In vitro Antimicrobial and NO Inhibitory Activities of A Thai Herbal Recipe Against Cutibacterium acnes, Staphylococcus aureus and Staphylococcus epidermidis
DOI:
https://doi.org/10.48048/tis.2025.9989Keywords:
Antimicrobial, NO inhibitory, Cutibacterium acnes, Staphylococcus aureus, Staphylococcus epidermidis, Traditional Thai medicineAbstract
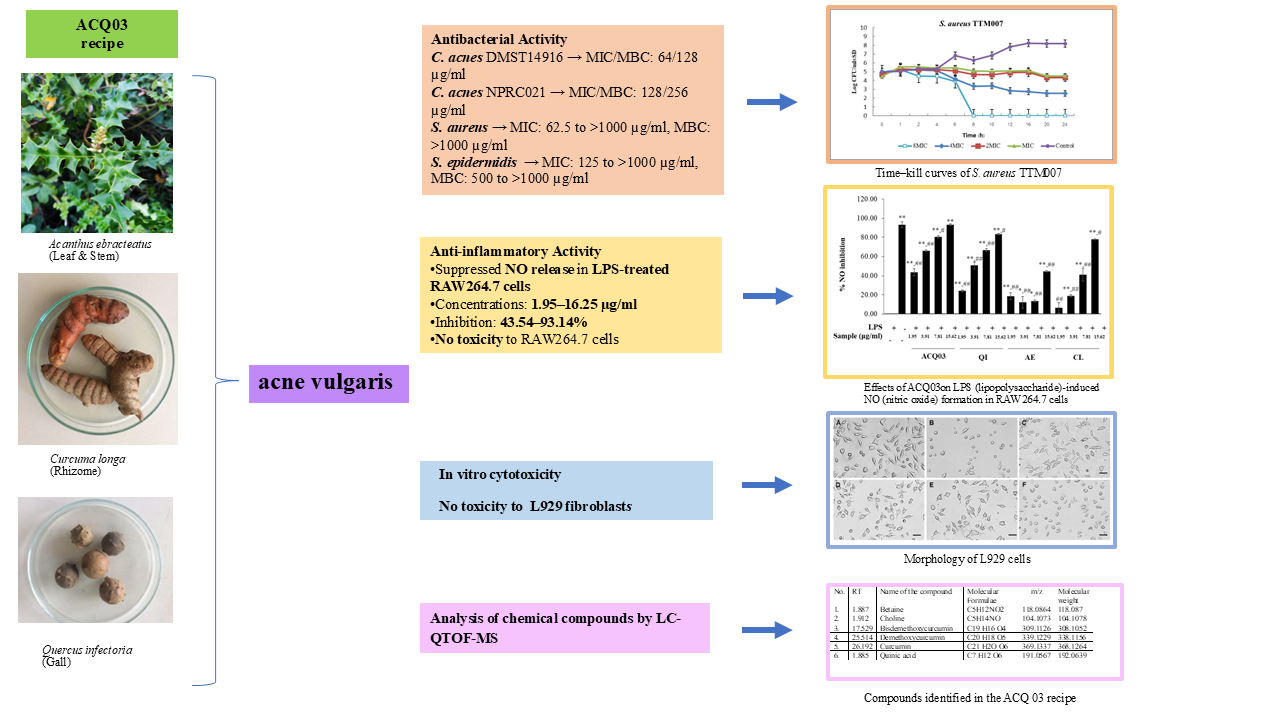
Introduction: Acanthus ebracteatus (AE), Curcuma longa (CL) (rhizome), (leaf and stem), and Quercus infectoria (QI) (gall) (ACQ03 recipe) have long been used to treat skin and inflammatory conditions. In addition, it has been used in traditional Thai medicine hospitals to treat acne. This study is aimed to evaluate the antibacterial and anti-inflammatory effects of ACQ03 recipe. Materials and methods: ACQ03 recipe was evaluated antibacterial using broth microdilution, a time-kill assay and nitric oxide inhibitory effects and cytotoxicity activities. The ingredients that make up the ethanol extract in the ACQ03 formulation were identified by LC-QTOF-MS method. Results and discussion: Antibacterial activity of ACQ03 against C. acnes DMST14916 had good results, with MIC/MBC values of 64/128 (µg/mL); for C. acnes NPRC021, with MIC/MBC values of 128/256 (µg/mL); against 20 strains of S. aureus, MICs ranging from 62.5 to > 1,000 and MBC values > 1,000 (µg/mL); of S. epidermidis had MIC values ranging from 125 to > 1,000 and MBC values ranging from 500 to > 1,000 µg/mL. NO release in LPS-treated RAW264.7 cells was strongly suppressed by the ACQ03 recipe ethanol extract at concentrations ranging from 1.95 to 16.25 µg/mL, with inhibition values ranging from 43.54 to 93.14 % without cytotoxicity to RAW264.7 cells or L929 cells. Conclusion: ACQ03 recipe could reduce antibacterial and anti-inflammatory effect. It might be suggested that this ACQ03 recipe could be useful for the treatment of acne.
