occlusion Method
DOI:
https://doi.org/10.48048/tis.2025.10251Keywords:
Neuron, GFAP, Neuroinflammation, Ischemic stroke model, Common carotid artery occlusionAbstract
Background: The middle cerebral artery occlusion (MCAO) model has been widely used as a tool for learning the focal ischemic stroke. However, the MCAO method has several limitations, including a lack of visibility and risk of subarachnoid hemorrhage. In addition, Common carotid artery (CCA) occlusion is another form of ischemic stroke type in human with a prevalence ranging from approximately 0.24 to 5 % among in ischemic stroke patients. Thus, CCA occlusion method can be developed as an alternative for creating ischemic stroke models to learn pathophysiology of ischemic stroke. Aim: The aim of this study is to prove whether an occlusion of the left CCA for 180 min in rat can induce ischemic stroke based on the change number of neuron and biomarker in brain tissue. Methods: The 15 male Wistar rats were randomly divided in to 3 groups. The first and second 5 rats, each of them were occluded on the left CCA for 60 and 180 min, then were categorized as the Group A and Group B respectively. Meanwhile, the remaining 5 rats without occlusion were included in the sham group. The assessment of the motoric dysfunction used LRWT tool to evaluate foot fault scoring (FFS) and foot placement accuracy, while the number and distribution of neuron was evaluated by H and E staining. The expression of MMP9 and S100B were assessed using Elisa technique, while GFAP was evaluated with immunohistochemistry technique. Results: The number of neuron in ischemic stroke groups (A and B) was significantly decreased compared to the sham group. Meanwhile, Expression of MMP9, S100B, GFAP was found to increase significantly in ischemic groups (A and B). Motoric function was found to decrease in ischemic stroke group (A and B) both in RHL and RFL. Conclusions: The occlusion of left unilateral CCA for 180 min yield in the declining number of the neurons, motoric deficit and in increasing of MMP9, S100B, GFAP expression. This finding was expected to give a new venue to develop preclinical research to learn pathophysiology of ischemic stroke.
HIGHLIGHTS
- 180-min left CCA occlusion induces brain ischemia.
- Ladder Rung Walking Test confirms movement impairment in ischemic stroke.
- Neuron loss indicates stroke impact on brain tissue.