HIGHLIGHTS
- Antibacterial activity of ACQ03 against C. acnes DMST14916 had good results, with MIC/MBC values of 64/128 (µg/ml); for C. acnes NPRC021, with MIC/MBC values of 128/256 (µg/ml); against 20 strains of S. aureus.
- NO release in LPS-treated RAW264.7 cells was strongly suppressed by the ACQ03 recipe ethanol extract at concentrations ranging from 1.95 to 16.25 µg/ml, with inhibition values ranging from 43.54 to 93.14 % without cytotoxicity to RAW264.7 cells or L929 cells
- Quadrupole time‒of-flight LC‒MS was used to analyze the chemical constituents of the ethanol extract from the ACQ03 recipe
GRAPHICAL ABSTRACT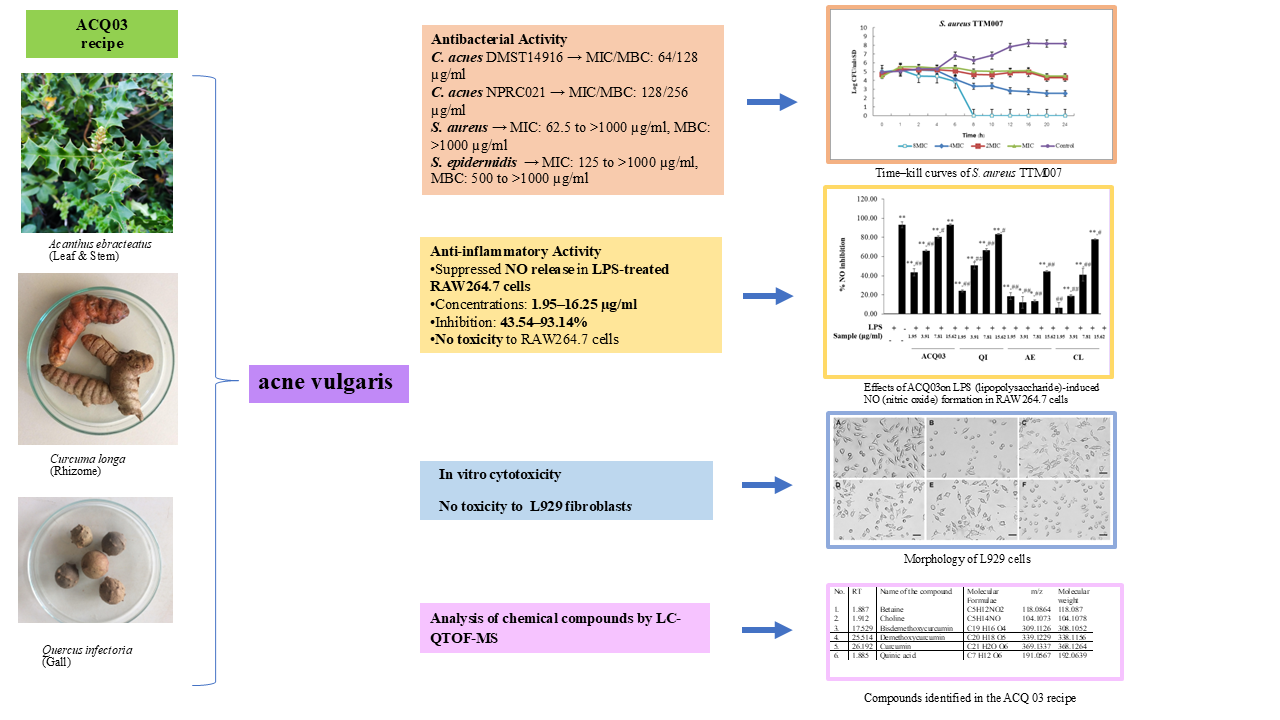
Downloads
References
C Feidenhansl, M Lund, A Poehlein, R Lood, HB Lomholt and H Brüggemann. Cutibacterium and Staphylococcus dysbiosis of the skin microbiome in acne and its decline after isotretinoin treatment. JEADV Clinical Practice 2024; 3(5), 1454-1466.