- Elevated MMP9, S100B, GFAP show neuroinflammation and astrocyte damage indicated brain damage.
- Findings support the use of this model for studying ischemic stroke pathophysiology.
GRAPHICAL ABSTRACT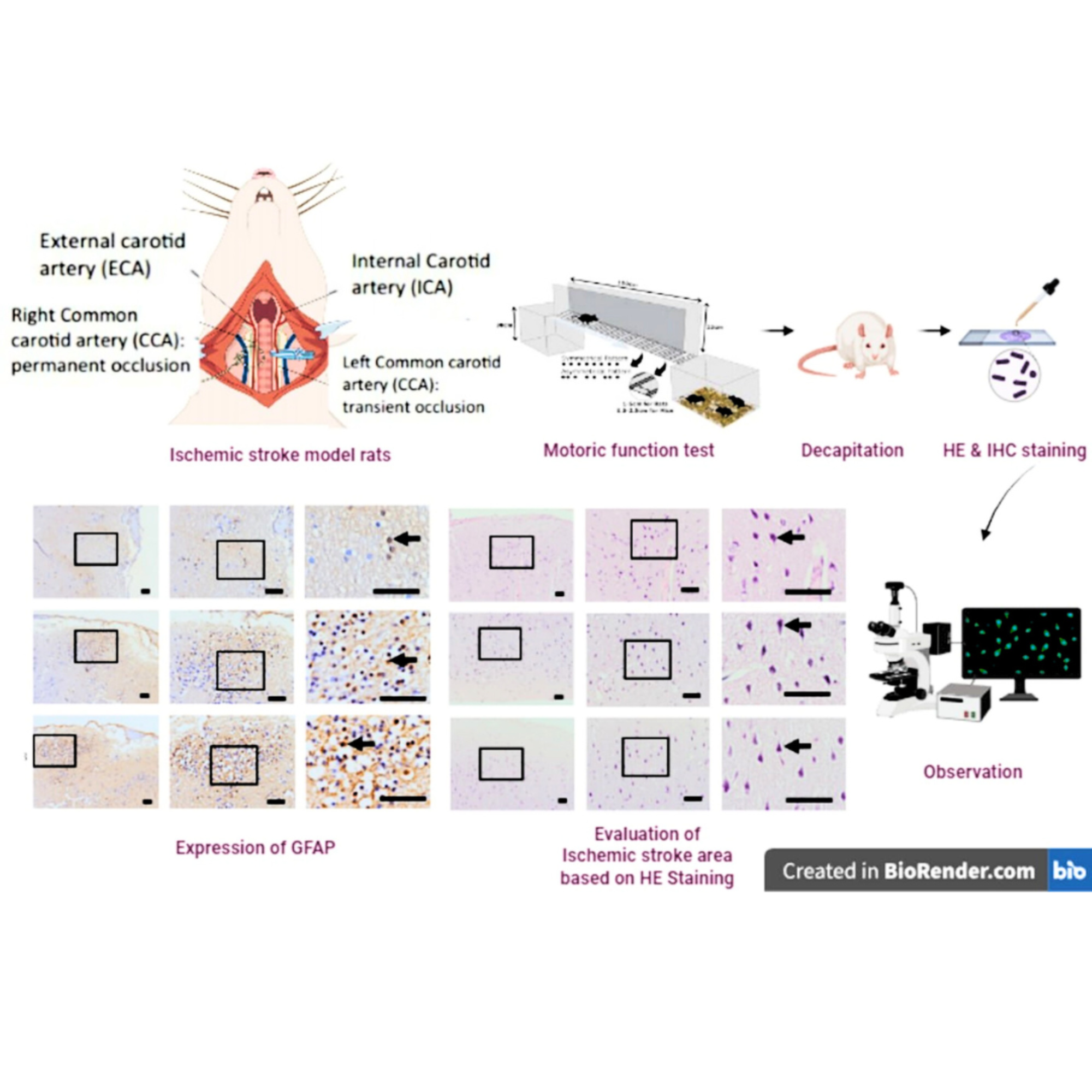
Downloads
References
L Pu, L Wang, R Zhang, T Zhao, Y Jiang and L Han. Projected global trends in ischemic stroke incidence, deaths, and disability-adjusted life years from 2020 to 2030. Stroke 2023; 54(5), 1330-1339.
L Zeng, S Hu, L Zeng, R Chen, H Li, J Yu and H Yang. Animal models of ischemic stroke with different forms of middle cerebral artery occlusion. Brain Sciences 2023; 13(7), 1007.
J Zhao, X Zhang, L Dong, Y Wen and L Cui. The many roles of statins in ischemic stroke. Current Neuropharmacolgy 2014; 12(6), 564-574.
F Fluri, MK Schuhmann and C Kleinschnitz. Animal models of ischemic stroke and their application in clinical research. Drug Design, Development and Therapy 2015; 9, 3445-3454.
IM Macrae. Translational neuropharmacology—using appropriate animal models to guide clinical drug development: preclinical stroke research—advantages and disadvantages of the most common rodent models of focal ischemia. British Journal of Pharmacology 2012; 164(4), 1062-1078.
M Rozanski and HJ Audebert. Glial fibrillary acidic protein in acute stroke: What we know and what we need to know. AME Medical Journal 2018; 3, 14.
Y Huang, Z Wang, Z Huang and Z Liu. Biomarkers and the outcomes of ischemic stroke. Frontiers in Molecular Neuroscience 2023; 16, 1006789.
P Khandare, A Saluja, RS Solanki, R Singh, K Vani, D Garg and RK Dhamija. Serum S100B and NSE levels correlate with infarct size and bladder-bowel involvement among acute ischemic stroke patients. Journal of Neurosciences in Rural Practice 2022; 13(2), 218-225.
AH Katsanos, K Makris, D Stefani, K Koniari, E Gialouri, M Lelekis, M Chondrogianni, C Zompola, E Dardiotis, I Rizos, J Parissis, E Boutati, K Voumvourakis and G Tsivgoulis. Plasma glial fibrillary acidic protein in the differential diagnosis of intracerebral hemorrhage. Stroke 2017; 48(9), 2586-2588.
SM Desai, M Rocha, TG Jovin and AP Jadhav. High variability in neuronal loss: Time is brain, requantified. Stroke 2019; 50(1), 34-37.
A Szymankiewicz-Szukala, J Huber, P Czarnecki, A Wiertel-Krawczuka and M Dabrowski. Temporary occlusion of common carotid arteries does not evoke total inhibition in the activity of corticospinal tract neurons in experimental conditions. Biomedicines 2023; 11(5), 1425.