HJ Kim and YH Kim. Exploring acne treatments: From pathophysiological mechanisms to emerging therapies. International Journal of Molecular Sciences 2024; 25(10), 5302.
LH Kircik. Individual article: Vehicles matter. Journal of Drugs in Dermatology 2024; 23(2), SF378969s3.
K Eawsakul and K Bunluepuech. Exploring synergistic inhibition of inflammatory and antioxidant potential: Integrated in silico and in vitro analyses of Garcinia mangostana, Curcuma comosa, and Acanthus ebracteatus. Advances in Pharmacological and Pharmaceutical Sciences 2024; 2024(1), 8584015.
W Wutthithammavet. Thai traditional medicine. Odean Store Press, Bangkok, Thailand, 1997.
R Bora, PP Adhikari, AK Das, N Raaman and GD Sharma. Ethnomedicinal, phytochemical, and pharmacological aspects of genus Acanthus. International Journal of Pharmacy and Pharmaceutical Sciences 2017; 9(12), 8.
K Larsen. Leguminosae-Caesalpinioideae. Flora of Thailand 1984; 4, 4.
F Vahdat-Lasemi, P Kesharwani and A Sahebkar. Curcumin as a pharmaceutical leader. In: P Kesharwani and A Sahebkar (Eds.). Curcumin-based nanomedicines as cancer therapeutics. Academic Press, London, 2024, p. 23-46.
M Cozmin, II Lungu, C Gutu, A Stefanache, LD Duceac, BD Șoltuzu, D Damir, G Calin, ERB Goroftei, C Grierosu and M Boev. Turmeric: From spice to cure. A review of the anti-cancer, radioprotective and anti-inflammatory effects of turmeric sourced compounds. Frontiers in Nutrition 2024; 11, 1399888.
T Ak and I Gülçin. Antioxidant and radical scavenging properties of curcumin. Chemico-Biological Interactions 2028; 174(1), 27-37.
S Llano, S Gómez, J Londoño and A Restrepo. Antioxidant activity of curcuminoids. Physical Chemistry Chemical Physics 2019; 21(7), 3752-3760.
B Kocaadam and N Şanlier. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Critical Reviews in Food Science and Nutrition 2017; 57(13), 2889-2895.
V Soleimani, A Sahebkar and H Hosseinzadeh. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances. Phytotherapy Research 2018; 32(6), 985-995.
S Shukla, K Sahu, P Sharma, A Joshi and D Bhatnagar. Assessment of the antioxidant activity of Quercus infectoria gall extract and its efficacy in wound healing using Optical Coherence Tomography in Swiss albino mice. European Journal of Biomedical 2018; 5(3), 367-376.
T Thammathirat. 2017, Antibacterial, tyrosinase inhibitory and antioxidant activities of Thai herbal formula (THF-AC003) used for acne and its application as soap. Master Thesis. Prince of Songkla University, Thailand.
Clinical and Laboratory Standards Institute. Methods for antimicrobial susceptibility testing of anaerobic bacteria, approved standard M11-A7. Clinical and Laboratory Standards Institute, Pennsylvania, 2007.
Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard in Clinical and Laboratory Standards Institute Document M07-A8. Clinical and Laboratory Standards Institute, Pennsylvania, 2009.
S Chusri and SP Voravuthikunchai. Quercus infectoria: A candidate for the control of methicillin‐resistant Staphylococcus aureus infections. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives 2008; 22(4), 560-562.
S Suriyaprom, P Srisai, V Intachaisri, T Kaewkod, J Pekkoh, M Desvaux and Y Tragoolpua. Antioxidant and anti-inflammatory activity on LPS-stimulated RAW 264.7 macrophage cells of white mulberry (Morus alba L.) leaf extracts. Molecules 2023; 28(11), 4395.
P Pooprommin, C Manaspon, A Dwivedi, A Mazumder, S Sangkaew, S Wanmasae, J Tangpongd, T Ongtanasupa and K Eawsakul. Alginate/pectin dressing with niosomal mangosteen extract for enhanced wound healing: Evaluating skin irritation by structure-activity relationship. Heliyon 2022; 8(12), e12032.
S Sangkaew, S Wanmasae, K Bunluepeuch, T Ongtanasup, S Srisang, C Manaspon, P Pooprommin and K Eawsakul. Development of nanoemulsions for wound dressings containing Cassia alata L. Leaf extraction. Evidence-Based Complementary and AlternativeMedicine 2022; 2022(1), 4282678.
KW Kim, YS Lee, D Yoon, GS Kim and DY Lee. The ethanolic extract of Curcuma longa grown in Korea exhibits anti-neuroinflammatory effects by activating of nuclear transcription factor erythroid-2-related factor 2/heme oxygenase-1 signaling pathway. BMC Complementary Medicine and Therapies 2022; 22(1), 343.
MY Um, KH Hwang, J Ahn and TY Ha. Curcumin attenuates diet‐induced hepatic steatosis by activating AMP‐activated protein kinase. Basic & Clinical Pharmacology & Toxicology 2013; 113(3), 152-157.
RL Thangapazham, S Sharad and RK Maheshwari. Skin regenerative potentials of curcumin. Biofactors 2013; 39(1), 141-149.
J Yahaufai, P Siripong and W Limpanasithikul. Immunomodulatory effect of Acanthus ebracteatus Vahl. Aqueous extract on macrophage function. Thai Cancer Journal 2011; 30(2), 59-67.
W Laupattarakasem, M Boonard, P Laupattarakasem and W Kosuwon. Tibial inlay technique using hamstring graft for posterior cruciate ligament reconstruction and remnant revision. Arthroscopy Techniques 2013; 2(1), e1-e7.
P Manop, P Keangin, N Nasongkla and K Eawsakul. In vitro experiments of microwave ablation in liver cancer cells (effects of microwave power and heating time). In: Proceedings of the 7th International Conference on Industrial Engineering and Applications (ICIEA), Bangkok, Thailand. 2020, p. 805-813.
HF Zhao, WD Jiang, Y Liu, J Jiang, P Wu, SY Kuang, L Tang, WN Tang, YA Zhang, XQ Zhou and L Feng. Dietary choline regulates antibacterial activity, inflammatory response and barrier function in the gills of grass carp (Ctenopharyngodon idella). Fish & Shellfish Immunology 2016; 52, 139-150.
HF Zhao, WD Jiang, Y Liu, J Jiang, P Wu, SY Kuang, L Tang, WN Tang, YA Zhang, XQ Zhou and L Feng. Betaine in inflammation: Mechanistic aspects and applications. Frontiers in Immunology 2018; 9, 1070.
SA Jang, DW Park, JE Kwon, HS Song, B Park, H Jeon, EH Sohn, HJ Koo and SC Kang. Quinic acid inhibits vascular inflammation in TNF-α-stimulated vascular smooth muscle cells. Biomedicine & Pharmacotherapy 2017; 96, 563-571.
Y Li, J Yao, C Han, J Yang, MT Chaudhry, S Wang, H Liu and Y Yin. Quercetin, inflammation and immunity. Nutrients 2016; 8(3), 167.
S Alam, N Singh, N Afaq and C Putatunda. Natural phenolics exhibit strong antimicrobial activity against clinical isolates obtained from patients of urinary tract infection. International Journal of Current Microbiology and Applied Sciences 2024; 13(3), 51-63.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