P Cowled and R Fitridge. Pathophysiology of reperfusion injury. University of Adelaide Press, South Australia, Australia, 2020.
LA Mentari, R Naufalina, M Rahmadi and J Khotib. Development of ischemic stroke model by right unilateral common carotid artery occlusion (RUCCAO) Method. Folia Medica Indonesiana 2018; 54(3), 200-206.
A Machin, DA Purwanto, Nasronuddin, P Sugianto, AN Hidayati and B Utomo. Camellia sinensis with its active compound EGCG can decrease necroptosis via inhibition of HO-1 expression. EurAsian Journal of BioSciences 2020; 14, 1813-1820.
I Antonow-Schlorke, J Ehrhardt and M Knieling. Modification of the ladder rung walking task-new options for analysis of skilled movements. Stroke Research and Treatment 2013; 2013(1), 418627.
LA Martins, A Schiavo, LL Xavier and RG Mestriner. The foot fault scoring system to assess skilled walking in rodents: A reliability study. Frontiers in Behavioral Neuroscience 2022; 16, 892010.
GA Metz and IQ Whishaw. The ladder rung walking task: A scoring system and its practical application. Journal of Visualized Experiments 2009; 28, 1204.
LF Romao, VO De Sousa, VM Neto and FCA Gomes. Glutamate activates GFAP gene promoter from cultured astrocytes through TGF-β1 pathways. Journal of Neurochemistry 2008; 106(2), 746-756.
S Xu, J Lu, A Shao, JH Zhang and J Zhang. Glial cells: Role of the immune response in ischemic stroke. Frontiers in Immunology 2020; 11, 294.
M Nour, F Scalzo and DS Liebeskind. Ischemia-reperfusion injury in stroke. Interventional Neurology 2012; 1(3-4), 185-199.
GA Metz and IQ Whishaw. Cortical and subcortical lesions impair skilled walking in the ladder rung walking test: a new task to evaluate fore- and hindlimb stepping, placing, and coordination. Journal of Neuroscience Methods 2002; 115(2), 169-179.
M Riek-Burchardt, P Henrich-Noack, GA Metz and KG Reymann. Detection of chronic sensorimotor impairments in the ladder rung walking task in rats with endothelin-1-induced mild focal ischemia. Journal of Neuroscience Methods 2004; 13(2), 227-133.
J Zielinska-Turek, M Dorobek, G Turek, J Dabrowski, A Ziemba, P Andziak and M Barcikowska-Kotowicz. MMP-9, TIMP-1 and S100B protein as markers of ischemic stroke in patients after carotid artery endarterectomy. Polish medical journal 2022; 50(297), 177-182.
Y Lu, R Wang, H Huang, H Qin, C Liu, Y Xiang, C Wang, H Luo, J Wang, Y Lan and Y Wei. Association of S100B polymorphisms and serum S100B with risk of ischemic stroke in a Chinese population. Scientific Reports 2018; 8(1), 971.
R Mao, N Zong, Y Hu, Y Chen and Y Xu. Neuronal death mechanisms and therapeutic strategy in ischemic stroke. Neuroscience Bulletin 2022; 38(10), 1229-1247.
GC Brown. Neuronal loss after stroke due to microglial phagocytosis of stressed neurons. International Journal of Molecular Sciences 2021; 22(24), 13442.
E Sekerdag, I Solaroglu and Y Gursoy-Ozdemir. Cell death mechanisms in stroke and novel molecular and cellular treatment options. Current Neuropharmacology 2018; 16(9), 1396-1315.
L Amalia. Glial fibrillary acidic protein (GFAP): Neuroinflammation biomarker in acute ischemic stroke. Journal of Inflammation Research 2021; 14, 7501-7506.
T Yang, YJ Dai, G Chen and S Cui. Dissecting the dual role of the glial scar and scar-forming astrocytes in spinal cord injury. Frontiers in Cellular Neuroscience 2020; 14, 78
M Herrmann, P Vos, MT Wunderlich, CHMM De Bruijn and KJB Lamers. Release of Glial Tissue-Specific Proteins After Acute Stroke: A Comparative Analysis of Serum Concentrations of Protein S-100B and Glial Fibrillary Acidic Protein. Stroke 2000; 31(11), 2670-2677.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






